Abstract
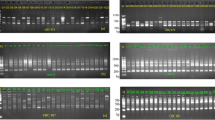
The main objective of this research was to add genotyping information to previous phenotyping findings of Pakistani guava for the purposes of selective breeding. Inter-primer binding site (iPBS) and microsatellite (SSR) markers were used to assess the molecular variation and genetic structure of 51 promising Pakistani guava (Psidium guajava L.) genotypes which were then compared with 19 others from different geographical regions across the world. PCR of 6 iPBS primers (dominant markers) produced a total of 97 bands (96.63 % polymorphic) ranging from 100 to 2800 bp and the mean PIC for primers ranged from 0.1687 to 0.3522. The mean unbiased expected heterozygosity (0.183), Shannon’s information index (0.275) and average fixation index (Fst or inbreeding) (0.925) indicated a high level of inbreeding among the accessions tested. Multi-locus DNA fingerprints using 18 SSR loci unambiguously differentiated all accessions and demonstrated an absence of duplicated samples. Diversity analysis revealed a total of 172 alleles (from 124 to 553 bp) ranging from 2 to 17 with a mean value of 9.56 alleles per locus. The mean unbiased expected heterozygosity (0.091), Shannon’s information index (0.130) and mean inbreeding coefficient (0.854) also indicated a high level of inbreeding among the accessions. Ordination and cluster analysis from both iPBS and SSR markers showed that the genetic relationships between all accessions could be separated into geographic origin, specifically Pakistan, Mexico, Hawaii and India. Guava accessions cultivated in Pakistan and wild guava germplasm are highly divergent and possess abundant genetic diversity. The iPBS PCR-based genome fingerprinting technology used in this study is low-cost and provides an effective alternative in differentiating accessions of guava and their related species or genera.







Similar content being viewed by others
References
Ahmed B, Mannan MA, Hossain SA (2011) Molecular characterization of guava (Psidium guajava L.) germplasm by RAPD analysis. Int J Nat Sci 1:62–67
Aranguren Y, Briceno A, Fermin G (2010) Assessment of the variability of Venezuelan guava landraces by microsatellites. In: Rohde W, Fermin G (eds) Acta Hortic, vol 849, pp 147–154
Baijnath H, Ramcharun S, Naicker S (1982) Psidium spp., (Myrtaceae); very successful weeds. S Afr J Bot 1:78
Baranek M, Meszaros M, Sochorova J, Cechova J, Raddova J (2012) Utility of retrotransposon-derived marker systems for differentiation of presumed clones of the apricot cultivar Velkopavlovicka. Sci Hortic 143:1–6. doi:10.1016/j.scienta.2012.05.022
Briceno A, Aranguren Y, Fermin G (2010) Assessment of guava-derived SSR markers for the molecular characterization of Myrtaceae from different ecosystems in Venezuela. In: Rohde W, Fermin G (eds) Acta Hortic, vol 849, pp 139–146
Chen T-W, Ng C-C, Wang C-Y, Shyu Y-T (2007) Molecular identification and analysis of Psidium guajava L. from indigenous tribes of Taiwan. J Food Drug Anal 15:82–88
Coser SM, da Silva Ferreira MF, Ferreira A, Mitre LK, Carvalho CR, Clarindo WR (2012) Assessment of genetic diversity in Psidium guajava L. using different approaches. Sci Hortic 148:223–229. doi:10.1016/j.scienta.2012.09.030
da Costa SR, Santos CAF, Castro JMC (2012) Assessing Psidium guajava × P. guineense hybrids tolerance to Meloidogyne enterolobii. In: Santos CAF, Mitra SK, Griffis Jr JL (eds) Acta Hortic, vol 959, pp 59–66
Dayanandan S, Bawa KS, Kesseli R (1997) Conservation of microsatellites among tropical trees (Leguminosae). Am J Bot 84:1658–1663. doi:10.2307/2446463
Dettori MT, Palombi MA (2000) Identification of Feijoa sellowiana Berg accessions by RAPD markers. Sci Hortic 86:279–290. doi:10.1016/s0304-4238(00)00157-6
FAO (2002) Commodity market: reviews 2001–2002. Food and Agriculture Organization of the United Nations, Rome
Felsenstein J (2005) PHYLIP (Phylogeny Inference Package), 3.6 edn. Distributed by the author, Department of Genetics, University of Washington, Seattle, Washington
