Abstract
Reliable short-term chilled sperm storage is a critical prerequisite to using advanced reproductive techniques for captive breeding of barramundi (Asian sea bass; Lates calcarifer). Marine Ringer's solution (MRS) is a common non-activating medium (NAM) and has previously been used to store sperm from wild-caught barramundi. However, MRS-stored spermatozoa from captive-bred barramundi were observed to lyse within 30 min incubation. Therefore, this study aimed to optimize the composition of NAM for short-term chilled storage by characterizing and mimicking the biochemical profile of seminal and blood plasma of captive-bred barramundi. To further understand the effect of each component, osmolality was first examined to determine its effect on sperm viability. Thereafter, the effects of NaHCO3, pH, and Na+ and K+ concentrations on sperm motility were investigated. Optimization of the NAM formula was achieved through iterative adaptions. The increase in NAM osmolality from 260 to 400 mOsm/kg led to a significant improvement in sperm viability. Moreover, using HEPES instead of NaHCO3 as buffering agent significantly enhanced sperm motility and velocity. As a result, sperm samples diluted with optimized NAM (185 mM NaCl, 5.1 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 10.0 mM HEPES, 5.6 mM D+ glucose, 400 mOsm/kg, pH 7.4) and stored at 4 °C showed no significant loss in total motility for up to 48 h and retained progressive motility for up to 72 h. The optimized NAM developed in this study significantly extended the functional lifespan of spermatozoa during chilled storage, permitting the ongoing development of advanced reproductive technologies for barramundi.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
Barramundi (Asian sea bass; Lates calcarifer) is an aquaculture species of commercial importance farmed throughout Australia, Southeast Asia, and increasingly globally (Moore 1979). Over the last decade, the increasing domestic demand for Australian-farmed barramundi has outstripped production (Savage 2016). Thus, the Australian barramundi aquaculture industry has set the ambitious goal of meeting market demand by 2025 and increasing annual production from 9,000 to 25,000 tonnes (FRDC 2020). One key driver to achieving this goal is to implement selective breeding programs to enhance traits of commercial interest. However, the operational efficiency of current selective breeding programs is hindered by complications associated with barramundi's (1) mass-spawning reproductive strategy (i.e., skewed parental contribution, necessitating DNA testing, etc.) (Robinson et al. 2010; Domingos et al. 2013, 2014, 2021) and (2) ability to sex change (i.e., barramundi are protandrous hermaphrodites) (Moore 1979; Budd et al. 2015, 2022b; Banh et al. 2017; Domingos et al. 2018; Budd 2020). Therefore, a priority for the barramundi industry is to achieve better control over reproduction to boost the efficiency of selective breeding programs (Robinson and Jerry 2009).
Methods to induce sex change in barramundi from male to female are now available, which will increase the efficiency of selective breeding programs by increasing the number of young broodstock females available for spawning (Banh et al. 2021a, b; Budd et al. 2022a; Guppy et al. 2022). Yet, unsynchronized gonadal maturation still occurs in males and females reared in captivity, skewing parental contribution in mass-spawning events (Frost et al. 2006; Robinson et al. 2010; Domingos et al. 2013, 2014, 2021; Loughnan et al. 2013) and preventing the reliable collection of mature spermatozoa and eggs for artificial fertilization (Marc et al. 2021; Guppy et al. 2022). Developing a reliable short-term sperm storage protocol could overcome these problems and allow temporary access to spermatozoa of barramundi broodstock (Contreras et al. 2020). Sperm storage coupled with refinement of egg-stripping and artificial fertilization will enhance control over mate pairings and, therefore, improve the effectiveness of barramundi breeding programs (Robinson et al. 2010; Domingos et al. 2013, 2014, 2021; Jerry et al. 2022).
Various non-activating media (NAM) have been used in teleosts for short-term sperm storage, including Stop-milt, Cortland, Ringer's solution, Mountib medium, and seminal-like solutions (Contreras et al. 2020). These NAM are used to ease sperm handling and enhance storage duration and are designed as a substitute for seminal plasma (Billard and Cosson 1992; Cabrita et al. 2010; Beirao et al. 2019; Contreras et al. 2020). In teleosts, seminal plasma provides spermatozoa the ability to activate and sustain motility once released from the genital tract into the external aqueous environment (Billard and Cosson 1992; Beirao et al. 2019; Cosson 2019; Dietrich et al. 2021). Seminal plasma also creates an optimal environment in the spermatic duct for spermatozoa to be stored immotile until the spawning event (Billard and Cosson 1992; Beirao et al. 2019; Cosson 2019; Dietrich et al. 2021). The seminal plasma composition differs among species (Ulloa-Rodríguez et al. 2017). For instance, the seminal plasma osmolality of teleosts ranges from 200 to 400 mOsm/kg, excluding sturgeons (i.e., 50 to 100 mOsm/kg) (Alavi and Cosson 2006; Alavi et al. 2011) and the seminal plasma pH ranges between 7.5 and 8.5 (Alavi and Cosson 2005; Islam and Akhter 2012; Cosson 2019; Contreras et al. 2020). Moreover, Na+, K+, Ca2+, and Mg2+ are the main mineral compounds in most seminal plasma, which play a role at species-specific concentrations in sperm motility-activating signaling (Cabrita et al. 2010; Beirao et al. 2019; Cosson 2019). Therefore, it is necessary to ensure the formulation of NAM is suitable for the species of interest because an ionic imbalance of NAM during chilled storage could alter sperm function or cause premature activation of motility (Dreanno et al. 1999; Alavi et al. 2004; Yang et al. 2006; Bozkurt et al. 2008; Jing et al. 2009).
Short-term chilled sperm storage (Leung 1987; Palmer et al. 1993; Vuthiphandchai et al. 2021) and artificial fertilization (Palmer et al. 1993; Haque et al. 2021; Vuthiphandchai et al. 2021) have been achieved using sperm collected from wild-caught barramundi. These studies used Ringer's solution as a base NAM (Leung 1987; Palmer et al. 1993; Vuthiphandchai et al. 2021). However, spermatozoa collected from captive-bred broodstock swelled and lysed within 30 min of incubation in marine Ringer's solution, requiring an osmolality increase from 260 to 400 mOsm/kg to prevent osmotic stress from occurring (Marc et al. 2021). Although sperm viability improved sufficiently to enable sperm quality assessment, total sperm motility remained low (mean: 24.5 ± 4.4%) (Marc et al. 2021). While low motility could be indicative of poor sperm quality in broodstock, it is necessary to ensure that the Ringer's-based NAM used to dilute sperm provides optimal conditions for triggering and maintaining sperm motility. Therefore, further investigations were required to determine the suitability of Ringer's-based NAM for handling and short-term chilled storage of captive-bred barramundi spermatozoa.
