Abstract
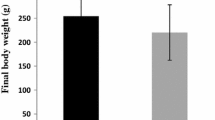
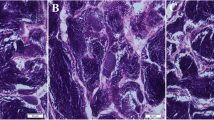
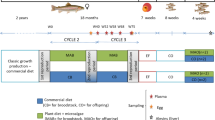
Although the immuno-modulatory and stress-relieving properties of β-glucan is well elucidated in humans and other animal models, including fish, its role as a dietary supplement on reproduction is extremely scarce. Therefore, in this study, adult female fish were fed one of four test diets having 0 (control), 0.5, 1, and 1.5% β-d-glucan for 130 days and its effect on reproductive performance, ovarian and liver histology, sex hormones, and transcript abundance of selected reproduction-related genes was assessed. Low dietary intake of β-glucan improved fertilization and hatching rates (p<0.05). The relative fecundity and percentage of spawning females were higher (non-significant) in 0.5% β-glucan-fed groups. Surprisingly, even after 130 days, spawning did not occur in 1.5% β-glucan-fed individuals. Irrespective of β-glucan intake, all the brooders recorded similar plasma 17β-estradiol and maturation-inducing hormone (p>0.05). Higher intake of β-glucan (1.5%) upregulated aromatase genes without a parallel increase in 17β-estradiol. However, plasma vitellogenin increased with increasing β-glucan up to 1.0% then declined at 1.5% (p<0.05). The fish that received control, 0.5, and 1.5% β-glucan recorded similar vitellogenin levels in their plasma. Significantly higher plasma cortisol was evidenced in 1.5% β-glucan fed brooders (p<0.05). Histologically, higher follicular atresia and leaking of yolk material was evidenced in 1.5% β-glucan-fed group. Liver histology revealed the highest nutrient/lipid accumulation in fish that received 1.0% and 1.5% β-glucan. This study demonstrated the stimulatory effect of β-glucan intake at a lower dose (0.5%) on reproduction. However, higher intake (1.5%) could perturb normal reproductive function in a fish model and caused an increased number of atretic follicles leading to spawning/reproductive failure.
Graphical Abstract









Similar content being viewed by others
Data availability
The data used in the manuscript will be made available from the corresponding author on reasonable request.
References
Abu-Elala NM, Ali TES, Ragaa NM, Ali SE, Abd-Elsalam RM, Younis NA, Abdel-Moneam DA, Hamdien AH, Bonato M, Dawood MAO (2021) Analysis of the productivity, immunity, and health performance of Nile tilapia (Oreochromis niloticus) broodstock-fed dietary fermented extracts sourced from Saccharomyces cerevisiae (Hilyses): A field trial. Animals 11:815–835. https://doi.org/10.3390/ani11030815
Akhtar MS, Ciji A, Sarma D, Rajesh M, Kamalam BS, Sharma P, Singh AK (2017) Reproductive dysfunction in females of endangered golden mahseer (Tor putitora) in captivity. Anim Reprod Sci 182:95–103. https://doi.org/10.1016/j.anireprosci.2017.05.004
Akhtar MS, Rajesh M, Ciji A, Sharma P, Kamalam BS, Patiyal RS, Singh AK, Sarma D (2018) Photo-thermal manipulations induce captive maturation and spawning in endangered golden mahseer (Tor putitora): a silver-lining in the strangled conservation efforts of decades. Aquaculture 497:336–347. https://doi.org/10.1016/j.aquaculture.2018.08.003
Akhtar MS, Manchi R, Kamalam BS, Ciji A (2020) Effect of photoperiod and temperature on indicators of immunity and wellbeing of endangered golden mahseer (Tor putitora) broodstock. J Therm Biol 93:102694. https://doi.org/10.1016/j.jtherbio.2020.102694
Akhtar MS, Tripathi HP, Pandey A, Ciji A (2021a) β-glucan modulates non-specific immune gene expression, thermal tolerance and elicits disease resistance in endangered Tor putitora fry challenged with Aeromonas salmonicida. Fish Shellfish Immunol 119:154–162. https://doi.org/10.1016/j.fsi.2021.09.038
Akhtar MS, Tripathi HP, Pandey A, Ciji A (2021b) Transgenerational effects of β-glucan on thermal tolerance, growth performance, and immune gene expression of endangered cyprinid Tor putitora progeny. J Therm Biol 102:103120. https://doi.org/10.1016/j.jtherbio.2021.103120
