Abstract
Crop wild relatives (CWR) are wild species that are more or less genetically related to crops that can be used to introgress useful genes for improvement of productivity, resistance to biotic and abiotic stresses and quality of cultivated crops. They are important in crop improvement to achieve food security for an increasing population and to overcome the challenges caused by climate change and the new virulence of major diseases and pests. These genetic resources are increasingly threatened in their natural habitats through over-exploitation and land reclamation and degradation. Therefore, their efficient and effective conservation would be taxonomically and genetically valuable and will contribute to maintaining and promoting the sustainability of crop diversity, facilitating agricultural production and supporting the increasing demand for food, feed and natural resources. A checklist of 5780 Crop Wild Relatives (CWR) taxa from North Africa was obtained using the CWR Catalogue for Europe and the Mediterranean (PGR Forum). Of which consists 76% of the flora of North Africa. The checklist contains 5588 (~97%) native taxa and 192 introduced. Families with higher taxa richness are Fabaceae, Asteraceae, and Poaceae. These three families constitute more than 33% of the total taxa included in the checklist. About 9% (502) CWR taxa identified as a priority for conservation in North Africa using four criteria, the economic value of the crop, the relatedness degree of wild relatives to their crop, threat status using IUCN red list assessment, and finally the centre of origin and/or diversity of the crop. Of these, 112 taxa were assigned high, 268 medium and 122 low priorities for effective conservation. Those assessed as threatened using IUCN Red list and national assessment represent approximately 2% (119 taxa) of the CWR in the region. However, 21 taxa are assessed as critically endangered (CR), 53 as endangered (EN), and 45 as vulnerable (VU). Wild relatives of some globally important crops are present, with those related to wheat (Triticum aestivum L. and T. durum L.) and barley (Hordeum vulgare L.) among the highest priority crops for the North Africa region. Amongst CWR assessed as threatened, only 8 (6.7%) CWR are related to food crops, Avena agadiriana B.R. Baum. et G. Fedak (VU), A. atlantica B.R. Baum et G. Fedak (VU), A. murphyi Ladiz. (EN), Beta macrocarpa Guss. (EN), Olea europaea subsp. maroccana Guss. (VU), Rorippa hayanica Maire (VU) and Aegilops bicornis (Forssk.) Jaub. et Spach (VU). The wild relative of Safflower Carthamus glaucus M. Bieb is restricted to Egypt and Libya and assessed as rare in Egypt. The information available about the conservation and threat status of CWR in North Africa still lags behind, and more investigations are required.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
North Africa is well-known as a hotspot of floristic richness and endemism (Myers et al. 2000; Cincotta et al. 2000), being part of the Mediterranean centre of diversity defined among the Vavilov centres of origin/diversity of cultivated crops (Vavilov 1926; Zeven and Zhukovsky 1975). Recent analysis has found it to be a centre of crop wild relative (CWR) diversity (Castañeda-Álvarez et al. 2016; Vincent et al. 2013). CWR are wild species genetically related to crops, including their progenitors. They are valuable genetic resources in developing new crop varieties capable of coping with the major biotic and abiotic stresses associated with climate change, because of the possibility to transfer these traits from the CWR to the related crop (Maxted et al. 2006). Several nationally and globally important crop genepools occur in the region, including many cereal plants, food legumes, vegetables, forage crops, fruit trees and nut crops (IUCN–EGA 1996–1998; Zehni 2006). However, currently CWR species and genetic diversity are threatened by genetic erosion and extinction due to desertification, drought, agricultural development, urbanization, and habitat destruction by overgrazing and forest clearing, plus the projected more considerably negative impact of climate change (Derneği 2010; El-Beltagy 2006; UNU-WIDER 2011). Thus there is an urgent need for systematic CWR conservation in North Africa to ensure their continued availability for contribution to the sustainable agricultural development and food security. In this context, as a crucial first step to achieving this goal, the aim of this paper is to create a regional CWR checklist, to prioritise the checklist and produce a CWR inventory for North Africa applying the methodology developed by Maxted et al. (2007).
