Abstract
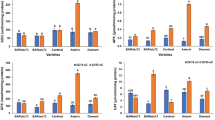
The ascorbate-glutathione system was studied during development and maturation of beech (Fagus sylvatica L.) seeds, the classification of which in the orthodox category is controversial. This study revealed an increase in glutathione content after acquisition of desiccation tolerance, which was more intensive in embryonic axes than in cotyledons. During seed maturation, the redox status of glutathione markedly changed toward the more reducing state, especially in cotyledons. Ascorbic acid content decreased during maturation, mostly in cotyledons. Activities of the enzymes of the ascorbate-glutathione cycle—ascorbate peroxidase (APX, EC 1.11.1.11), monodehydroascorbate reductase (MR, EC 1.6.5.4), dehydroascorbate reductase (DHAR, EC 1.8.5.1) and glutathione reductase (GR, EC 1.6.4.2)—were markedly higher in embryonic axes than in cotyledons throughout the study period. In the course of seed maturation, the activities of these enzymes decreased. Importance of the ascorbate-glutathione cycle in desiccation tolerance of beech seeds was discussed in relation to results for typical orthodox and recalcitrant seeds of other broadleaved species.





Similar content being viewed by others
References
Allen RD, Webb RP, Schake SA (1997) Use of transgenic plants to study antioxidant defenses. Free Radic Biol Med 23:473–479
Arrigoni O (1994) Ascorbate system in plant development. J Bioenerg Biomembr 26:407–419
Arrigoni O, De Gara L, Tommasi F, Liso R (1992) Changes in ascorbate system during seed development of Vicia faba L. Plant Physiol 99:235–238
Bailly C, El-Maarouf-Bouteau H, Corbineau F (2008) From intracellular signaling networks to cell death: the dual role of reactive oxygen species in seed physiology. C R Biol 331:806–814
Bonner FT (1990) Storage of seeds: potential and limitations for germplasm conservation. For Ecol Manage 35:35–43
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254
De Gara L, de Pinto MC, Moliterni VMC, D’Egidio MG (2003) Redox regulation and storage processes during maturation in kernels of Triticum durum. J Exp Bot 54:249–258
De Tullio MC, Arrigoni O (2003) The ascorbic acid system in seeds: to protect and to serve. Seed Sci Res 13:249–260
Desikan R, Hancock JT, Neill SJ (2005) Reactive oxygen species as signalling molecules. In: Smirnoff N (ed) Antioxidants and reactive oxygen species in plants. Blackwell Publishing, New York, pp 169–197
Dickie JB, Pritchard HW (2002) Systematic and evolutionary aspects of desiccation tolerance in seeds. In: Black M, Pritchard HW (eds) Desiccation and survival in plants. Drying without dying. CABI Publishing, pp 239–259
Esterbauer H, Grill D (1978) Seasonal variation of glutathione and glutathione reductase in needles of Picea abies. Plant Physiol 61:119–121
Foyer CH, Noctor G (2003) Redox sensing and signaling associated with reactive oxygen in chloroplast, peroxisomes and mitochondria. Physiol Plant 119:355–364
Foyer CH, Descouvrières P, Kunert KJ (1997) Protection against oxygen radicals: an important defence mechanism studied in transgenic plants. Plant Cell Environ 1717:507–523
Gosling P (1991) Beechnuts storage: a review and practical interpretation of the scientific literature. Forestry 64:51–59
Helmerhorst E, Stokes GB (1980) Microcentrifuge desalting: a rapid, quantitative method for desalting small amounts of protein. Anal Biochem 104:130–135
Kalemba E, Janowiak F, Pukacka S (2009) Desiccation tolerance acquisition in developing beech (Fagus sylvatica L.) seeds: the contribution of dehydrin-like protein. Trees 23:305–315
Kampfenkel K, VanMontagu M, Inzé D (1995) Extraction and determination of ascorbate and dehydroascorbate from plant tissue. Anal Biochem 225:165–167
Kermode AR, Finch Savage BE (2002) Desiccation sensitivity in orthodox and recalcitrant seeds in relation to development. In: Black M, Pritchard HW (eds) Desiccation and survival in plants. Drying without dying. CABI Publishing, pp 149–184
Kranner I, Birtić S (2005) A modulating role for antioxidants in desiccation tolerance. Integr Comp Biol 45:734–740
Kranner I, Grill D (1993) Content of low-molecular weight thiols during the imbibition of pea seeds. Physiol Plant 88:557–562