Akhtar MS, Ciji A, Tripathi HP, Sharma P (2021c) Dietary β-glucan influences the expression of testicular aquaporins, antioxidative defence genes and sperm quality traits in endangered golden mahseer, Tor putitora (Hamilton, 1822). Int J Biol Macromol 193:1286–1293. https://doi.org/10.1016/j.ijbiomac.2021.10.177
Akhtar MS, Tripathi PH, Ciji A (2022) Light spectra influence the reproductive performance and expression of immune and anti-oxidative defense genes in endangered golden mahseer (Tor putitora) female brooders. Aquaculture 547:737355. https://doi.org/10.1016/j.aquaculture.2021.737355
AOAC (2012) Official methods of analysis. Arlington, Association of Official Analytical Chemists, p 1141
Arantes FP, Santos HB, Rizzo E, Sato Y, Bazzoli N (2010) Profiles of sex steroids, fecundity, and spawning of the curimatã-pacu Prochilodus argenteus in the São Francisco River, downstream from the Três Marias Dam, Southeastern Brazil. Anim Reprod Sci 118:330–336. https://doi.org/10.1016/j.anireprosci.2009.07.004
Blazer VS (2002) Histopathological assessment of gonadal tissue in wild fishes. Fish Physiol Biochem 2:85–101. https://doi.org/10.1023/A:1023332216713
Bobe J, Labbé C (2010) Egg and sperm quality in fish. Gen Comp Endocrinol 165:535–548. https://doi.org/10.1016/j.ygcen.2009.02.011
Bombardelli RA, Mewes JK, Buzzi AH, Pedreira ACO, Syperreck MA, Dalmaso ACS, Chagas TV, Chiella RJ, Meurer F (2021) Diets containing crude glycerin modify the ovary histology, cause reproductive harm on Nile tilapia females and impair the offspring quality. Aquaculture 533:736098. https://doi.org/10.1016/j.aquaculture.2020.736098
Bowers EK, Bowden RM, Sakaluk SK, Thompson CF (2015) Immune activation generates corticosterone-mediated terminal reproductive investment in a wild bird. Am Nat 185(6):769–783. https://doi.org/10.1086/681017
Cerda J, Zapater C, Chauvigné F, Finn RN (2013) Water homeostasis in the fish oocyte: new insights into the role and molecular regulation of a teleost-specific aquaporin. Fish Physiol Biochem 39(1):19–27. https://doi.org/10.1007/s10695-012-9608-2
Cheshenko K, Pakdel F, Segner H, Kah O, Eggen RIL (2008) Interference of endocrine disrupting chemicals with aromatase CYP19 expression or activity, and consequences for reproduction of teleost fish. Gen Comp Endocrinol 55(1):31–62. https://doi.org/10.1016/j.ygcen.2007.03.005
Ciji A, Akhtar MS (2021) Stress management in aquaculture: a review of dietary interventions. Rev Aquac 13:2190–2247. https://doi.org/10.1111/raq.12565
Ciji A, Sharma P, Rajesh M, Kamalam BS, Sharma A, Dash P, Akhtar MS (2021) Intra-annual changes in reproductive indices of male and female Himalayan snow trout, Schizothorax richardsonii (Gray, 1832). Aquac Res 52:130–141. https://doi.org/10.1111/are.14875
Ciji A, Akhtar MS, Dubey MK, Pandey A, Tripathi PH, Kamalam BS, Rajesh M, Sharma P (2022) Comparative assessment of egg and larval quality traits of progeny from wild-collected and captive-matured brooders of endangered golden mahseer, Tor putitora: A prelude to quality broodstock development and seed production. Aquaculture 552:737949. https://doi.org/10.1016/j.aquaculture.2022.737949
Corriero A, Zupa R, Mylonas CC, Letizia P (2022) Atresia of ovarian follicles in fishes, and implications and uses in aquaculture and fisheries. J Fish Dis 44:1271–1291. https://doi.org/10.1111/jfd.13469
Drummond CD, Bazzoli N, Rizzo E, Sato Y (2000) Postovulatory follicle: a model for experimental studies of programmed cell death or apoptosis in teleosts. J Exp Zool 287:176–182. https://doi.org/10.1002/1097-010X(20000701)287:2<176::AID-EZ8>3.0.CO;2-2
Forsatkar MN, Nematollahi MA, Rafiee G, Farahmand H, Lawrence C (2018) Effects of the prebiotic mannan-oligosaccharide on feed deprived zebrafish: growth and reproduction. Aquac Res 49:2822–2832. https://doi.org/10.1111/are.13745