Materials and methods
A preliminary CWR checklist for North Africa was extracted from CWR Catalogue of Europe and Mediterranean (http://www.pgrforum.org/) established by PGR Forum (Kell et al. 2005; Kell et al. 2008) for the five countries in North Africa. This initial checklist was then enhanced using information from USDA, ARS, Germplasm Resources Information Network (Gregory et al. 2009); the Harlan and de Wet Crop Wild Relative inventory (Vincent et al. 2013), the flora of Tunisia (Le Floch et al. 2010) and Flora of Libya (Jafri et al. 1976). The number of CWR in the checklist is relatively large, exceeding the available resources (human and financial) required for their conservation. Therefore, prioritising the checklist was necessary for an effective and efficient conservation. The prioritisation process used four criteria for establishing immediate conservation priorities: the relative economic value of the related crop, the actual or potential ease of use in crop breeding (i.e. Gene Pool and Taxon Group Concepts), relative threats using IUCN red list assessment and centre of origin and/or diversity of the crop. Several authors suggested these criteria as common criteria that could be used to prioritise CWR taxa and develop strategies for CWR conservation (e.g. Barazani et al. 2008; Ford-Lloyd et al. 2008; Kell et al. 2012; Maxted et al. 1997).
The related crop value was obtained from FAOSTAT (http://faostat.fao.org/site/567/default.aspx#ancor) for human food, animal forage and fodder, industrial and ornamental crops to establish the relative economic value of CWR for the five countries in North Africa. Crops were identified using the crop category then value attributed using: (i) crop production in tonnes, (ii) gross production value in the US $ and (iii) crop surface area of cultivation in hectares. The gross production value of the crop was estimated using a five-year period between 2009 and 2013. To establish relative crop/CWR relatedness Gene Pool concept (Harlan and de Wet 1971) and Taxon Group concept (Maxted et al. 2006) were used as a proxy for the CWR ease of use in breeding programmes. Primarily focusing on CWR taxa in the primary gene pool (GP1B) and secondary gene pool, the coenospecies (GP2), but also including more remote tertiary gene pool (GP3) taxa if they have previously been used in breeding. Where genepool information was unavailable the taxonomic hierarchy was used as a proxy and CWR taxa in taxon group one (TG1b), taxon group two (TG2), and taxon group three (TG3) of their related crop were prioritised. The global IUCN red list (IUCN 2016) was consulted along with national Red Lists to establish CWR threat assessment. Among the 5780 CWR in the CWR checklist for North Africa, 119 native taxa had been assessed as threatened using the latest red list criteria (IUCN 2001) and national assessments (IUCN 2016; Garzuglia 2006; Rankou et al. 2015), and these were prioritised. Wild relatives of crops that have North Africa as their centre of origin and/or diversity were also prioritised as they are well adapted to the region and possess desired features for crop improvement. Centre of origin and/or diversity was taken from Vavilov and Dorofeev (1992), Zeven and Zhukovsky (1975) and Vincent et al. (2013). CWR within the priority list were scored from one to ten for each used criterion to enable them to be ranked high, medium and low priority taxa (Table 1). When the CWR is related to more than one crop with different relatedness degree as in the case of Brassica complex, the CWR was assigned the score of the closest wild relative to the crop. A final priority score (FPS) was then assigned for each CWR by averaging the four individual prioritisation criterion scores. High priority was assigned for CWR taxa where FPS 5–10; medium priority where 2 ≤ FPS < 5 and low priority where FPS < 2.
Results and discussion
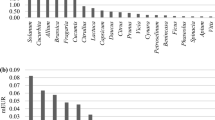
The North African CWR checklist included 5780 taxa belonging to 136 families, 765 genera, 4866 species and 914 infraspecific taxa (See Supplementary data, Table 2). The checklist consists of 5588 (~97%) native and 192 introduced taxa. Figure 1 shows families with the highest number of CWR taxa, Fabaceae, Asteraceae and Poaceae representing more than 33% of all taxa included in the checklist.
Using prioritisation criteria 502 CWR taxa belonging to 153 crop genera were identified (See Supplementary data, Table 3) with 112 CWR taxa assigned high, 268 medium and 122 low priorities. Families with a greater number of priority CWR taxa were Fabaceae (115), Poaceae (87) and Brassicaceae (82). Seven species introduced to the region are also included in the priority list. They are: Amaryllidaceae (Allium porrum L.), Brassicaceae (Camelina sativa (L.) Crantz and Diplotaxis tenuisiliqua Delile), Chenopodiaceae (Chenopodium urbicum L.), Fabaceae (Acacia farnesiana (L.) Willd.), Poaceae (Setaria italica (L.) P. Beauv.) and Solanaceae (Nicotiana glauca Graham). Table 2 shows a summary of priority CWR diversity.