This study investigated: (i) the biochemical properties of barramundi seminal and blood plasma to gain a greater understanding of the microenvironment to which testicular spermatozoa are exposed in the sperm duct, and the general internal body physiology of captive broodstock reared according to the Australian aquaculture industry standards; (ii) the influence of the Ringer's-based NAM composition, including the effect of osmolality on sperm viability, and the effects of pH, Na+ and K+ concentrations, and NaHCO3 vs. HEPES buffering agents on sperm motility; and (iii) the ability of the optimized NAM, modified based on outcomes of (i) and (ii), to retain sperm motility after short-term chilled storage.
Methods
Animals
Captive-bred barramundi were maintained at the Marine and Aquaculture Research Facility (MARF), James Cook University, Townsville, Queensland, Australia. Two cohorts of male barramundi were used in this study: (i) broodstock (n = 8) for blood plasma (mean body weight 3.5 ± 0.2 kg, length 62.0 ± 1.4 cm, maintained in 2,500 L tanks at 28 °C, 30 ppt salinity, and 16 h light: 8 h dark cycle), and (ii) broodstock (n = 10) for seminal plasma and spermatozoa (mean body weight 3.9 ± 0.6 kg, length 70.1 ± 4.8 cm, maintained in 28,000 L tanks under standard breeding conditions at 30 °C, 30 ppt salinity, and 16 h light: 8 h dark cycle). Experiments operated under James Cook University Animal Ethics Permit (A2406).
Sample collection and preparation
Blood was collected from the caudal vein of sedated fish using 18G 3.5" spinal needles (Provet Qld Pty Ltd, Brisbane, Australia). A total blood volume of 3 mL was extracted from each fish. Blood samples were centrifuged immediately after collection, and plasma samples were held on ice before storage at -80 ℃ until analysis. Sperm samples were collected following methods described by Marc et al. (2021). Broodstock were sedated in an anaesthetic bath containing 40 mg/L iso-eugenol (AQUI-S®, New Zealand), and the gonopore of males was rinsed with distilled water before sperm collection. Sperm samples were retrieved from the gonads by cannulation, and an aliquot was assessed for quality control (i.e., presence of blood, urine contamination, premature sperm activation, or failure to activate on contact with saltwater) before being deemed suitable for inclusion in subsequent experiments.
Biochemical composition and osmolality
Blood and seminal plasma were analyzed for 12 biochemical parameters, including sodium (Na+), potassium (K+), calcium (Ca2+), magnesium (Mg2+), chlorine (Cl−), phosphate (PO43−), partial pressure of carbon dioxide (pCO2), total protein, cholesterol, glucose, triglyceride, and urea, using an Olympus AU 480 Clinical Chemistry Analyzer (Beckman Coulter, USA). Osmolality was measured by a freezing point depression osmometer (Osmomat 030; Genotec GmbH, Berlin, Germany), and pH was recorded using a micro-pH electrode (inlab ultra-micro-ISM; Mettler‐Toledo, Melbourne, Victoria, Australia).
Assessment of sperm function
Sperm motility
Sperm motility was assessed following methods described by Marc et al. (2021). Specifically, the computer-assisted sperm analysis (CASA) system consisted of an Olympus BX53/CoolLED pE-300W fluorescent phase-contrast microscope (Olympus, Tokyo, Japan) equipped with a 20 X negative phase-contrast objective, 0.5 X C-Mount adaptor, and a Basler avA1000-100gc high frame-rate area scan camera (Basler AG, Ahrensburg, Germany), coupled with AndroVision®, version 1.1 software (Minitüb GmbH, Tiefenbach, Germany). Videos of spermatozoa were recorded at 10 X final magnification.
Multiple sperm motility parameters were evaluated, including total motility (TM; %), progressive motility (PM; %), slow, medium, and fast motility (%), curvilinear velocity (VCL; μm/s), average path velocity (VAP; μm/s), straight-line velocity (VSL; μm/s), the amplitude of lateral head displacement (ALH; μm), straightness of the average path (STR; % of VSL/VAP), the linearity of the curvilinear path (LIN; % of VSL/VCL), wobble (WOB; % of VAP/VCL) and beat-cross frequency (BCF; Hz). Sperm motility categories were classified based on VCL thresholds, including total motility (VCL ≥ 15 μm/s), progressive motility (VCL ≥ 35 μm/s), slow motility (VCL ≥ 15 and < 35 μm/s), medium motility (VCL ≥ 35 and < 100 μm/s), and fast motility (VCL ≥ 100 μm/s).
Sperm motility assessment was conducted in triplicate for each fish and assessed 15 s after activation in natural saltwater (30 ppt) consisting of 404.9 mM Na+, 459.1 mM Cl−, 10.1 mM K+, 9.7 mM Ca2+, 0.1 mM PO43−, pH 8.1, and 900 mOsm/kg using a two-step dilution to a final ratio of 1:1000 (1:10 in NAM and 1:100 in saltwater). Leja 4-chamber linear-flow slides (Leja Products B. V., Nieuw Vennep, Amsterdam, Netherlands) pre-warmed at 30 ˚C were used to contain sperm samples during analysis. Sperm motility was performed on 1 s videos recorded at a frame rate of 60 fps.
Sperm viability
Sperm viability was assessed following methods described by Marc et al. (2021) using Hoechst 33,342/Propidium Iodine (PI) dual nuclear stains and flow cytometry with 20 mW, 488 nm; 27.5 mW, 635 nm; and 27 mW, 405 nm lasers (CyanADP flow cytometer, Beckman Coulter, Fullerton, CA, USA). Both FSC and SSC were set to Lin, and FL3 (PE-Texas Red) and FL6 (DAPI) were set to Log in the Detector/Amps window. Detector voltages were set to: FSC = 400 V, SSC = 600 V, FL6 = 750 V, and FL3 = 780 V.