Feria-Romero IA, Astudillo-de la Vega H, Chavez-Soto MA, Rivera-Arce E, Lopez M, Serrano H, Lozoya X (2009) RAPD markers associated with quercetin accumulation in Psidium guajava. Biol Plant 53:125–128. doi:10.1007/s10535-009-0017-z
Gailite A, Rungis D (2012) An initial investigation of the taxonomic status of Saussurea esthonica Baer ex Rupr. utilising DNA markers and sequencing. Plant Syst Evol 298:913–919. doi:10.1007/s00606-012-0600-1
Guo D-L, Guo M-X, Hou X-G, Zhang G-H (2014) Molecular diversity analysis of grape varieties based on iPBS markers. Biochem Syst Ecol 52:27–32. doi:10.1016/j.bse.2013.10.008
Hayes WB (1970) Fruit growing in India, 3rd edn. Kitabistan, Allahabad, p 72
Hernandez-Delgado S, Padilla-Ramirez JS, Nava-Cedillo A, Mayek-Perez N (2007) Morphological and genetic diversity of Mexican guava germplasm. Plant Genet Resour Charact Util 5:131–141. doi:10.1017/s1479262107827055
Jaiswal U, Jaiswal VS (2005) Psidium guajava guava. In: Litz RE (ed) Biotechnology of fruit and nut crops. Biotechnology in agricultural series, vol 29. CAB Publishers, Cambridge, pp 394–401
Kalendar R, Antonius K, Smykal P, Schulman A (2010) iPBS: a universal method for DNA fingerprinting and retrotransposon isolation. Theor Appl Genet 121:1419–1430. doi:10.1007/s00122-010-1398-2
Kalendar R, Flavell AJ, Ellis THN, Sjakste T, Moisy C, Schulman AH (2011) Analysis of plant diversity with retrotransposon-based molecular markers. Heredity 106:520–530. doi:10.1038/hdy.2010.93
Landrum LR (1990) Accara—a new genus of Myrtaceae, Myrtinae from Brazil. Syst Bot 15:221–225. doi:10.2307/2419176
Lepitre V et al (2010) The microsatellite (SSR)/AFLP reference linkage map of guava. In: Rohde W, Fermin G (eds) Acta Hortic, vol 849, pp 183–192
Lewontin RC (1972) The apportionment of human diversity. Evol Biol 6:381–398
Mehmood A, Jaskani MJ, Khan IA, Ahmad S, Ahmad R, Luo S, Ahmad NM (2014) Genetic diversity of Pakistani guava (Psidium guajava L.) germplasm and its implications for conservation and breeding. Sci Hortic 172:221–232. doi:10.1016/j.scienta.2014.04.005
Mehmood A, Jaskani MJ, Saeed A, Rashid A (2013) Evaluation of genetic diversity in open pollinated guava by iPBS primers. Pak J Agric Sci 50:591–597
Menzel CM (1985) Guava: an exotic fruit with potential in Queensland. Qld Agric J 111:93–98
Nei M, Li WH (1979) Mathematical-model for studying genetic-variation in terms of restriction endonucleases. Proc Natl Acad Sci USA 76:5269–5273. doi:10.1073/pnas.76.10.5269
Odong TL, van Heerwaarden J, Jansen J, van Hintum TJL, van Eeuwijk FA (2011) Determination of genetic structure of germplasm collections: are traditional hierarchical clustering methods appropriate for molecular marker data? Theor Appl Genet 123:195–205. doi:10.1007/s00122-011-1576-x
Padilla-Ramirez JS, Gonzalez-Gaona E (2010) Collection and characterization of Mexican guava (Psidium guajava L.) germplasm. In: Rohde W, Fermin G (eds) Acta Hortic, vol 849, pp 49–54
Pakistan Statistical Yearbook (2010) Government of Pakistan, Islamabad
Pathak RK, Ojha CM (1993) Genetic resources of guava. Advances in horticulture: fruit crops, vol 1. Malhotra Publishing House, New Delhi
Peakall R, Smouse PE (2006) GenAlEx 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295. doi:10.1111/j.1471-8286.2005.01155.x
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research—an update. Bioinformatics 28:2537–2539. doi:10.1093/bioinformatics/bts460
Pommer CV (2012) Guava world-wide breeding: major techniques and cultivars and future challenges. In: Santos CAF, Mitra SK, Griffis Jr JL (eds) Acta Hortic, vol 959, pp 81–88
Pommer CV, Murkami KRN (2009) Breeding guava (Psidium guajava L.). In: Jain SM, Pommer CV (eds) Breeding plantation tree crop: tropical species. Springer, Berlin, pp 83–120
Prakash DP, Narayanaswamy P, Sondur SN (2002) Analysis of molecular diversity in guava using RAPD markers. J Hortic Sci Biotechnol 77:287–293
Risterucci AM, Duval MF, Rohde W, Billotte N (2005) Isolation and characterization of microsatellite loci from Psidium guajava L. Mol Ecol Notes 5:745–748. doi:10.1111/j.1471-8286.2005.01050.x