The closest wild relatives to the crop (GP1b and TG1b) represent 17% (16.% GP1b and 1% TG1b) of the priority CWR list; these could be used in breeding programs easily as there are no hybridization barriers between them and their relative crops for gene introgression. Both GP2 and TG2 comprise a total of 98 taxa, 89 (18%) and 9 (2%) respectively. The more remote taxa GP3, TG3 and TG4 consist of 40% (200) of the priority taxa. They are of potential use as gene donors to improve cultivated crops and, thus, were included in the priority list. The degree of relatedness of the remaining priority CWR 112 (23%) is not confirmed yet; most of these taxa are threatened species (see Fig. 2). Although many of these CWR species have yet to have their crop breeding use confirmed, some have already been used to improve crop traits. Aegilops triaristata Willd. has been used to confer Hessian fly resistance on cultivated wheat (El Khlifi et al. 2004) and Pennisetum orientale Rich. has provided earliness, long inflorescence, leaf size and male fertility improvement to pearl millet (Dujardin and Hanna 1989). Medicago rugosa Desr. and Medicago scutellata (L.) Mill. have potential use in alfalfa weevil resistance, disease resistance and agronomic traits for Medicago sativa subsp. sativa and Medicago truncatula respectively (Mizukami et al. 2006; Tian and Rose 1999). The wild relatives Pistacia saportae Burnat., and Prunus padus L. are used as graft stock for their related crop Pistacia vera L. and Prunus cerasus L. respectively (USDA 2016).
The priority list contains CWR of several economically important crops for the region and worldwide. A number of wild gene pools of cereal crops are found in North Africa, particularly for wheat (Triticum L.), barley (Hordeum L.) and oat (Avena L.); this is also the case for food legumes such as pea (Pisum L.) and lupins (Lupinus L.). Many fruit crops are widely cultivated in the region along the coastal strip, such as citrus (Citrus L.), grapes (Vitis L.), olive (Olea L.), date (Phoenix L.), fig (Ficus L.), apple (Malus Mill.), pear (Pyrus L.), and plums and cherries (Prunus L.). Several cultivated vegetable crops have CWR within the North Africa region, notably lettuce (Lactuca L.), carrot (Daucus L.), cabbage and other brassicas (Brassica L.), beet (Beta L.), celery (Apium L.), asparagus (Asparagus Tourn. ex L.) and artichoke (Cynara L.). The region also is very rich in other condiments crops, aromatic plants, such as mustards (Sinapis L., Brassica L.), chives and leek (Allium L.). Forestry resources such as pine (Pinus L.), fir (Abies Mill.), fodder plants, clover (Trifolium L.), alfalfa (Medicago L.), are also distributed in the region. North Africa is a centre of origin and/or diversity for many of these crops (Supplementary data, Table 5).
Wheat (Triticum aestivum L. and Triticum durum L.) and barley (Hordeum vulgare L.) are among the crops with the highest value for North Africa in terms of their contribution to food and agriculture (see, ICARDA and UNEP 2000; Zehni 2006). Therefore, their wild relatives are of high priority for conservation. Wheat is the highest value crop in terms of cultivated area and production, with averages of 6,976,478.40 ha and 18,528,355.40 ton/year respectively (from 2009 to 2013) and is second after tomato regarding production value in US $ (Fig. 3). However, currently, wheat production in the region does not reach its full agricultural potential due to an erratic distribution of rain and the prevalence of many diseases and pests (ICARDA 2004). Twenty-four wild relatives of cultivated wheat (Triticum aestivum L.) occur in North Africa and are included in the prioritised inventory. They are represented by five genera, 15 taxa in the genus Aegilops, 2 in Agropyron Gaertn., 2 in Elymus L., 1 in Secale L. and 4 in Thinopyrum A. Löve. Ten (43%) out of 23 Aegilops species occur in the region; these are in the secondary gene pool. The rest are in the tertiary gene pool, Agropyron cristatum (L.) Beauv., Elymus repens (L.) Gould, Elymus repens (L.) Gould subsp. repens, Secale strictum (C. Presl) C. Presl, Thinopyrum elongatum (Host) D. R. Dewey, Thinopyrum junceum (L.) A. Love, Thinopyrum pycnanthum (Godr.) Barkworth, Thinopyrum scirpeum (C. Presl) D. R. Dewey. Taxa in the secondary gene pool are: Aegilops bicornis (Forssk.) Jaub. et Spach, Ae. bicornis (Forssk.) Jaub. et Spach var. anathera Eig, Ae bicornis (Forssk.) Jaub. et Spach var. bicornis, Ae. biuncialis Vis., Ae. geniculata Roth, Ae. kotschyi Boiss., Ae. longissima Schweinf. et Muschl., Ae. neglecta Req. ex Bertol., Ae. peregrina (Hack.) Maire et Weiller, Ae. peregrina (Hack.) Maire et Weiller var. brachyathera (Boiss.) Maire et Weiller, Ae. peregrina (Hack.) Maire et Weiller var. peregrina, Ae. triasistata Willd., Ae. triuncialis L., Ae. Triuncialis L. var. triuncialis, Ae. ventricosa Tausch, (see Supplementary data, Table 3).