The concentration of each sperm sample was standardized to 100 × 106 sperm/mL. An aliquot from each sample was used to generate an unstained control (U1), heat-treated Hoechst single-stained positive control (P1), heat-treated Hoechst/PI double-stained positive control (P2), and test sample. Positive controls were incubated at 70 °C for 5 min to induce perforation of the sperm plasma membrane and subsequent complete PI staining. Positive controls and test samples were each incubated at room temperature for 30 min in 10 μg/mL Hoechst 33,342 and incubated for 7 min in the dark in 10 μg/mL PI. Debris was excluded from the sperm population using side and forward scatter profiles from unstained samples. The viable sperm subpopulation was gated based on threshold quadrants set on unstained and stained positive controls. Flow cytometry data were analyzed using Summit version 4.4 software (Beckman Coulter, Fullerton, California, USA).
Experimental design
Marine Ringer's solution (MRS), described by Palmer et al. (1993), was used as a starting medium for developing an optimized NAM. MRS consisted of 124.1 mM NaCl, 5.1 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 0.1 mM NaHCO3, 2.6 mM NaH2PO4·2H2O, and 5.6 mM D+ glucose with a final osmolality of 260 mOsm/kg adjusted to pH 7.4 using 0.1 M NaOH. This study used an iterative experimental design to optimize the NAM to suit sperm samples collected from captive-bred barramundi reared in 30 ppt saltwater.
Experiment 1. Effect of NaHCO3-buffered NAM osmolality on sperm viability
In this experiment, sperm samples (n = 5) were collected from barramundi broodstock. Sperm volume was 37.4 ± 8.1 µL, sperm concentration was 29.8 ± 5.8 × 109 sperm/mL, and total sperm count was 1,144.1 ± 285.6 × 106 sperm. Each sperm sample was individually diluted 1:10 in 260 mOsm/kg NAM (MRS original osmolality control) and 300, 350, 400, and 450 mOsm/kg NAM treatments using NaCl. The range of osmolality of NaHCO3-buffered NAM was modified based on the osmolality of captive-bred barramundi seminal plasma determined in this study. Sperm viability analyses were performed after 1 h incubation at 4 ℃.
Experiment 2. Effect of NaHCO3-buffered NAM pH on sperm motility
For this experiment, the optimal NAM determined in Experiment 1 was selected, which consisted of 182.4 mM NaCl, 5.1 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 0.1 mM NaHCO3, 2.6 mM NaH2PO4·2H2O, and 5.6 mM D+ glucose with a final osmolality of 400 mOsm/kg adjusted to the required pH level using 0.1 M NaOH. For the control, NAM was adjusted to pH 7.4 based on the original formula (Palmer et al. 1993). For the treatments, NAM was adjusted to pH 6.5 to determine if acidic conditions inhibited sperm motility (Chantzaropoulos et al. 2015), pH 7.8 based on the pH of barramundi blood plasma, pH 8.1 to replicate the pH of saltwater rearing tank, and pH 8.5 to mimic the pH of natural saltwater. Sperm samples from barramundi (n = 10) were collected. Sperm volume was 8.0 ± 1.3 µL, sperm concentration was 29.9 ± 3.9 × 109 sperm/mL, and total sperm count was 245.1 ± 55.1 × 106 sperm. Each sperm sample was individually incubated at 4 ℃ for 1 h and 24 h undiluted and diluted at 1:10 in the different NAM before sperm motility was assessed. Undiluted sperm samples were activated directly following 1:1000 dilution in 30 ppt saltwater and were used as a control to quantify the effect of NAM on sperm motility.
Experiment 3. Effect of HEPES-buffered NAM pH on sperm motility
Based on Experiment 2 and Supplementary Material 1 (Fig. S1 and Table S1), we hypothesized that the NaHCO3 buffering agent might be the element inhibiting sperm motility when diluted in NAM. Therefore, the pH experiment was repeated using a zwitterionic organic chemical buffering agent, HEPES, 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid, which has improved buffering stability at low temperatures compared to NaHCO3-based buffers (Williams-Smith et al. 1977; Matthews et al. 2018). Moreover, the monobasic sodium phosphate (NaH2PO4·2H2O) buffering agent was also removed from the NAM to prevent further unwanted interactions. For this experiment, NAM consisted of 182.4 mM NaCl, 5.1 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 10.0 mM HEPES, and 5.6 mM D+ glucose with a final osmolality of 400 mOsm/kg adjusted to the required pH value using 0.1 M NaOH. Sperm samples from barramundi (n = 10) were collected. Sperm volume was 13.9 ± 3.4 µL, sperm concentration was 16.0 ± 2.5 × 109 sperm/mL, and total sperm count was 239.0 ± 69.3 × 106 sperm. Each sperm sample was individually incubated at 4 ℃ for 1 h and 24 h undiluted and diluted at 1:10 in the different NAM before sperm motility was assessed. Undiluted sperm samples were activated directly following 1:1000 dilution in saltwater and were used as a control to quantify the effect of NAM on sperm motility. In addition, based on the Experiment 2 outcome, the NaHCO3-buffered NAM adjusted at pH 6.5 was also used as a control.
Experiment 4. Effect of HEPES-buffered NAM sodium and potassium concentration on sperm motility
For this experiment, NAM consisted of NaCl and KCl, each adjusted to their required concentrations, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 10.0 mM HEPES, and 5.6 mM D+ glucose with a final osmolality of 400 mOsm/kg adjusted to pH 7.4 using 0.1 M NaOH. Different treatments were prepared with the following NaCl/KCl concentrations: 0 mM NaCl/190 mM KCl, 140 mM NaCl/50 mM KCl, 160 mM NaCl/30 mM KCl, 185 mM NaCl/5 mM KCl, and 190 mM NaCl/0 mM KCl. NAM was adjusted to pH 7.4 with 1 M KOH for the 0 mM NaCl/190 mM KCl treatment and 0.1 M NaOH for the other groups. The first and last treatments, 0 mM NaCl/190 mM KCl and 190 mM NaCl/0 mM KCl, were designed to determine whether the motility of barramundi spermatozoa was Na+ or K+ dependent. The intermediate treatments spanned the breadth of seminal plasma ionic measurements. Sperm samples from barramundi (n = 6) were collected. Sperm volume was 14.2 ± 3.4 µL, sperm concentration was 19.0 ± 2.0 × 109 sperm/mL, and total sperm count was 281.6 ± 73.9 × 106 sperm. Each sperm sample was individually incubated at 4 ℃ for 1 h and 24 h undiluted and diluted at 1:10 in the different NAM before sperm motility was assessed. Undiluted sperm samples were activated directly following 1:1000 dilution in saltwater and were used as a control to quantify the effect of NAM on sperm motility.
