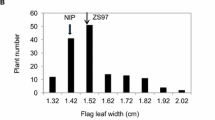
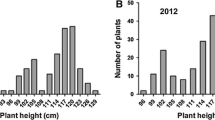
Abstract
Wild relatives of rice (Oryza sativa L.) represent enormous genetic diversity for many traits. Many domestication related traits found only in wild species that are lost from cultivated species during domestication. In this study, we report high-resolution mapping of two domestications related quantitative trait locus (QTL) controlling purple sheath color and plant height using high-density SNP based linkage map in an interspecific F2 population derived from a cross between O. nivara Sharma & Shastry and O. sativa. The segregation ratio of purple sheath color fit into 3:1 indicated that the presence of a dominant gene. The segregation of plant height trait exhibited a normal distribution in F2 and F2:3 generations. QTL analysis showed the identification of two QTLs controlling purple sheath color (designated as qPsh6.1 and qPsh6.2) on chromosome 6 explaining 27.12% and 14% phenotypic variance, respectively. The only plant height QTL (tentatively designated as qPh1.9) was mapped on chromosome 1 contributing 18.3% phenotypic variance. The qPsh6.1 covers 25.17 Kb region flanked by AX-95928721 and AX-95954573 containing five open reading frames (ORFs) while qPsh6.2 spanned in 118.2 Kb region containing 19 ORFs flanked by markers AX-95956260 and AX-95928295. The qPh1.9 gene controlling plant height was mapped between two SNP markers, AX-95944819 and AX-95964796 co-segregated with the locus LOC_Os01g66690 encoding for ZIP4/SPO22 protein. The detailed genetic study of the location of these genes will be useful for the map-based cloning.

Similar content being viewed by others
References
Ahmed N, Maekawa M, Utsugi S, Rikiishi K et al (2006) The wheat Rc gene for red coleoptile colour codes for a transcriptional activator of late anthocyanin biosynthesis genes. J Cereal Sci 44:54–58
Bharaj TS, Mangat GS, Kaur R, Singh K, Singh N (2014) PR122: a new high yielding variety of rice (Oryza sativa L.). J Res Punjab Agric Univ 51:204–205
Broadley M, Brown P, Cakmak I, Rengel Z, Zhao F (2012) Function of nutrients: micronutrients. In: Marschner P (ed) Mineral nutrition of higher plants, 3rd edn. Academic Press, London, pp 191–248
Chang TT, Li CC (1984) Semidwarfing genes in rice germplasm collection. Rice Genet Newsl 1:93–94
Chin HS, Wu YP, Hour AL, Hong CY, Lin YR (2016) Genetic and evolutionary analysis of purple leaf sheath in rice. Rice 9:8. https://doi.org/10.1186/s12284-016-0080-y
Choi JY, Platts AE, Fuller DQ, Hsing YI, Wing RA, Purugganan MD (2017) The rice paradox: multiple origins but single domestication in Asian Rice. Mol Biol Evol 34:969–979. https://doi.org/10.1093/molbev/msx049
Choudhury BI, Khan ML, Dayanandan S (2014) Patterns of nucleotide diversity and phenotypes of two domestication related genes (OsC1 and Wx) in indigenous rice varieties in Northeast India. BMC Genet 15:71. https://doi.org/10.1186/1471-2156-15-71
Chu Y, Xu N, Wu Q, Yu B, Li X, Chen R, Huang J (2019) Rice transcription factor OsMADS57 regulates plant height by modulating gibberellin catabolism. Rice 12:38. https://doi.org/10.1186/s12284-019-0298-6
Cone KC, Burr FA, Burr B (1986) Molecular analysis of the maize anthocyanin regulatory locus C1. Proc Natl Acad Sci USA 83:9631–9635. https://doi.org/10.1073/pnas.83.24.9631
de los Reyes BG (2019) Genomic and epigenomic bases of transgressive segregation—new breeding paradigm for novel plant phenotypes. Plant Sci 288:110213. https://doi.org/10.1016/j.plantsci.2019.110213
Edwards J, Stoltzfus D, Peterson PA (2001) The C1 locus in maize (Zea mays L.): Effect on gene expression. Theor Appl Genet 103:718–724. https://doi.org/10.1007/s001220100587