Ritter E (2012) Guava biotechnologies, genomic achievements and future needs. In: Santos CAF, Mitra SK, Griffis Jr JL (eds) Acta Hortic, vol 959, pp 131–140
Rodriguez-Medina NN, Valdes-Infante J, Gonzalez G, Fuentes V, Canizares J (2010) Genetic resources of guava (Psidium guajava) in Cuba: germplasm characterization and breeding. In: Rohde W, Fermin G (eds) Acta Hortic, vol 849, pp 341–348
Rodríguez N, Valdés-Infante J, Becker D, Velázquez B, Coto O, Ritter E, Rohde W (2004) Morphological, agronomic and molecular characterization of Cuban accessions of guava (Psidium guajava L.). J Genet Breed 58:79–90
Rodriguez N et al (2007) Characterization of guava accessions by SSR markers, extension of the molecular linkage map, and mapping of QTLs for vegetative and reproductive characters. In: Singh G, Kishun R, Chandra R (eds) Proceedings of the 1st international Guava symposium. Acta Hortic, vol 735, pp 201–215
Sanchez-Teyer LF, Barraza-Morales A, Keb L, Barredo F, Quiroz-Moreno A, O’Connor-Sanchez A, Padilla-Ramirez JS (2010) Assessment of genetic diversity of Mexican guava germplasm using DNA molecular markers. In: Rohde W, Fermin G (eds) Acta Hortic, vol 849, pp 133–138
Sitther V, Zhang D, Harris DL, Yadav AK, Zee FT, Meinhardt LW, Dhekney SA (2014) Genetic characterization of guava (Psidium guajava L.) germplasm in the United States using microsatellite markers. Genet Resour Crop Evol 61:829–839. doi:10.1007/s10722-014-0078-5
Smykal P, Bacova-Kerteszova N, Kalendar R, Corander J, Schulman AH, Pavelek M (2011) Genetic diversity of cultivated flax (Linum usitatissimum L.) germplasm assessed by retrotransposon-based markers. Theor Appl Genet 122:1385–1397. doi:10.1007/s00122-011-1539-2
Valdés-Infante D et al (2003) Molecular characterization of Cuban accessions of guava (Psidium guajava L.), establishment of a first molecular linkage map and mapping of QTLs for vegetative characters. J Genet Breed 57:349–358
Valdes-Infante J et al (2010) Comparison of the polymorphism level, discriminating capacity and informativeness of morpho-agronomic traits and molecular markers in guava (Psidium guajava L.). In: Rohde W, Fermin G (eds) Acta Hortic, vol 849, pp 121–132
Van Wyk AE (1980) A note on the seed morphology of the genus Eugenia Myrtaceae in Southern Africa. S Afr J Bot 46:115–120
Van Wyk AE, Botha R (1984) The genus Eugenia (Myrtaceae) in Southern-Africa—ontogeny and taxonomic value of the seed. S Afr J Bot 3:63–80
Watson L, Dallwitz MJ (2007) The families of flowering plants: descriptions, illustrations, identification, and information retrieval. DELTA—DEscription Language for TAxonomy. http://delta-intkey.com. Accessed June 28, 2015
Wright S (1965) The interpretation of population structure by F-statistics with special regards to systems of mating. Evolution 19:395–420
Wuensch A (2009) Cross-transferable polymorphic SSR loci in Prunus species. Sci Hortic 120:348–352. doi:10.1016/j.scienta.2008.11.012
Acknowledgments
This project is part of Ph.D. research funded by the Higher Education Commission (HEC) of Pakistan. We thank Professor Emeritus R.A. McIntosh for assistance in the preparation of the manuscript. The authors would also like to thank the Editor and reviewers for their most helpful comments and suggestions. We would like also to thank Dr. Urmil Bansal for her help with LI-COR DNA analyzer. Our thanks are also due to Dr. Xiaochun Zhao, Dr. Hanif Miah, Dr. Akram Khan, Mr. Muhammad B. Gill and Mr. Keshab Kandel for providing some of the plant samples. We greatly appreciate the help of Dr. Abdul Karim in the DNA extractions of the Pakistani guava accessions. Our thanks are also due to Dr. Faisal Saeed Awan (CABB) who provided useful materials for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mehmood, A., Luo, S., Ahmad, N.M. et al. Molecular variability and phylogenetic relationships of guava (Psidium guajava L.) cultivars using inter-primer binding site (iPBS) and microsatellite (SSR) markers. Genet Resour Crop Evol 63, 1345–1361 (2016). https://doi.org/10.1007/s10722-015-0322-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-015-0322-7