Economic values of Northern Africa crops. The mean value of agriculture production (Int. $1000), production quantity and area harvested between 2009 and 2013 (FAO 2016)
Archaeological evidence shows that barley was the most dominant crop historically in North Africa (ICARDA 2004), where it was cultivated as human food and animal feed. Today it remains an important crop for food security and livelihood sustainability. The annual average production is 4,837,338.20 tons/year and average area harvested is 3,712,252.00 ha. The prioritised inventory includes five wild taxa in North Africa from the barley gene pool, one in the primary gene pool (GP1B) H. vulgare subsp. spontaneum (K. Koch) Thell., one in the secondary gene pool (GP2) H. bulbosum L., and three in the tertiary gene pool (GP3) H. secalinum Schreber, H. murinum L. and H. marinum Hudson. These wild relatives in GP1b and GP2 are valuable genetic resources contributing disease resistance and drought tolerance in barley cultivars (Nevo and Chen 2010; Lakew et al. 2011).
Olive trees are of socioeconomic importance and may be the most important agricultural oil crop in the Mediterranean region (Terzopoulos et al. 2005). Olive is the third highest crop regarding the value of production (2,573,984.06 US $) in North Africa; it has a wide distribution and covers 3,213,376.40 ha. Southern Europe, North Africa, and the Middle East are the source of about 95% of the world olive oil production (Hatzopoulos et al. 2002; Jain and Priyadarshan 2009). North Africa produces more than 11.5% of the world production of olive oil (FAO 2016). Tunisia is among the largest olive oil producers in the world. The primary gene pool (GP1) of Olea europaea L. includes both wild and cultivated forms. Olea europaea L. subsp. europaea L. var. sylvestris (Mill.) Lehr. (GP1B) and O. europaea L. subsp. europaea L. var. sativa (GP1A) that are genetically closely related (Sesli and Yegenoglu 2010; Brennan 2012). The secondary gene pool (GP2) is made up of sub-species O. europaea L. subsp. cuspidata (Wall. et G. Don) Cif, O. europaea L. subsp laperrinei (Batt et Trab.) Cif. and O. europaea subsp. maroccana (Greuter et Burdet) P. Vargas et al. (Brennan 2012). The primary gene pool (Olea europaea L. subsp. europaea L. var. sylvestris (Mill.) Lehr.) is used as a rootstock for grafting cultivated cultivars, and a gene source for improving resistance against changing environments and diseases (Sesli and Yegenoglu 2010).
Other important crops such as sugarcane (Saccharum officinarum L.) and sugar beet (Beta vulgaris L.) are cultivated mainly in Egypt and Morocco. Sugarcane has four wild relatives in the prioritised inventory, two in the secondary gene pool (GP2), S. spontaneum L. and its subsp. aegyptiacum (Willd) Hack., and two in the tertiary gene pool (GP3) S. ravennae (L.) L. and Imperata cylindrica (L.) P. Beauv. Beta vulgaris L. is represented in the region by B. vulgaris var. maritima (L.) Arcang., and B. macrocarpa Guss., both in the primary gene pool and by Patellifolia patellaris (Moq.) A.J. Scott et al. in the tertiary gene pool. The latter is a potential source of sugar beet nematode resistance (USDA 2016). Several fruit crops such as citrus (Citrus spp.), grape (Vitis vinifera L.), date (Phoenix dactylifera L.) and fig (Ficus carica L.) are widely cultivated all over the region from Morocco to Egypt. Date, as well as olive, has a long history of cultivation in the area and is of high cultural value. The production of dates and grapes are approximately 2,561,080.20 and 2,390,561.80 (MT/year), respectively. One wild relative taxon has been found for both date (Phoenix dactylifera L.), and grape (Vitis vinifera L.), P. humilis Royle (TG4) and V. vinifera L. var. sylvestris (C.C. Gmel.) Hegi (GP1B) respectively. Cultivated grapes V. vinifera L. is a primary gene pool for itself. North Africa is a centre of diversity for some vegetables such as onion, garlic, lettuce and safflower. Melons are a major summer crop. Pea, oat, rapeseed, carrot, brassica and carob are also economically important. The region is the centre of their origin and/or diversity, and a substantial number of their wild relatives are present and have been given the priority for conservation (see Supplementary data, Table 3 and 5).