Fan FJ, Fan YY, Du JH, Zhuang JY (2008) Fine mapping of C (chromogen for anthocyanin) gene in rice. Rice Sci 15:1–6. https://doi.org/10.1016/S1672-6308(08)60012-8
Furukawa T, Maekawa M, Oki T et al (2007) The Rc and Rd genes are involved in proanthocyanidin synthesis in rice pericarp. Plant J 49:91–102. https://doi.org/10.1111/j.1365-313X.2006.02958.x
Gao D, He B, Zhou Y, Sun L (2011) Genetic and molecular analysis of a purple sheath somaclonal mutant in japonica rice. Plant Cell Rep 30:901–911. https://doi.org/10.1007/s00299-011-1004-3
Gao J, Dai G, Zhou W et al (2019) Mapping and identifying a candidate gene Plr4, a recessive gene regulating purple leaf in rice, by using bulked segregant and transcriptome analysis with next-generation sequencing. Int J Mol Sci 20:4335. https://doi.org/10.3390/ijms20184335
Gordeeva EI, Glagoleva AY, Kukoeva TV et al (2019) Purple-grained barley (Hordeum vulgare L.): marker-assisted development of NILs for investigating peculiarities of the anthocyanin biosynthesis regulatory network. BMC Plant Biol 19:52. https://doi.org/10.1186/s12870-019-1638-9
Hadagal BN, Manjunath A, Goud JV (1981) Linkage of genes for anthocyanin pigmentation in rice (Oryza sativa L.). Euphytica 30:747–754. https://doi.org/10.1007/BF00038804
Han Z, Hu W, Tan C, Xing Y (2017) QTLs for heading date and plant height under multiple environments in rice. Genetica 145:67–77. https://doi.org/10.1007/s10709-016-9946-6
Himi E, Nisar A, Noda K (2005) Colour genes (R and Rc) for grain and coleoptile upregulate flavonoid biosynthesis genes in wheat. Genome 48:747–754
Iorizzo M, Cavagnaro PF, Bostan H et al (2019) A cluster of MYB transcription factors regulates anthocyanin biosynthesis in carrot (Daucus carota L.) root and petiole. Front Plant Sci 9:1927. https://doi.org/10.3389/fpls.2018.01927
Ishimaru K, Ono K, Kashiwagi T (2004) Identification of a new gene controlling plant height in rice using the candidate-gene strategy. Planta 218:388–395. https://doi.org/10.1007/s00425-003-1119-z
Kadambari G, Vemireddy LR, Srividhya A et al (2018) QTL-Seq-based genetic analysis identifies a major genomic region governing dwarfness in rice (Oryza sativa L.). Plant Cell Rep 37:677–687. https://doi.org/10.1007/s00299-018-2260-2
Khlestkina EK, Röder MS, Börner A (2010) Mapping genes controlling anthocyanin pigmentation on the glume and pericarp in tetraploid wheat (Triticum durum L.). Euphytica 171:65–69. https://doi.org/10.1007/s10681-009-9994-4
Khush GS (1999) Green revolution: preparing for the 21st century. Genome 42:646–655
Kinoshita T (1995) Report of committee on gene symbolization, nomenclature and linkage groups. Rice Genet Newsl 12:9–153
Koide Y, Sakaguchi S, Uchiyama T et al (2019) Genetic properties responsible for the transgressive segregation of days to heading in rice. G3 Genes Genom Genet 9:1655–1662. https://doi.org/10.1534/g3.119.201011
Konishi S, Izawa T, Lin SY et al (2006) An SNP caused loss of seed shattering during rice domestication. Science 312:1392–1396. https://doi.org/10.1126/science.1126410
Kovach MJ, Sweeney MT, McCouch SR (2007) New insights into the history of rice domestication. Trends Genet 23:578–587. https://doi.org/10.1016/j.tig.2007.08.012
Kumar K, Sarao PS, Bhatia D et al (2018) High-resolution genetic mapping of a novel brown planthopper resistance locus, Bph34 in Oryza sativa L. X Oryza nivara (Sharma & Shastry) derived interspecific F2 population. Theor Appl Genet 131:1163–1171. https://doi.org/10.1007/s00122-018-3069-7
Lei L, Zheng HL, Wang JG et al (2018) Genetic dissection of rice (Oryza sativa L.) tiller, plant height, and grain yield based on QTL mapping and metaanalysis. Euphytica 214:109. https://doi.org/10.1007/s10681-018-2187-2