Experiment 5. Evaluation of the optimized NAM for short-term chilled storage
This experiment compared the motility of sperm samples that were undiluted (control) or diluted in optimized NAM. Based on Experiments 1–4, optimized NAM consisted of 185 mM NaCl, 5.0 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 10.0 mM HEPES, and 5.6 mM D+ glucose with a final osmolality of 400 mOsm/kg adjusted to pH 7.4 using 0.1 M NaOH. Sperm motility was assessed after 1, 24, 48, 72, and 96 h incubation at 4 ℃.
Statistical analysis
Statistical analyses were performed using RStudio version 1.0.153 (RStudio, Inc, Boston, MA, USA). Percentage data for motility (TM, PM, Slow, Medium, and Fast), kinetic parameters (LIN, STR, and WOB), and viability were normalized using arcsine \(\left(\sqrt{value/100}\right)\) transformation. All data were assessed for normality and homogeneity of variance using Shapiro–Wilk and Levene tests. Where data were normally distributed with homogeneous variance, one-way ANOVA, followed by Tukey's multiple comparison test, was used to test differences between treatments. A Kruskal–Wallis H test was performed when these assumptions were not met, followed by Fisher's Least Significant Difference test to compare treatment pairs. The paired samples Wilcoxon test (Wilcoxon signed-rank test) was used to evaluate significant differences between 1 and 24 h incubation for Experiments 2, 3, and 4. Kruskal–Wallis rank-sum test followed by pairwise Mann–Whitney U-test (Wilcoxon rank-sum test) with Bonferroni correction was used to assess the significance between groups at each time point for Experiment 5. Data are displayed as mean ± standard error (SEM). The level of significance was set at P < 0.05.
Results
Biochemical analysis
Ionic and metabolite concentrations of blood plasma were relatively homogenous between male barramundi. However, these parameters in seminal plasma varied substantially between individuals (Table 1). The concentration of Na+, Cl−, pCO2, glucose, triglyceride, and urea in blood and seminal plasma did not differ (Table 1). However, the concentration of K+ and PO43− were tenfold higher in seminal than blood plasma (Welch t-test, K+: t(7.02) = -8.74, P < 0.001; PO43−: t(7.02) = -6.26, P < 0.001; Table 1). Mg2+ concentration was also significantly higher in seminal than blood plasma (Wilcoxon's test, P < 0.001, r = -0.89; Table 1). The higher ionic concentration in seminal plasma is consistent with an overall 18.8% increase in osmolality (mean: 396.1 ± 13.4 mOsm/kg) over blood plasma (mean: 333.5 ± 2.1 mOsm/kg; Table 1). By contrast, Ca2+, total protein, cholesterol, and pH were significantly lower in seminal than in blood plasma (Table 1).
Experiment 1. Effect of NaHCO3-buffered NAM osmolality on sperm viability
The highest sperm viability rates occurred in NaHCO3-buffered NAM with osmolalities, closely mimicking the range of seminal plasma (350–450 mOsm/kg; Table 1; Fig. 1). In particular, viability was significantly higher after 1 h incubation in NaHCO3-buffered NAM adjusted to 400 mOsm/kg (mean: 78.2 ± 2.9%) when compared to the original NAM at 260 mOsm/kg (mean: 44.8 ± 3.3%; ANOVA, F(1, 23) = 11.18, P < 0.05; Fig. 1).
Effect of NaHCO3-buffered non-activating medium (NAM) osmolality on sperm viability. Percentage of live barramundi (Lates calcarifer) spermatozoa was analyzed using Hoechst 33,342/Propidium Iodine (PI) dual nuclear stains and flow cytometry 1 h post-incubation at 4 °C in NaHCO3-buffered NAM with an osmolality of 260 to 450 mOsm/kg (n = 5). Osmolality of NAM (Marine Ringer's solution consisted of 124.1 mM NaCl, 5.1 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 0.1 mM NaHCO3, 2.6 mM NaH2PO4·2H2O, and 5.6 mM D+ glucose at pH 7.4 and osmolality 260 mOsm/kg) was modified using NaCl. Data are mean ± SEM. Different lowercase letters indicate a significant difference between treatments (P < 0.05)
Experiment 2. Effect of NaHCO3-buffered NAM pH on sperm motility
After 1 h incubation, spermatozoa incubated in NaHCO3-buffered NAM with a pH adjusted between 7.4 and 8.5 showed a significant decline in TM compared to the control (ANOVA, F(5, 54) = 9.81, P < 0.01; Fig. 2). However, there were no significant differences in sperm swimming velocities (VCL, VSL, and VAP) between NaHCO3-buffered NAM treatments (Supplementary Table S2). After 24 h incubation, spermatozoa motility was decreased to less than 5% in NAM with pH adjusted between 7.4 and 8.5, while it was higher than 15% in control and NAM with pH of 6.5. For the same pH, TM was significantly decreased at 24 h post-incubation compared to 1 h post-incubation in NaHCO3-buffered NAM (Wilcoxon's test, Control: P = 0.002, r = 0.89; NAM pH 6.5: P = 0.0039, r = 0.854; Fig. 2; Supplementary Table S2). The motility of spermatozoa stored in NaHCO3-buffered NAM adjusted at pH 6.5 did not differ from the control at either incubation period (Fig. 2). Of note, biochemical analysis of NaHCO3-buffered NAM showed an increase of pCO2 with the rise in pH (pH 6.5: 0.0 mmHg; pH 7.4: 1.3 mmHg; pH 7.8: 2.4 mmHg; pH 8.1: 3.2 mmHg; pH 8.5: 4.5 mmHg).
