Abstract
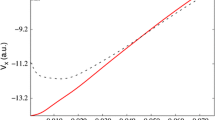
Slater’s method is an integral part of the undergraduate experience. In actuality, Slater’s method is part of an atomic model and not simply a set of rules. Slater’s rules are a simple means for computing the effective nuclear charge experienced by an orbital. These rules are based on the shell-like structure of the Slater atom in which outer shell electrons are incapable of shielding inner electrons. Slater’s model provides a qualitative description of the valence electrons in multi-electron atoms with tremendous ease. The model is useful for explaining atomic properties such as ionisation energy, electron affinity and atomic radius qualitatively. Slater’s rules also correctly reproduce the Madelung rule of filling and the ionisation sequence (4s before 3d); however, these rules are not able to reproduce the anomalous configurations of atoms such as Cr and Cu. It is found that the atomic properties that Slater’s model reproduces are all related to the exponential decay factor of the Slater orbital. We find—from estimating the polarity of a diatomic molecule using a simple model—that molecular polarity is related to the difference in the exponential decay factors of the valence orbitals of the two atoms, implying that the decay factor acts as the electronegativity of the atom.















Similar content being viewed by others
References
Allen, L.C.: Electronegativity is the average one-electron energy of the valence-shell electrons in ground-state free atoms. J. Am. Chem. Soc. 111, 9003–9014 (1989). https://doi.org/10.1021/ja00207a003
Allred, A.L., Rochow, E.G.: A scale of electronegativity based on electrostatic force. J. Inorg. Nucl. Chem. 5, 264–268 (1958). https://doi.org/10.1016/0022-1902(58)80003-2
Alvarez, S.: A cartography of the van der Waals territories. Dalt. Trans. 42, 8617 (2013). https://doi.org/10.1039/c3dt50599e
Batsanov, S.: Van der Waals radii of elements. Inorg. Mater. 37, 871–885 (2001). https://doi.org/10.1023/a:1011625728803
Bohórquez, H.J., Boyd, R.J.: Is the size of an atom determined by its ionization energy? Chem. Phys. Lett. 480, 127–131 (2009). https://doi.org/10.1016/j.cplett.2009.08.034
Bohr, N.: On the constitution of atoms and molecules. Lond. Edinb. Dublin Philos. Mag. J. Sci. 26, 1–25 (1913). https://doi.org/10.1080/14786441308634955
Bratsch, S.G.: Revised Mulliken electronegativities: I. Calculation and conversion to Pauling units. J. Chem. Educ. 65, 34 (1988). https://doi.org/10.1021/ed065p34
Brink, C.P.: The use of effective nuclear charge (Z*) calculations to illustrate the relative energies of ns and (n − 1)d orbitals. J. Chem. Educ. 68, 376 (1991). https://doi.org/10.1021/ed068p376
Brown, T.L., LeMay, H.E., Jr., Bursten, B.E., Murphy, C.J., Woodward, P.M., Stoltzfus, M.W., Lufaso, M.W.: Chemistry: The Central Science. Pearson, New York (2018)
Chakraborty, T., Gazi, K., Ghosh, D.C.: Computation of the atomic radii through the conjoint action of the effective nuclear charge and the ionization energy. Mol. Phys. 108, 2081–2092 (2010). https://doi.org/10.1080/00268976.2010.505208
Clementi, E., Raimondi, D.L.: Atomic screening constants from SCF functions. J. Chem. Phys. 38, 2686–2689 (1963)
Clementi, E., Raimondi, D.L., Reinhardt, W.P.: Atomic screening constants from SCF functions. II. Atoms with 37 to 86 electrons. J. Chem. Phys. 47, 1300–1307 (1967)
DeKock, R.L., Strikwerda, J.R., Yu, E.X.: Atomic size, ionization energy, polarizability, asymptotic behavior, and the Slater–Zener model. Chem. Phys. Lett. 547, 120–126 (2012). https://doi.org/10.1016/j.cplett.2012.07.072
Fock, V.: Näherungsmethode zur Lösung des quantenmechanischen Mehrkörperproblems. Z. Phys. 61, 126 (1930)
Goudsmit, S.A., Richards, P.I.: The order of electron shells in ionized atoms. Proc. Natl. Acad. Sci. 51, 664–671 (1964). https://doi.org/10.1073/pnas.51.4.664
Hartree, D.R.: The wave mechanics of an atom with a non-coulomb central field. Part II. Some results and discussion. Math. Proc. Cambridge Philos. Soc. 24, 111–132 (1928). https://doi.org/10.1017/S0305004100011920
Hehre, W.J., Stewart, R.F., Pople, J.A.: Self-consistent molecular-orbital methods. I. Use of Gaussian expansions of Slater-type atomic orbitals. J. Chem. Phys. 51, 2657–2664 (1969). https://doi.org/10.1063/1.1672392