Li Z, Pinson SRM, Stansel JW, Park WD (1995) Identification of quantitative trait loci (QTLs) for heading date and plant height in cultivated rice (Oryza sativa L.). Theor Appl Genet 91:374–381. https://doi.org/10.1007/BF00220902
Li C, Zhou A, Sang T (2006) Rice domestication by reducing shattering. Science 311:1936–1938. https://doi.org/10.1126/science.1123604
Lin Z, Griffith ME, Li X et al (2007) Origin of seed shattering in rice (Oryza sativa L.). Planta 226:11–20. https://doi.org/10.1007/s00425-006-0460-4
Lin-Wang K, Bolitho K, Grafton K et al (2010) An R2R3 MYB transcription factor associated with regulation of the anthocyanin biosynthetic pathway in Rosaceae. BMC Plant Biol 10:50. https://doi.org/10.1186/1471-2229-10-50
Mao H, Sun S, Yao J et al (2010) Linking differential domain functions of the GS3 protein to natural variation of grain size in rice. Proc Natl Acad Sci USA 10:19579–19584. https://doi.org/10.1073/pnas.1014419107
Mooney BP (2009) The second green revolution? Production of plant-based biodegradable plastics. Biochem J 418:219–232. https://doi.org/10.1042/bj20081769
Oshima M, Taniguchi Y, Akasaka M, Abe K, Ichikawa H, Tabei Y, Tanaka J (2019) Development of a visible marker trait based on leaf sheath-specific anthocyanin pigmentation applicable to various genotypes in rice. Breed Sci 69:244–254. https://doi.org/10.1270/jsbbs.18151
Palme K, Li X, Teale WD (2014) Towards second green revolution: engineering nitrogen use efficiency. J Genet Genom 41:315–316. https://doi.org/10.1016/j.jgg.2014.05.003
Paz-Ares J, Ghosal D, Saedler H (1990) Molecular analysis of the C1-I allele from Zea mays: a dominant mutant of the regulatory C1 locus. EMBO J 9:315–321. https://doi.org/10.1002/j.1460-2075.1990.tb08113.x
Reddy VS, Goud KV, Sharma R, Reddy AR (1994) Ultraviolet-B-responsive anthocyanin production in a rice cultivar is associated with a specific phase of phenylalanine ammonia lyase biosynthesis. Plant Physiol 105:1059–1066. https://doi.org/10.1104/pp.105.4.1059
Reddy VS, Dash S, Reddy AR (1995) Anthocyanin pathway in rice (Oryza sativa L): identification of a mutant showing dominant inhibition of anthocyanins in leaf and accumulation of proanthocyanidins in pericarp. Theor Appl Genet 91:301–312. https://doi.org/10.1007/BF00220892
Reddy VS, Scheffler BE, Wienand U, Wessler SR, Reddy AR (1998) Cloning and characterization of the rice homologue of the maize C1 anthocyanin regulatory gene. Plant Mol Biol 36:497–498
Saitoh K, Onishi K, Mikami I, Thidar K, Sano Y (2004) Allelic diversification at the C (OsC1) locus of wild and cultivated rice: nucleotide changes associated with phenotypes. Genetics 168:997–1007. https://doi.org/10.1534/genetics.103.018390
Sakamoto W, Ohmori T, Kageyama K, Miyazaki C, Saito A, Murata M, Noda K, Maekawa M (2001) The Purple leaf (Pl) locus of rice: the Pl w allele has a complex organization and includes two genes encoding basic helix-loop-helix proteins involved in anthocyanin biosynthesis. Plant Cell Physiol 42:982–991. https://doi.org/10.1093/pcp/pce128
Sanchez AC, Khush GS (1994) Chromosomal location of some marker genes in rice using the primary trisomics. J Hered 85:297–300. https://doi.org/10.1093/oxfordjournals.jhered.a111461
Shomura A, Izawa T, Ebana K et al (2008) Deletion in a gene associated with grain size increased yields during rice domestication. Nat Genet 40:1023–1028. https://doi.org/10.1038/ng.169
Sugimoto K, Takeuchi Y, Ebana K et al (2010) Molecular cloning of Sdr4, a regulator involved in seed dormancy and domestication of rice. Proc Natl Acad Sci USA 107:5792–5797. https://doi.org/10.1073/pnas.0911965107
Sun X, Zhang Z, Chen C et al (2018) The C–S–A gene system regulates hull pigmentation and reveals evolution of anthocyanin biosynthesis pathway in rice. J Exp Bot 69:1485–1498. https://doi.org/10.1093/jxb/ery001