Effect of NaHCO3-buffered non-activating medium (NAM) pH on sperm motility. Total motility of barramundi (Lates calcarifer) spermatozoa was analyzed at 1 and 24 h post-incubation at 4 °C in NaHCO3-buffered NAM with different pH (n = 10). Undiluted sperm was used as a control. pH of NAM (Marine Ringer's solution consisted of 182.4 mM NaCl, 5.1 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 0.1 mM NaHCO3, 2.6 mM NaH2PO4·2H2O, and 5.6 mM D+ glucose, osmolality 400 mOsm/kg) was modified using NaOH. Data are mean ± SEM. Different lowercase letters indicate a significant difference between pH treatments within an incubation period, and different capital letters indicate a significant difference between incubation periods at the same pH (P < 0.05)
Experiment 3. Effect of HEPES-buffered NAM pH on sperm motility
After 1 h incubation, sperm motility and velocity remained high, with no significant differences between NAM treatments (Fig. 3; Supplementary Table S3). Spermatozoa in the HEPES-buffered NAM adjusted at pH 7.8 showed the highest TM, followed by pH 7.4. For the same pH, TM was decreased at 24 h post-incubation compared to 1 h post-incubation (Wilcoxon's test, P = 0.031, r = 0.898; Fig. 3). However, HEPES-buffered NAM at pH 7.4 and pH 7.8 and the control retained higher TM than other treatments (ANOVA, F(6, 35) = 12.23, P < 0.001; Fig. 3; Supplementary Table S3). Spermatozoa held in HEPES-buffered NAM adjusted at pH 6.5, pH 8.1, and pH 8.5 showed a significant decline in sperm motilities (TM, PM, and Fast) and kinetics (VCL, VSL, VAP, and ALH) compared to pH 7.4 and 7.8 treatments (Supplementary Table S3). Except for BCF, there were no significant differences in sperm motility characteristics between spermatozoa held in NaHCO3- and HEPES-buffered NAM at pH 6.5 after 1 h and 24 h incubation (Fig. 3; Supplementary Table S3).
Effect of HEPES-buffered non-activating medium (NAM) pH on sperm motility. Total motility of barramundi (Lates calcarifer) spermatozoa was analyzed at 1 and 24 h post-incubation at 4 °C in HEPES-buffered NAM with different pH (n = 7). Undiluted sperm was used as a control as well as sperm diluted in the NaHCO3-buffered NAM pH 6.5 for comparison. pH of NAM (Marine Ringer's solution consisted of 182.4 mM NaCl, 5.1 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 10.0 mM HEPES, and 5.6 mM D+ glucose, osmolality 400 mOsm/kg) was modified using NaOH. Data are mean ± SEM. Different lowercase letters indicate a significant difference between pH treatments within an incubation period, and different capital letters indicate a significant difference between incubation periods at the same pH (P < 0.05)
Experiment 4. Effect of HEPES-buffered NAM sodium and potassium concentration on sperm motility
After 1 h incubation, spermatozoa incubated in Na+ free HEPES-buffered NAM showed a large decline in TM (mean: 4.6 ± 0.6%) compared to the undiluted sperm (control) and all other treatments (mean: 43.5 ± 4.5%; ANOVA, F(5, 30) = 20.88, P < 0.001; Fig. 4; Supplementary Table S4). By contrast, the K+ free (0 mM) and low K+ (5 mM) media with the highest Na+ concentrations had the highest TM, although these did not differ from control or Na+-containing HEPES-buffered NAM (mean: 56.5 ± 4.3% and 56.8 ± 5.0% respectively; Fig. 4; Supplementary Table S4). A similar trend was observed for PM, Fast, VCL, VSL, and VAP (Supplementary Table S4). In particular, the 185/5 mM NaCl/KCl medium showed the highest PM (mean: 28.5 ± 2.4%), with most spermatozoa classified as Fast (mean: 26.4 ± 2.3%) and having high overall sperm velocities (VCL: 112.1 ± 7.4 μm/s, VSL: 79.1 ± 6.8 μm/s, and VAP: 102.6 ± 7.6 μm/s). For all media containing Na+, the swimming trajectory of spermatozoa (LIN, STR, WOB, ALH, and BCF) indicated little deviation from the primary path (Supplementary Table S4). After 24 h incubation, the highest TM (mean: 47.7 ± 7.2%) was also recorded in the NAM containing 185 mM NaCl/5 mM KCl, although this did not differ from the control or NAM containing 190 mM NaCl. Furthermore, there was no significant difference between TM measured after 1 h and 24 h incubation in NAM containing 185 mM NaCl/5 mM KCl (Wilcoxon's test, P = 0.313, r = 0.469; Fig. 4; Supplementary Table S4). Of note, this experiment was initially conducted using the NaHCO3-buffered NAM at pH 7.4, 400 mOsm/kg (Supplementary Material 1, Fig. S1, and Table S1). However, the influence of NaCl and KCl concentration on sperm motility was masked due to the presence of NaHCO3 (Figs. 2 and 3; Supplementary Material 1, Fig. S1, and Table S1).
Effect of HEPES-buffered non-activating medium (NAM) NaCl and KCl concentration on sperm motility. Total motility of barramundi (Lates calcarifer) spermatozoa was analyzed at 1 and 24 h post-incubation at 4 °C in HEPES-buffered NAM with different NaCl and KCl concentrations (n = 6). Undiluted sperm was used as a control. NaCl and KCl concentrations of NAM (Marine Ringer's solution consisted of 0–190 mM NaCl, 0–190 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 10.0 mM HEPES, and 5.6 mM D+ glucose at pH 7.4 and osmolality 400 mOsm/kg) were modified accordingly to maintain osmolality of 400 mOsm/kg. Data are mean ± SEM. Different lowercase letters indicate a significant difference between concentrations within an incubation period, and different capital letters indicate a significant difference between incubation periods at the same concentration (P < 0.05)
Experiment 5. Evaluation of the optimized HEPES-buffered NAM for short-term chilled storage
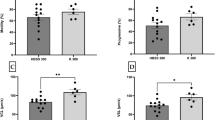
After 1 h incubation, spermatozoa in the optimized HEPES-buffered NAM showed significantly higher motilities (TM, PM, and Fast) and velocities (VCL, VSL, VAP, LIN, and WOB) than those in the undiluted sperm control (Fig. 5a and b; Supplementary Table S5). Other swimming parameters (STR, ALH, and BCF) did not differ between treatments (Supplementary Table S5). TM only declined significantly after 72 h in the optimized HEPES-buffered NAM (Kruskal–Wallis Rank Sum test; H = 17.54, df = 4, P < 0.002) compared to 48 h for the undiluted sperm control (Kruskal–Wallis Rank Sum test; H = 18.45, df = 4, P = 0.001; Fig. 5a; Supplementary Table S5). However, VCL significantly declined within 24 h in the optimized HEPES buffered NAM (Kruskal–Wallis Rank Sum test; H = 18.43, df = 4, P = 0.001; Fig. 5b; Supplementary Table S5). Of note, for both time points, the decreases in average motility and velocity were mainly caused by the loss of sperm motility in samples from two out of six fish at 48 h and three out of six fish at 72 h rather than a ubiquitous drop in motility and velocity from all six fish (Supplementary Table S6). When samples that failed activation were excluded, TM was 42.6 ± 6.0% at 48 h and 31.6 ± 2.2% at 72 h, respectively (Supplementary Table S6). After incubation of spermatozoa for 96 h, TM was less than 5%, and PM was absent for both groups (Fig. 5a; Supplementary Table S5).