In terms of forage and fodder crops, cultivated species in Lathyrus L., Medicago L., Trifolium L. and Vicia L. are of priority importance, both alfalfa (Medicago sativa L.) and clover (Trifolium repens L.) are widely cultivated in North Africa. The average production quantity of forage in Egypt alone is 26,490,000.0 tonnes/year over a five-year period (2009–2013). Egyptian clover or berseem (Trifolium alexandrinum L.) was domesticated in Egypt millennia ago and then spread to the west and south Asia, it is an essential crop for fodder and soil fertility maintenance, and is now cultivated extensively in irrigated cropping systems (Muhammad et al. 2014). The region is a centre of origin for these crops where a significant number of wild relatives are present (Supplementary data, Table 3 and 5).
The precise degree of threat for CWR in North Africa is unclear as there has been no specific red list assessment for CWR or wild plant species in the region. However, some globally assessed species are found in the region and other species have been assessed by national initiatives. The total number of threatened CWR using IUCN red list assessment and national assessment is 119 taxa (see supplementary data, Table 3), of which 21 critically endangered (CR), 53 endangered (EN) and 45 vulnerable (VU) (Fig. 4). These represent less than 2% of the CWR found in the region. Amongst the 119 threatened CWR, 21 are threatened at a global level, 14 (EN) and 7 (VU), all of which are native to North Africa. The information needed to assess further CWR threat status is largely unavailable, only 8 CWR related to food crops have been assessed as threatened, Avena agadiriana B.R. Baum et G. Fedak (VU), Avena atlantica B.R. Baum et G. Fedak (VU), Avena murphyi Ladiz. (EN), Beta macrocarpa Guss., (EN), Olea europaea L. subsp. maroccana (Greuter et Burdet) P. Vargas et al. (VU), Rorippa hayanica Maire (VU) at a national level and Aegilops bicornis (Forssk.) Jaub. et Spach (VU) at a global level. Carthamus glaucus M. Bieb. is restricted to Egypt and Libya and assessed as rare in Egypt (Radford et al. 2011). The lack of threat assessment is hampering conservation planning, and gathering of necessary data should be a future regional priority.
The highest number of threatened CWR is found in Morocco with 2 (CR) Lotus benoistii (Maire) Lassen. and Puccinellia distans (Jacq.) Parl. subsp. font-queri Maire, plus 7 (EN) and 12 (VU), flowed by Algeria with 4 (CR) Abies numidica de Lannoy ex Carrière, Epilobium numidicum Batt., Pulicaria filaginoides Pomel and Vicia fulgens Batt., and 3 (EN). The next highest threatened CWR was in Egypt with 2 (CR) Medemia argun (Martius) Wurtt. ex H.A. Wendl. and Primula boveana Decne ex Duby, 3 (EN) and 2 (VU); and finally one critically endangered (CR) Rumex tunetanus Barratte et Murb. ex Murb. in Tunisia. One vulnerable species Domasonium alisma Mill. occurs in all North African countries.
At the national level, a very recent conservation assessment and a Red listing of the endemic Moroccan flora (monocotyledons) was conducted (Rankou et al. 2015). The assessment comprises about 60 threatened taxa of which 16 species are CWR, 12 belong to the Family Poaceae, 3 to Amaryllidaceae and one to Xanthorrhoeaceae. Three are critically endangered (CR) Allium valdecallosum Maire et Weiller, Bromopsis maroccana (Pau et Font Quer) Holub and Vulpia litardiereana (Maire) A. Camus, 10 endangered (EN) and 3 vulnerable (VU) (see Supplementary data, Table 3). Some forest trees are also assessed as threatened at the national level. Six assessed as CR in Morocco and Tunisia Acacia tortilis (Forssk.) Galasso et Banfi., Acer monspessulanum L., Celtis australis L. Pyrus communis L. and Rhamnus serpyllacea L., 15 (EN) in Egypt, Morocco and Tunisia, and 13 (VU) in Algeria, Morocco and Tunisia (Garzuglia 2006).
The floristic diversity of Northern Africa countries is reflected in its CWR diversity. Morocco is the richest country in the region with 3,284 taxa (30%), followed by Algeria 2810 (26%), Tunisia 1792 (16%), Egypt 1673 (15%) and Libya 1392 (13%) (Fig. 5). Similarly, the geographic distribution of priority CWR in the region varies, some are native to just one country and others are native to two or more countries. Around 48.4% (243) of the priority taxa occur in more than one country while 13.3% (67) present in all countries. Some are native to individual countries 19% (96) in Morocco, 6% (30) in Algeria, 9.4% (47) in Egypt, 2.4% (12) in Libya and 1.4% (7) in Tunisia (Fig. 6). Approximately half of the 502 priority CWR is shared by the five Northern African countries. This highlights the need for establishing closer cooperation and coordination between these countries to conserve CWR diversity effectively and efficiently throughout the region.