Gioacchini G, Maradonna F, Lombardo F, Bizzaro D, Olivotto I, Carnevali O (2010) Increase of fecundity by probiotic administration in zebrafish (Danio rerio). Reprod 140:953–959. https://doi.org/10.1530/REP-10-0145
Grier HJ, Uribe Aranzábal MC, Patiño R (2009) The ovary, folliculogenesis, and oogenesis in teleosts. In: Jamieson BGM (ed) Reproductive Biology and Phylogeny of Fishes. Science Publishers, Enfield, New Hampshire, USA, pp 25–87
Han SK, Gottsch ML, Lee KJ, Popa SM, Smith JT, Jakawich SK, Clifton DK, Steiner RA, Herbison AE (2005) Activation of gonadotropin-releasing hormone neurons by kisspeptin as a neuoendocrine switch for the onset of puberty. J Neurosci 25:11349–11356. https://doi.org/10.1523/JNEUROSCI.3328-05.2005
Jang YD, Kang KW, Piao LG, Jeong TS, Auclair E, Jonvel S, D’Inca R, Kim YY (2013) Effects of live yeast supplementation to gestation and lactation diets on reproductive performance, immunological parameters and milk composition in sows. Livest Sci 152:167–173. https://doi.org/10.1016/j.livsci.2012.12.022
Khan AA, Gani A, Masoodi FA, Amin F, Wani IA, Khanday FA, Gani A (2016) Structural, thermal, functional, antioxidant and antimicrobial properties of β-D-glucan extracted from baker’s yeast (Saccharomyces cereviseae) - Effect of γ-irradiation. Carbohydr Polym 140:442–450. https://doi.org/10.1016/j.carbpol.2016.01.003
Meena DK, Das P, Kumar S, Mandal SC, Prusty AK, Singh SK, Akhtar MS, Behera BK, Kumar K, Pal AK, Mukherjee SC (2013) Beta-glucan: an ideal immunostimulant in aquaculture (a review). Fish Physiol Biochem 39:431–457. https://doi.org/10.1007/s10695-012-9710-5
Mehdinejad N, Imanpou MR, Jafari V (2019) Combined or individual effects of dietary probiotic, Pediococcus acidilactici and nucleotide on reproductive performance in goldfish (Carassius auratus). Probiotics Antimicrob Proteins 11:233–238. https://doi.org/10.1007/s12602-017-9377-4
Mehrim AI, Khalil FF, Hassan ME (2015) Hydroyeast Aquaculture ® as a reproductive enhancer agent for the adult Nile tilapia (Oreochromis niloticus Linnaeus, 1758). Fish Physiol Biochem 41:371–381. https://doi.org/10.1007/s10695-014-9989-5
Mona MH, Alm-Eldeen AA, Elgayar EE, Heneish AM, El-feky MMM (2015) Evaluation the effect of local and imported yeasts as supplementary food on the African catfish (Clarias gariepinus) in Egypt. J Aquac Mari Biol 2(3):00024. https://doi.org/10.15406/jamb.2015.02.00024
Monsang SJ, Parhi J, Choudhury J, Deka A, Pal P (2019) Molecular characterisation of carp aquaporin-3a: expression profiling during hormone-induced spawning. Aquaculture 500:569–575. https://doi.org/10.1016/j.aquaculture.2018.10.069
Nagahama Y (1994) Endocrine regulation of gametogenesis in fish. Int J Dev Biol 38(2):217–229
Ohga H, Selvaraj S, Matsuyama M (2018) The roles of kisspeptin system in the reproductive physiology of fish with special reference to chub mackerel studies as main axis. Front Endocrinol 9:147. https://doi.org/10.3389/fendo.2018.00147
Okumura S, Okamoto K, Oomori R, Nakazono A (2002) Spawning behavior and artificial fertilization in captive reared red spotted grouper, Epinephelus akaara. Aquaculture 206:165–173
Peng X, Yan C, Hu L, Huang Y, Fang Z, Lin Y, Xu S, Feng B, Li J, Zhuo Y, Wu D (2020) Lianqiang Che Live yeast supplementation during late gestation and lactation affects reproductive performance, colostrum and milk composition, blood biochemical and immunological parameters of sows. Anim Nutr 6:288–292. https://doi.org/10.1016/j.aninu.2020.03.001
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29(9):e45. https://doi.org/10.1093/nar/29.9.e45
Rajakumar A, Senthilkumaran B (2020) Steriogenesis and its regulation in teleost-a review. Fish Physiol Biochem 46(3):803–818. https://doi.org/10.1007/s10695-019-00752-0