Evaluation of the optimized NAM post-short-term chilled storage on sperm motility. (a) Total motility and (b) VCL of barramundi (Lates calcarifer) spermatozoa were analyzed for up to 96 h at 4 °C in an optimized HEPES-buffered non-activating medium (n = 6). Undiluted sperm was used as a control. Optimized NAM consisted of 185 mM NaCl, 5.0 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 10 mM HEPES and 5.6 mM D+ glucose at pH 7.4 and osmolality 400 mOsm/kg. Data are mean ± SEM. Different lowercase letters indicate a significant difference between extender treatments within an incubation period, and different capital letters indicate a significant difference between incubation periods for the same medium (P < 0.05)
Discussion
Reliable sperm handling and storage protocols are required to establish artificial breeding techniques (e.g., artificial fertilization and sperm banking) for the barramundi aquaculture industry. Previously published sperm handling and storage protocols were developed using sperm samples from wild-caught barramundi (Leung 1987; Palmer et al. 1993; Vuthiphandchai et al. 2021). However, these methods used Ringer's-based NAM and results could not be replicated using sperm samples collected from captive-reared broodstock. Therefore, to provide fundamental knowledge toward optimizing a suitable NAM for captive-bred barramundi, the ionic and metabolite profile of seminal plasma was characterized for the first time. Furthermore, this study investigated the effects of osmolality, pH, Na+ and K+ concentrations, and NaHCO3 vs. HEPES buffering agents on sperm motility. As a result, it was confirmed that an osmotic imbalance in previously used marine Ringer's solution caused spermatozoa to lyse, and the presence of NaHCO3 buffering agent inhibited sperm motility. Further, it was demonstrated that spermatozoa incubated in an optimized NAM (185 mM NaCl, 5.1 mM KCl, 1.6 mM CaCl2·2H2O, 1.1 mM MgSO4·7H2O, 10.0 mM HEPES, and 5.6 mM D+ glucose, 400 mOsm/kg, pH 7.4) could be stored at 4 ℃ for up to 48 h without significant loss of total motility.
Characterizing the biochemical parameters of blood and seminal plasma provided valuable insight into determining the root causes inducing sperm damage when stored in Ringer's-based NAM. The MRS used by Palmer et al. (1993) contained 124.1 mM of Na+ and had an osmolality of 260 mOsm/kg. The MRS osmolality was lower than seminal (mean: 396.1 ± 13.4 mOsm/kg) and blood plasma (mean: 333.5 ± 2.1 mOsm/kg) derived from captive-bred barramundi reared in 30 ppt saltwater. However, MRS sodium content and osmolality were similar to levels found in the blood plasma of barramundi reared in freshwater (Na+: 105.6 ± 2.8 mM and 218.3 ± 3.3 mOsm/kg; Supplementary Table S7) and seminal plasma of individuals reared in brackish water (15–17 ppt; osmolality range: 295 ± 4 to 306 ± 4 mOsm/kg) (Vuthiphandchai et al. 2021).
Palmer et al. (1993) previously used MRS as a base NAM for a cryopreservation experiment using sperm samples from wild-caught barramundi. Sperm samples incubated in MRS without cryoprotectant were not assessed for motility. However, they were used as a control group for the artificial fertilization experiment, resulting in an ~ 81% hatching rate (Palmer et al. 1993), confirming that the MRS successfully maintained barramundi sperm integrity during chilled storage. These findings directly contrasted our attempts to utilize MRS with spermatozoa collected from captive-bred barramundi reared in 30 ppt. In comparison, Palmer et al. (1993) caught barramundi in a coastal area, with the GPS coordinates of the fishing sites suggesting broodstock were captured in the freshwater plume or brackish section of the estuary. In addition, the seminal plasma osmolality was not assessed by Palmer et al. (1993) during the published experiment. However, Vuthiphandchai et al. (2021) found that the seminal plasma osmolality of barramundi reared in brackish conditions (15–17 ppt) ranged from 295 to 306 mOsm/kg, which differs from our findings (mean: 396.1 ± 13.4 mOsm/kg). While the effect of environmental salinity on seminal plasma osmolality remains to be determined in barramundi, an increase in seminal plasma osmolality with the increase in rearing salinity has been shown in tilapia, species which also has a high tolerance to a wide range of environmental salinity (Harvey and Kelley 1984; Linhart et al. 1999; Legendre et al. 2016). The seminal plasma of tilapia (Sarotherodon melanotheron heudelotii) reared in freshwater (0 ppt) was an average of 318 mOsm/kg compared to 330 mOsm/kg at 35 ppt and 349 mOsm/kg at 70 ppt (Legendre et al. 2016). A similar result was found in another tilapia species (Oreochromis mossambicus), where the osmolality of testicular seminal plasma increased by 15 mOsm/kg from 336.63 mOsm/kg to 351.0 mOsm/kg in fish reared in saltwater compared to freshwater (Linhart et al. 1999). Moreover, tilapia (O. mossambicus) transferred from freshwater to seawater showed an increase of seminal plasma osmolality from 283 mOsm/kg to 330 mOsm/kg after 30–90 days acclimation (Harvey and Kelley 1984). Consequently, we hypothesized that (1) the difference in environmental salinity is a driver of the discrepancy between Palmer et al. (1993) and our study, and (2) the optimal NAM osmolality is contingent on the rearing environment.
However, additional factors could be at the origin of the difference in seminal plasma osmolality and, thus, the suitability of the NAM; it is well documented that seminal plasma characteristics fluctuate within species. For example, in Atlantic cod (Gadus morhua), seminal plasma composition can differ significantly across spawning seasons and origin (wild-caught vs. captive-bred individuals) (Butts et al. 2010; Anthony et al. 2011). Hulak et al. (2008) also recorded a drop in seminal plasma osmolality when Northern pike (Esox Lucius) sperm samples were collected using manual stripping (mean: 273 ± 21 mOsm/kg) compared to cannulation (mean: 358 ± 77 mOsm/kg). Therefore, it will be of interest to investigate the influence of environmental salinity and collection methods (cannulation vs. manual stripping) on barramundi spermatozoa and seminal plasma to improve the chilled storage procedure.