All species in the secondary gene pool of cultivated wheat (Triticum aestivum L.) are distributed throughout the region, however, the gene pool three (GP3) Agropyron cristatum (L.) Gaetn. is present only in Morocco. Wild barley Hordeum vulgare L. subsp. spontaneum (C. Koch.) Thell., is present in Libya, Egypt and Morocco while the secondary gene pool (GP2) Hordeum bulbosum L. occurs in all countries except Egypt. The tertiary gene pool (GP3) Hordeum secalinum Schreber. is present only in Algeria while Hordeum marinum Hudson, and Hordeum murinum L., occur in the whole region. The cultivated olive Olea europaea L. subsp. europaea L. var. sativa is a primary gene pool (GP1A) of O. europea L., it is widely distributed in the region except in Egypt. All other wild relatives of the olive crop in North Africa are in the secondary gene pool (GP2), Olea europaea L. subsp. laperrinei (Batt et Trab.) Cif., in Algeria, Olea europaea L. subsp. cuspidata (Wall. et G. Don) Cif., in Egypt and Olea europaea L. subsp. maroccana (Greuter et Burdet) P. Vargas et al., in Morocco.
Conclusion
North Africa is a hotspot of plant diversity and endemism. Many areas of high plant richness are recognised in the region, for instance within, the Atlas Mountains in Morocco, the coastal strips of Morocco and Algeria, and Cyrenaica in Libya (Pons and Quézel 1985). It is also a hotspot of CWR diversity (Vincent et al. 2013) and here the taxa are well adapted to the extreme climatic conditions, such as high temperature, drought and high salinity. The unique geographic location of the North Africa as a transition zone from mesic to xeric habitats offers unique habitats for plant diversity (Derneği 2010). Growing under extreme conditions CWR native to this region are likely to have a wide array of adaptive importance to breeders for crop improvement, enabling crops to better survive the changing environment and climate change (Redden 2015). However, CWR conservation in North Africa is currently neglected and CWR use is undervalued. The information available on their diversity and threat status, required to develop a systematic conservation, is scarce. Therefore, one of the achievements of this study has been for the first time to identify the CWR diversity in the region and establish conservation priorities to help lay the foundations for future ex situ and in situ conservation, and subsequent use. The preliminary comprehensive CWR checklist of food and fodder species comprised 5780 taxa, 76% of the flora of North Africa, underlining that the region is a hotspot of CWR diversity, especially those related to food and fodder crops. Although 119 CWR priority taxa have been assessed as threatened using IUCN Red criteria, the number of threatened CWR taxa is thought to be an underestimate as there is no specific red listing activity in the region even though there is evidence of significant taxonomic and genetic erosion. This study will serve as a platform to establish and develop further conservation priorities for North Africa at regional and national levels and will form an exemplary to other countries in the wider region.