Kranner I, Birtić S, Anderson KM, Pritchard HW (2006) Glutathione half-cell reduction potential: a universal stress marker and modulator of programmed cell death? Free Radic Biol Med 40:2155–2165
León-Lobos P, Ellis RH (2002) Seed storage behaviour of Fagus sylvatica and Fagus crenata. Seed Sci Res 12:31–37
Liso R, Calabrese G, Bitonti MB, Arrigoni O (1984) Relationship between ascorbic acid and cell division. Exp Cell Res 150:314–320
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Nambara E, Marion-Poll A (2003) ABA action and interactions in seeds. Trends Plant Sci 8:213–217
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Oracz K, El-Maarouf-Bouteau H, Kranner I, Bogatek R, Corbineau F, Bailly C (2009) The mechanisms in seed dormancy alleviation by hydrogen cyanide unravel the role of reactive oxygen species as key factors of cellular signaling during germination. Plant Physiol 150:494–505
Paciolla C, De Tullio MC, Chiappetta A, Innocenti AM, Bitonti MB, Liso R, Arrigoni O (2001) Short- and long-term effects of dehydroascorbate on Lupinus albus and Allium cepa roots. Plant Cell Physiol 42:857–863
Pastori GM, Kiddle G, Antoniw J, Bernard S, Veljovic-Jovanovic S, Verrier PJ, Noctor G, Foyer CH (2003) Leaf vitamin C contents modulate plant defense transcripts and regulate genes that control development through hormone signaling. Plant Cell 15:939–951
Peng JR, Harberd NP (2002) The role of GA-mediated signalling in the control of seed germination. Curr Opin Plant Biol 5:376–381
Potters G, De Gara L, Asrad H, Horemans N (2002) Ascorbate and glutathione: guardians of the cell cycle, partners in crime? Plant Physiol Biochem 40:537–548
Poulsen KM (1993) Predicting the storage life of beech nuts. Seed Sci Technol 21:327–337
Poulsen KM, Knudsen H (1999) Viability constants based on eight years storage of beech nuts (Fagus sylvatica L.). Seed Sci Technol 27:1037–1039
Pukacka S, Ratajczak E (2006) Antioxidative response of ascorbate-glutathione pathway and metabolites to desiccation of recalcitrant Acer saccharinum seeds. J Plant Physiol 163:1259–1266
Pukacka S, Ratajczak E (2007a) Ascorbate and glutathione metabolism during development and desiccation of orthodox and recalcitrant seeds of the genus Acer. Funct Plant Biol 34:601–613
Pukacka S, Ratajczak E (2007b) Age-related biochemical changes during storage of beech (Fagus sylvatica L.) seeds. Seed Sci Res 17:45–53
Pukacka S, Wojkiewicz E (2003) The effect of temperature of drying on viability and some factors affecting storability of Fagus sylvatica seeds. Acta Physiol Plant 25:163–169
Pukacka S, Hoffmann SK, Goslar J, Pukacki PM, Wójkiewicz E (2003) Water and lipid relations in beech (Fagus sylvatica L.) seeds and its effect on storage behaviour. Biochim Biophys Acta 1621:48–56
Rodriguez-Gacio MD, Matilla AJ (2001) The last step of the ethylene biosynthesis pathway in turnip tops (Brassica rapa) seeds: alterations related to development and germination and its inhibition during desiccation. Physiol Plant 112:273–279
Sattler SE, Gilliland LU, Magallanes-Lundback M, Pollard M, Della Penna D (2004) Vitamin E is essential for seed longevity and for preventing lipid peroxidation during germination. Plant Cell 16:1419–1432
Schafer FQ, Buettner GR (2001) Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple. Free Radic Biol Med 30:1191–1212
Smith I (1985) Stimulation of glutathione synthesis in photorespiring plants by catalase inhibitors. Plant Physiol 79:1044–1047
Tommasi F, Paciolla C, Arrigoni O (1999) The ascorbate system in recalcitrant and orthodox seeds. Physiol Plant 105:193–198
Vertucci CW, Farrant JM (1995) Acquisition and loss of desiccation tolerance. In: Kigel J, Galili G (eds) Seed development and germination. Marcel Dekker, New York, pp 237–271
Zhang J, Kirkham MB (1996) Enzymatic response of the ascorbate-glutathione cycle to drought in sorghum and sunflower plants. Plant Sci 113:139–147
Acknowledgments
This study was supported by research funds of the Polish Ministry of Science and Higher Education.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pukacka, S., Ratajczak, E. Ascorbate and glutathione metabolism during development and desiccation of beech (Fagus sylvatica L.) seeds. Plant Growth Regul 62, 77–83 (2010). https://doi.org/10.1007/s10725-010-9492-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-010-9492-7