Santos HB, Thomé RG, Arantes FP, Sato Y, Bazzoli N, Rizzo E (2008) Ovarian follicular atresia is mediated by heterophagy, autophagy, and apoptosis in Prochilodus argenteus and Leporinus taeniatus (Teleostei: Characiformes). Theriogenology 70:1449–1460
Shahi N, Singh AK, Sahoo M, Mallik SK, Thakuria D (2017) Molecular cloning, characterization and expression profile of kisspeptin1 and kisspeptin1 receptor at brain-pituitary-gonad (BPG) axis of golden mahseer, Tor putitora (Hamilton, 1822) during gonadal development. Comp Biochem Physiol Part B 205:13–29. https://doi.org/10.1016/j.cbpb.2016.11.010
Stier H, Ebbeskotte V, Gruenwald J (2014) Immune-modulatory effects of dietary yeast Beta-1,3/1,6-D-glucan. Nutr J 13:38–46 http://www.nutritionj.com/content/13/1/3
Vainikka A, Jokinen EI, Kortet R, Paukku S, Pirhonen J, Rantala MJ, Taskinen J (2005) Effects of testosterone and β-glucan on immune functions in tench. J Fish Biol 66:348–361. https://doi.org/10.1111/j.1095-8649.2004.00598.x
Vannucci L, Krizan J, Sima P, Stakheev D, Caja F, Rajsiglova L, Horak V, Saieh M (2013) Immunostimulatory properties and antitumor activities of glucans (Review). Int J Oncol 43:357–364. https://doi.org/10.3892/ijo.2013.1974
Vetvicka V (2011) Glucan-immunostimulant, adjuvant, potential drug. World J Clin Oncol 2(2):115–119. https://doi.org/10.5306/wjco.v2.i2.115
Vetvicka V, Vannucci L, Sima P (2020) β-glucan as a new tool in vaccine development. Scand J Immunol 91:e12833. https://doi.org/10.1111/sji.12833
Zaleska B, Milewski S, Zabek K (2015) Impact of Saccharomyces cerevisiae supplementation on reproductive performance, milk yield in ewes and offspring growth. Arch Anim Breed 58:79–83. https://doi.org/10.5194/aab-58-79-2015
Zhen W, Shao Y, Wu Y, Li L, Pham VH, Abbas W, Wan Z, Guo Y, Wang Z (2020) Dietary yeast β-glucan supplementation improves eggshell color and fertile eggs hatchability as well as enhances immune functions in breeder laying hens. Int J Biol Macromol 159:607–621. https://doi.org/10.1016/j.ijbiomac.2020.05.134
Acknowledgements
The authors duly acknowledge the Director, ICAR-DCFR, for facilitating all the logistics support during the study. The sincere help extended by Sh. Harish Chandra and Sh. Prem Pandey for rearing and maintenance of the experimental fish is greatly acknowledged.
Funding
The study was financially supported by the Department of Biotechnology (DBT), Ministry of Science and Technology, Government of India (vide reference no. BT/PR26920/AAQ/3/884/2017).
Author information
Authors and Affiliations
Contributions
Alexander Ciji: conceptualization; experiment execution; data analysis; manuscript’s original draft preparation; M.S. Akhtar: conceptualization; fund acquisition; experiment execution; formal analysis; manuscript review and editing; Priyanka H. Tripathi: gene expression analyses; Maneesh Kumar Dubey: histological sample processing; Prakash Sharma: histological data interpretations; manuscript reviewing and editing.
Corresponding author
Ethics declarations
Ethical approval
This research complies with current animal welfare laws in India, and the animals were used in accordance with the CPCSEA ((Committee for the Purpose of Control and Supervision of Experiments on Animals), Ministry of Environment & Forests (Animal Welfare Division), Govt. of India) guidelines on animal care and use in scientific research. The study was carried out with the approval of competent authorities of the ICAR-Directorate of Coldwater Fisheries Research at Bhimtal, Uttarakhand, India.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ciji, A., Akhtar, M.S., Tripathi, P.H. et al. Higher intake of β-glucan impairs reproduction in a female teleost, Tor putitora (Hamilton, 1822). Fish Physiol Biochem 50, 589–603 (2024). https://doi.org/10.1007/s10695-023-01292-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01292-4