References
Barazani O, Perevolotsky A, Hadas R (2008) A problem of the rich: prioritizing local plant genetic resources for ex situ conservation in Israel. Biol Conserv 141(2):596–600
Brennan R (2012) Wild crop relatives: genomic and breeding resources. Temp Fruits Exp Agric 48(01):1–88
Castañeda-Álvarez NP, Khoury CK, Achicanoy HA, Bernau V, Dempewolf H, Eastwood RJ, Guarino L, Harker RH, Jarvis A, Maxted N, Müller JV, Ramirez-Villegas J, Sosa CC, Struik PC, Vincent H, Toll J (2016) Global conservation priorities for crop wild relatives. Nat Plants 2:16022
Cincotta RP, Wisnewski J, Engelman R (2000) Human population in the biodiversity hotspots. Nature 404:990–992
Derneği D (2010) Ecosystem profile: Mediterranean basin biodiversity hotspot. CEPF—Critical Ecosystem Partnership Fund. Available at http://www.cepf.net/Documents/Mediterranean_EP_FINAL.pdf
Dujardin M, Hanna WW (1989) Crossability of pearl millet with wild Pennisetum species. Crop Sci 29:77–80
El Khlifi OK, Sharma H, Benlhabib O (2004) Transfer of Hessian fly resistance through interspecific crosses between cultivated wheat and four Aegilops species. Plant Genet Resour News 138:43–49
El-Beltagy A (2006) Central and West Asia and North Africa: challenges and opportunities. ICARDA, Aleppo
FAO (2016) FAOSTAT. Food and Agriculture of the United Nations, Rome, Italy. http://faostat3.fao.org/download/Q/QV/E (Accessed Feb 2016)
Ford-Lloyd BV, Kell SP, Maxted N (2008) Establishing conservation priorities for crop wild relatives. In: Maxted N, Ford-Lloyd BV, Kell SP, Iriondo JM, Dulloo ME, Turok J (eds) Crop wild relative conservation and use. CAB International, Wallingford, pp 110–119
Garzuglia M (2006) Threatened, endangered and vulnerable tree species: a comparison between FRA 2005 and the IUCN Red List. FAO, Forestry Department, working paper 108/E, Rome, Italy
Gregory PJ, Bengough AG, Grinev D, Schmidt S, Thomas WTB, Wojciechowski T, Young IM (2009) Root phenomics of crops: opportunities and challenges. Funct Plant Biol 36:922–929
Guo Y, Chen S, Li Z, Cowling WA (2014) Center of origin and centers of diversity in an ancient crop, Brassica rapa (Turnip Rape). J Hered, esu021
Harlan J, de Wet J (1971) Toward a rational classification of cultivated plants. Taxon 20:509–517
Hatzopoulos P, Banilas G, Giannoulia K, Gazis F, Nikoloudakis N, Milioni D, Haralampidis K (2002) Breeding, molecular markers and molecular biology of the olive tree. Eur J Lipid Sci Technol 104:574–586
ICARDA (2004) Annual Report (2003) International Center for Agricultural Research in the Dry Areas, Aleppo, Syria
ICARDA and UNEP (2000) Feasibility study on establishment of a Genebank for plant genetic resources in the Arab World. ICARDA, Aleppo
IUCN (2001) IUCN Red List categories and criteria: Version 3.1. IUCN, Cambridge
IUCN (2012) IUCN North Africa Programme: 2013–2016. IUCN, Cambridge
IUCN (2016) IUCN Red List. IUCN, Cambridge and Gland, UK and Switzerland. www.iucnredlist.org (Accessed Feb 2016)
IUCN–EGA (1996–1998) North Africa Biodiversity Programme, Phase 1, Final Summary Report
Jafri SMH, Ali SI, El-Ghadi AA (1976–1989) Flora of Libya. Al-Faateh University. Faculty of Sciences. Vol 1–147. Al-Faateh University, Tripoli, Libya
Jain SM, Priyadarshan PM (eds) (2009) Breeding plantation tree crops: temperate species, vol 2. Springer, Berlin
Kell SP, Maxted N, Ford-Lloyd BV (2005) PGR Forum: a project ends, but the mission continues. Crop Wild Relat 5:12
Kell SP, Knüpffer H, Jury SL, Ford-Lloyd BV, Maxted N (2008) Crops and wild relatives of the Euro-Mediterranean region: making and using a conservation catalogue. In: Maxted N, Ford-Lloyd BV, Kell SP, Iriondo J, Dulloo E, Turok J (eds) Crop wild relative conservation and use. CAB International, Wallingford, pp 69–109
Kell SP, Maxted N, Frese L, Iriondo JM (2012) In situ conservation of crop wild relatives: a strategy for identifying priority genetic reserve sites. In: Maxted N, Dulloo ME, Ford-Lloyd BV, Frese L, Iriondo JM, de Carvalho MAA (eds) Agrobiodiversity conservation: securing the diversity of crop wild relatives and landraces. CAB International, Wallingford, pp 7–19
Lakew B, Eglinton J, Henry RJ, Baum M, Grando S, Ceccarelli S (2011) The potential contribution of wild barley (Hordeum vulgare ssp. spontaneum) germplasm to drought tolerance of cultivated barley (H. vulgare ssp. vulgare). Field Crops Res 120:161–168
Le Floch É, Boulos L, Vela E (2010) Catalogue synonymique commenté de la flore de Tunisie. Simpact, Tunis