In this study, we found that the NaHCO3 buffering agent in the NAM inhibited barramundi sperm motility. Specifically, NaHCO3 buffer caused a significant decline in motility at pH 7.4 or greater but not pH 6.5. The only discernible difference between the NAM at pH 6.5 and NAM at pH ≥ 7.4 was the presence of pCO2 in NAM at pH ≥ 7.4. It is known that NaHCO3 dissociates in an aqueous solution into CO2 + H2CO3 (free CO2), HCO3−, and CO32−, with the equilibrium ratio of each chemical species present being pH-dependent (Burt and Rau 1994). At pH 7.4 and above, free-CO2 transmutes into HCO3− and H+, whereas at pH 6.5, free-CO2 remains a gas at equilibrium. These results suggest three possible scenarios. Firstly, barramundi spermatozoa could take up HCO3− into the cytoplasm through osmosis using Cl−/HCO3− and Na+/HCO3− exchanger proteins similar to mammals (Nishigaki et al. 2014). Thus, an increase in internal HCO3− concentration will cause an intracellular accumulation of H+ through the K+ channel, leading to a drop in internal pH levels limiting further H+ uptake necessary for the initiation of motility (Tanaka et al. 2002; Inaba et al. 2003). Secondly, like in the Japanese eel, the internal increase in HCO3− and H+ could be mediated by the conversion of CO2 through carbonic anhydrase, limiting further H+ uptake (Tanaka et al. 2002). Alternatively, like in turbot (Scophthalmus maximus), sperm motility could be inhibited by HCO3− through its direct action on the flagellum axoneme, in which an intracellular increase in HCO3− concentration was mediated by CO2 conversion (Inaba et al. 2003). Lastly, in the presence of sulphate ions (SO42−), the rise in CO32− concentration in NAM adjusted at pH ≥ 7.4 could cause precipitation of Ca2+ in the form of CaCO3 (Mayorga et al. 2019), reducing Ca2+ bioavailability in NAM and inhibiting sperm motility. Biochemical analysis of our NAM did not show a difference in Ca2+ concentration at different pH levels (unpublished data). However, some precipitation during chilled storage was observed in the stock solutions that contained NaHCO3 buffers. The concentration of bioavailable Ca2+ may have differed between NAM adjusted at different pH levels during sperm storage, causing sperm motility to drop at a pH greater than 7.4. Therefore, further studies are required to elucidate the detailed molecular and ionic mechanism regulating sperm motility in barramundi.
Although the interaction of NaHCO3 with the sperm molecular pathway remains to be elucidated, in our study, we demonstrated that HEPES was a suitable substitute for NaHCO3 as a pH buffering agent. The use of the HEPES buffering agent permitted the investigation of the effect of pH on barramundi sperm motility after chilled storage. After 24 h incubation, sperm motility was significantly higher in HEPES-buffered NAM with pH 7.4 and 7.8 than in NAM at pH 6.5, 8.1, and 8.5. These results contrast slightly with what has been found in other species. For instance, in goldfish (Carassius auratus) and common carp (Cyprinus carpio), a significant decrease in sperm motility was observed when spermatozoa were stored in acidic (pH 6.5) but not alkaline conditions (pH 8.0 and 8.5) (Billard et al. 1995; Chantzaropoulos et al. 2015). These different responses could be due to the species-specific pH requirement of the mitochondrial ATP-synthase to function (Alavi and Cosson 2005; Vílchez et al. 2017). It was also noted that optimal sperm motility was found in either NAM adjusted at pH 7.4 or 7.8 and was consistent for each male barramundi over time. Changes in seminal plasma pH from 7.5 to 8.0 have been linked to the progressive maturation of spermatozoa in rainbow trout (Oncorhynchus mykiss) (Morisawa and Morisawa 1988) and Japanese eel (Billard et al. 1995; Ohta et al. 1997). In turn, variation in optimal NAM pH could indicate asynchronous sperm maturation amongst males, supporting previous observations of skewed paternity during mass-spawning (Frost et al. 2006; Robinson et al. 2010; Domingos et al. 2013, 2014, 2021; Loughnan et al. 2013; Marc et al. 2021; Guppy et al. 2022). Further studies should focus on (1) validating a mitochondrial membrane potential assay to monitor sperm mitochondrial function (e.g., JC-1 staining); and (2) characterizing sperm quality and seminal plasma characteristics of males at different gonadal maturation stages. Ultimately, this information will permit the refinement of pH in NAM and ensure optimal chilled storage of barramundi spermatozoa.
The results of the present study demonstrated the dependency of sperm motility on specific ions in barramundi for the first time. Barramundi sperm motility was inhibited within 1 h in Na+ free medium like in European eel (Vílchez et al. 2016), which contrasts with results in pufferfish (Takifugu alboplumbeus) where no decline in sperm motility was observed (Pérez et al. 2022). However, in contrast to the Japanese eel (Ohta et al. 2001) and the European eel (Vílchez et al. 2016, 2017), barramundi sperm motility improved at low K+ concentration (5 mM), similar to that in blood plasma (mean: 3.7 ± 0.1 mM), rather than at higher K+ concentration (30 mM), similar to that in seminal plasma. Although incubation in K+ free NAM did not deplete barramundi spermatozoa of their potential to become motile as it did in Japanese eel (Ohta et al. 1997), higher K+ concentration in NAM could alkalinize sperm intracellular pH, also affecting sperm mobility upon activation (Alavi et al. 2019). Other variables in seminal plasma, such as the Ca2+/K+ ratio, could also affect sperm motility performance, as demonstrated in Persian sturgeon (Acipenser persicus) (Alavi et al. 2006). Further research is needed to determine whether smaller increments of K+ between 5 and 30 mM in NAM could improve sperm motility further and how it affects sperm intracellular pH and ionic content during storage.
Overall, while the total motility achieved in the optimized NAM was substantially improved throughout this study from 20% initially to ~ 60%, it is lower than reported in many species (e.g., 90% in common carp (Cejko et al. 2018) and Prochilodus lineatus (Viveiros et al. 2016), ~ 80% in red seabream, Pagrus major (Liu et al. 2007)). Yet, it is concurrent with levels found in turbot (Suquet et al. 2000) and Japanese eel (Ohta et al. 1997) that used cannulation to collect spermatozoa. Therefore, further research is warranted to develop hormone-induced methods to strip spermatozoa for barramundi to understand better the influence of the collection method and sperm maturation on motility. Lastly, the development of hormone-induced final oocyte maturation will also enable the striping of eggs from females, thus testing the fertility potential of chilled spermatozoa.