Heitler, W., London, F.: Wechselwirkung neutraler Atome und homöopolare Bindung nach der Quantenmechanik. Zeitschrift Für Phys. 44, 455–472 (1927). https://doi.org/10.1007/BF01397394
Koopmans, T.: The distribution of wave function and characteristic value among the individual electrons of an atom. Physica 1, 104–113 (1933)
Kumari, D., Saloni, Tandon, H., Labarca, M., Chakraborty, T.: An FSGO based electronegativity scale invoking the electrophilicity index. ChemistrySelect (2022a). https://doi.org/10.1002/slct.202202238
Kumari, V., Singh, T., Devi, S., Tandon, H., Labarca, M., Chakraborty, T.: Atomic electronegativity based on hardness and floating spherical Gaussian orbital approach. J. Math. Chem. (2022b). https://doi.org/10.1007/s10910-021-01306-7
Lias, S.G., Rosenstock, H.M., Draxl, K., Steiner, B.W., Herron, J.T., Holmes, J.L., Levin, R.D., Liebman, J.F., Kafafi, S.A.: Ionization Energetics (IE, AE) data. In: Linstrom, P.J. and Mallard, W.G. (eds.) NIST Standard Reference Database Number 69. National Institute of Standards and Technology, Gaithersburg MD, 20899 (2018)
Lide, D.R. (ed.): CRC Handbook of Chemistry and Physics. CRC Press, Boca Raton (2003)
Löwdin, P.-O.: On the long way from the Coulombic Hamiltonian to the electronic structure of molecules. Pure Appl. Chem. 61, 2065–2074 (1989). https://doi.org/10.1351/pac198961122065
Mann, J.B., Meek, T.L., Allen, L.C.: Configuration Energies of the Main Group Elements. J. Am. Chem. Soc. 122, 2780–2783 (2000a). https://doi.org/10.1021/ja992866e
Mann, J.B., Meek, T.L., Knight, E.T., Capitani, J.F., Allen, L.C.: Configuration energies of the d-block elements. J. Am. Chem. Soc. 122, 5132–5137 (2000b). https://doi.org/10.1021/ja9928677
Mulliken, R.S.: A new electroaffinity scale; together with data on valence states and on valence ionization potentials and electron affinities. J. Chem. Phys. 2, 782–793 (1934). https://doi.org/10.1063/1.1749394
Pauling, L.: The nature of the chemical bond. IV. The energy of single bonds and the relative electronegativity of atoms. J. Am. Chem. Soc. 54, 3570–3582 (1932). https://doi.org/10.1021/ja01348a011
Pauling, L.: The Nature of the Chemical Bond and the Structure of Molecules and Crystals: An Introduction to Modern Structural Chemistry. Cornell University Press, Ithaca (1960)
Petrucci, R.H., Herring, F.G., Madura, J.D., Bissonnette, C.: General Chemistry: Principles and Modern Applications. Pearson Canada Inc., Toronto (2017)
Pilar, F.L.: 4s is always above 3d! Or, how to tell the orbitals from the wavefunctions. J. Chem. Educ. 55, 2–6 (1978). https://doi.org/10.1021/ed055p2
Pople, J.A., Nesbet, R.K.: Self-consistent orbitals for radicals. J. Chem. Phys. 22, 571–572 (1954)
Roothaan, C.C.J.: New developments in molecular orbital theory. Revs. Mod. Phys. 23, 69–89 (1951)
Shankar, R.: Principles of Quantum Mechanics. Kluwer Academic, New York (1994)
Silberberg, M.S.: Principles of General Chemistry. McGraw-Hill Publishing Company, Dubuque (2010)
Slater, J.C.: Atomic shielding constants. Phys. Rev. 36, 57–64 (1930)
Tandon, H., Chakraborty, T., Suhag, V.: A scale of atomic electronegativity in terms of atomic nucleophilicity index. Found. Chem. 22, 335–346 (2020). https://doi.org/10.1007/s10698-020-09358-4
Tudela, D.: Slater’s rules and electron configurations. J. Chem. Educ. 70, 956 (1993). https://doi.org/10.1021/ed070p956
Viswanathan, B., Gulam Razul, M.S.: Orthogonality properties of states, configurations, and orbitals. Found. Chem. 24, 73–86 (2022). https://doi.org/10.1007/s10698-022-09417-y
Viswanathan, B., Gulam Razul, M.S.: Electronegativity provides the relationship between formal charge, oxidation state, and actual charge. Found. Chem. 25, 5–28 (2023). https://doi.org/10.1007/s10698-022-09447-6
Zener, C.: Analytic atomic wave functions. Phys. Rev. 36, 51–56 (1930)
Funding
There are no funding sources to disclose.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Viswanathan, B., Gulam Razul, M.S. Relating screening to atomic properties and electronegativity in the Slater atom. Found Chem (2023). https://doi.org/10.1007/s10698-023-09492-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s10698-023-09492-9