Maxted N, Hawkes JG, Guarino L, Sawkins M (1997) The selection of taxa for plant genetic conservation. Genet Resour Crop Evolut 44:337–348
Maxted N, Ford-Lloyd BV, Jury S, Kell SP, Scholten M (2006) Towards a definition of a crop wild relative. Biol Conserv 15:2673–2685
Maxted N, Scholten M, Codd R, Ford-Lloyd B (2007) Creation and use of a national inventory of crop wild relatives. Biol Conserv 140:142–159
Maxted N, Dulloo ME, Ford-Lloyd BV, Frese L, Iriondo, J, de Carvalho MAP (2011) Agrobiodiversity conservation securing the diversity of crop wild relatives and landraces. CABI. Page 65 In: Vavilov NI, Dorofeev VF, 1992. Origin and geography of cultivated plants. Cambridge University Press
Mizukami Y, Kato M, Takamizo T, Kanbe M, Inami S, Hattori K (2006) Interspecific hybrids between Medicago sativa L. and annual Medicago containing Alfafa weevil resistance. Plant Cell Tissue Organ Culture 84:79–88
Muhammad D, Misri B, EL-Nahrawy M, Khan S, Serkan A (2014) Egyptian Clover (Trifolium alexandrinum) King of Forage Crops. FAO, Regional Office for the Near East and North Africa, Cairo, Egypt
Myers N, Mittermeier RA, Mittermeier CG, Da Fonseca GA, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Nevo E, Chen G (2010) Drought and salt tolerances in wild relatives for wheat and barley improvement. Plant Cell Environ 33:670–685
Pons A, Quézel P (1985) The history of the flora and vegetation and past and present human disturbance in the Mediterranean region. In: Gomes-Campo C (ed) Plant Conservation in the Mediterranean area, W. Junk, Dordrecht, pp 25–43
Prescott-Allen R, Prescott-Allen C (2013) Genes from the wild: using wild genetic resources for food and raw materials. Routledge, Lomdon
Radford EA, Catullo G, de Montmollin B (2011) Important plant areas of the south and east Mediterranean region: priority sites for conservation. IUCN, Gland, p 108
Rankou H, Culham A, Sghir Taleb M, Ouhammou A, Martin G, Jury SL (2015) Conservation assessments and Red Listing of the endemic Moroccan flora (monocotyledons). Bot J Linn Soc 177:504–575
Redden R (2015) Wild relatives for the crop improvement challenges of climate change: the adaptation range of crops. In: Redden R, Yadav SS, Maxted N, Dulloo ME, Guarino L, Smith P (eds) Crop Wild Relatives and Climate Change. Wiley, New Jersey, pp 61–76
Sesli M, Yegenoglu ED (2010) Genetic relationships among and within wild and cultivated olives based on RAPDs. Genet Mol Res 9(3):1550–1556. doi:10.4238/vol9-3gmr866
Terzopoulos PJ, Kolano B, Bebeli PJ, Kaltsikes PJ, Metzidakis I (2005) Identification of olea europaea L. cultivars using inter-simple sequence repeat markers. Sci Hortic 105:45–51. doi:10.1016/j.scienta.2005.01.011
Tian D, Rose RJ (1999) Asymmetric somatic hybridisation between the annual legumes Medicago truncatula and Medicago scutellata. Plant Cell Rep 18:989996
UNU-WIDER (2011) Climate Change Compounding Risks in North Africa. 32
USDA (2016) Germplasm Resources Information Network—(GRIN) [Online Database]. National Germplasm Resources Laboratory, ARS, Beltsville, Maryland. http://www.ars-grin.gov/[Accessed March 2016]
Vavilov NI (1926) Studies in the origin of cultivated plants. Institute of Applied Botany and Plant Breeding, Leningrad
Vavilov NI, Dorofeev VF (1992) Origin and geography of cultivated plants. Cambridge University Press, Cambridge
Vincent H, Wiersema J, Kell SP, Fielder H, Dobbie S, Castañeda-Álvarez NP, Guarino L, Eastwood R, Leόn B, Maxted N (2013) A prioritised crop wild relative inventory to help underpin global food security. Biol Conserv 167:265–275
Zehni M (2006) Towards a Regional Strategy for the Conservation of Plant Genetic Resources in West Asia and North Africa (WANA). Association of Agricultural Research Institutes of the Near East and North Africa (AARINENA) www.aarinena.org/aarinena/documents/Strategy.pdf (Accessed March 2016)
Zeven AC, Zhukovsky PM (1975) Dictionary of cultivated plants and their centers of diversity. Center for Agricultural Publishing and Documentation, Wageningen
Acknowledgements
We acknowledge the financial support of the Government of Libya.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Lala, S., Amri, A. & Maxted, N. Towards the conservation of crop wild relative diversity in North Africa: checklist, prioritisation and inventory. Genet Resour Crop Evol 65, 113–124 (2018). https://doi.org/10.1007/s10722-017-0513-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-017-0513-5