Sweeney MT, Thomson MJ, Pfeil BE, McCouch S (2006) Caught red-handed: Rc encodes a basic helix-loop-helix protein conditioning red pericarp in rice. Plant Cell 18:283–294. https://doi.org/10.1105/tpc.105.038430
Thomson MJ, de Ocampo M, Egdane J et al (2010) Characterizing the Saltol quantitative trait locus for salinity tolerance in rice. Rice 3:148–160. https://doi.org/10.1007/s12284-010-9053-8
Wang WY, Ding HF, Li GX, Jiang MS, Li RF, Liu X, Zhang Y, Yao FY (2009) Delimitation of the PSH1(t) gene for rice purple leaf sheath to a 23.5 kb DNA fragment. Genome 52:268–274. https://doi.org/10.1139/G08-121
Wang S, Wu K, Yuan Q et al (2012) Control of grain size, shape and quality by OsSPL16 in rice. Nat Genet 44:950–954. https://doi.org/10.1038/ng.2327
Wang S, Li S, Liu Q et al (2015) The OsSPL16-GW7 regulatory module determines grain shape and simultaneously improves rice yield and grain quality. Nat Genet 47:949–954. https://doi.org/10.1038/ng.3352
Wollenweber B, Porter JR, Lübberstedt T (2005) Need for multidisciplinary research towards a second green revolution. Curr Opin Plant Biol 8:337–341. https://doi.org/10.1016/j.pbi.2005.03.001
Yamaji N, Xia J, Mitani-Ueno N, Yokosho K, Feng Ma J (2013) Preferential delivery of zinc to developing tissues in rice is mediated by P-type heavy metal ATPase OsHMA2. Plant Physiol 162:927–939. https://doi.org/10.1104/pp.113.216564
Yue B, Cui K, Yu S, Xue W, Luo L, Xing Y (2006) Molecular marker-assisted dissection of quantitative trait loci for seven morphological traits in rice (Oryza sativa L.). Euphytica 150:131–139. https://doi.org/10.1007/s10681-006-9101-z
Zhang XW, Jiang QT, Wei YM, Liu C (2017a) Inheritance analysis and mapping of quantitative trait loci (QTL) controlling individual anthocyanin compounds in purple barley (Hordeum vulgare L.) grains. PLoS One 12:e0183704. https://doi.org/10.1371/journal.pone.0183704
Zhang Y, Yu C, Lin J, Liu J, Liu B, Wang J, Huang A, Li H, Zhao T (2017b) OsMPH1 regulates plant height and improves grain yield in rice. PLoS ONE 12:e0180825. https://doi.org/10.1371/journal.pone.0180825
Zhao S, Wang C, Ma J et al (2016) Map-based cloning and functional analysis of the chromogen gene C in rice (Oryza sativa L.). J Plant Biol 59:496–505. https://doi.org/10.1007/s12374-016-0227-9
Zheng J, Wu H, Zhu H et al (2019) Determining factors, regulation system, and domestication of anthocyanin biosynthesis in rice leaves. New Phytol 223:705–721. https://doi.org/10.1111/nph.15807
Zhou L, Liu S, Wu W et al (2016) Dissection of genetic architecture of rice plant height and heading date by multiple-strategy-based association studies. Sci Rep 6:29718. https://doi.org/10.1038/srep29718
Acknowledgements
The authors are thankful to the Punjab Agricultural University, Ludhiana, Punjab, India for extending field and laboratory facility.
Funding
Funding was provided by Indian Council of Agricultural Research (Grant No. 10(9)2011-EPD).
Author information
Authors and Affiliations
Contributions
KS and KN designed the experiments. KK and KN conducted the experiments, analyzes the data and wrote the manuscript. DB, RK, RK, GSM and YV involved in maintaining mapping population. KN reviewed the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumar, K., Neelam, K., Bhatia, D. et al. High resolution genetic mapping and identification of a candidate gene(s) for the purple sheath color and plant height in an interspecific F2 population derived from Oryza nivara Sharma & Shastry × Oryza sativa L. cross. Genet Resour Crop Evol 67, 97–105 (2020). https://doi.org/10.1007/s10722-019-00869-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-019-00869-4