Conclusion
This study provides an optimized NAM for the short-term chilled storage of barramundi spermatozoa collected by testicular cannulation. We demonstrated that adjusting the osmolality to 400 mOsm/kg and pH to 7.4, in line with values found in seminal plasma, was optimal for maintaining sperm viability and motility. Furthermore, the replacement of NaHCO3 with HEPES as a pH buffer was able to overcome NaHCO3−-induced inhibition of sperm motility. Finally, Na+ at 185 mM and K+ at 5 mM in the medium retained the best sperm motility activation potential in captive-bred barramundi. These results lay the foundation for future studies looking at the regulatory role of different ions in barramundi sperm motility to further improve medium composition. More importantly, this optimized NAM facilitates safe handling and short-term storage of testicular spermatozoa, thereby facilitating the development of advanced reproductive technologies, including artificial fertilization programs, for barramundi.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Code availability
Not applicable
References
Alavi SMH, Cosson J (2006) Sperm motility in fishes. (II) Effects of ions and osmolality: a review. Cell Biol Int 30:1–14. https://doi.org/10.1016/j.cellbi.2005.06.004
Alavi SMH, Cosson J (2005) Sperm motility in fishes. (I) Effects of temperature and pH: a review. Cell Biol Int 29:101–110. https://doi.org/10.1016/j.cellbi.2004.11.021
Alavi SMH, Cosson J, Bondarenko O, Linhart O (2019) Sperm motility in fishes: (III) diversity of regulatory signals from membrane to the axoneme. Theriogenology 136:143–165. https://doi.org/10.1016/j.theriogenology.2019.06.038
Alavi SMH, Cosson J, Karami M et al (2004) Chemical composition and osmolality of seminal fluid of Acipenser persicus; their physiological relationship with sperm motility. Aquac Res 35:1238–1243. https://doi.org/10.1111/j.1365-2109.2004.01132.x
Alavi SMH, Cosson J, Kazemi R (2006) Semen characteristics in Acipenser persicus in relation to sequential stripping. J Appl Ichthyol 22:400–405. https://doi.org/10.1111/j.1439-0426.2007.00994.x
Alavi SMH, Gela D, Rodina M, Linhart O (2011) Roles of osmolality, calcium - potassium antagonist and calcium in activation and flagellar beating pattern of sturgeon sperm. Comp Biochem Physiol - A Mol Integr Physiol 160:166–174. https://doi.org/10.1016/j.cbpa.2011.05.026
Anthony I, Butts IAE, Alexander E et al (2011) Seminal plasma biochemistry and spermatozoa characteristics of Atlantic cod (Gadus morhua L.) of wild and cultivated origin. Comp Biochem Physiol 159:16–24. https://doi.org/10.1016/j.cbpa.2011.01.014
Banh QQ, Domingos JA, Zenger KR, Jerry DR (2017) Morphological changes and regulation of the genes dmrt1 and cyp11b during the sex differentiation of barramundi (Lates calcarifer. Aquaculture 479:75–84. https://doi.org/10.1016/j.aquaculture.2017.05.022
Banh QQT, Domingos JA, Pinto RCC et al (2021a) Dietary 17 β-oestradiol and 17 α-ethinyloestradiol alter gonadal morphology and gene expression of the two sex-related genes, dmrt1 and cyp19a1a, in juvenile barramundi (Lates calcarifer Bloch). Aquac Res 52:1414–1430. https://doi.org/10.1111/are.14996
Banh QQT, Guppy JL, Domingos JA et al (2021b) Induction of precocious females in the protandrous barramundi (Lates calcarifer) through implants containing 17β-estradiol - effects on gonadal morphology, gene expression and DNA methylation of key sex genes. Aquaculture 539:736601. https://doi.org/10.1016/j.aquaculture.2021.736601
Beirao J, Boulais M, Gallego V et al (2019) Sperm handling in aquatic animals for artificial reproduction. Theriogenology 133:161–178. https://doi.org/10.1016/j.theriogenology.2019.05.004
Billard R, Cosson J, Perchec G, Linhart O (1995) Biology of sperm and artificial reproduction in carp. Aquaculture 129:95–112. https://doi.org/10.1016/0044-8486(94)00231-C
Billard R, Cosson MP (1992) Some problems related to the assessment of sperm motility in freshwater fish. J Exp Zool 261:122–131. https://doi.org/10.1002/jez.1402610203
Bozkurt Y, Öǧretmen F, Erçin U, Yildiz Ü (2008) Seminal plasma composition and its relationship with physical spermatological parameters of Grass carp (Ctenopharyngodon idella) semen: with emphasis on sperm motility. Aquac Res 39:1666–1672. https://doi.org/10.1111/j.1365-2109.2008.02041.x
Budd AM (2020) Epigenetic effects of temperature on sex change in barramundi, Lates calcarifer. James Cook University, PhD Thesis. https://doi.org/10.25903/95mj-pk34
Budd AM, Banh QQ, Domingos JA, Jerry DR (2015) Sex control in fish: approaches, challenges and opportunities for aquaculture. J Mar Sci Eng 3:329–355. https://doi.org/10.3390/jmse3020329
Budd AM, Guppy JL, Thépot V et al (2022a) Delayed effect of low rearing temperature on gonadal DNA methylation in juvenile barramundi (Lates calcarifer). Aquaculture 560. https://doi.org/10.1016/j.aquaculture.2022.738541
Budd AM, Robins JB, Whybird O, Jerry DR (2022b) Epigenetics underpins phenotypic plasticity of protandrous sex change in fish. Ecol Evol 12:1–16. https://doi.org/10.1002/ece3.8730
Burt EE, Rau AH (1994) The determination of the level of bicarbonate, carbonate, or carbon dioxide in aqueous solutions. Drug Dev Ind Pharm 20:2955–2964. https://doi.org/10.3109/03639049409041960
Butts IAE, Litvak MK, Trippel EA (2010) Seasonal variations in seminal plasma and sperm characteristics of wild-caught and cultivated Atlantic cod, Gadus morhua. Theriogenology 73:873–885. https://doi.org/10.1016/j.theriogenology.2009.11.011
Cabrita E, Sarasquete C, Martínez-Páramo S et al (2010) Cryopreservation of fish sperm: applications and perspectives. J Appl Ichthyol 26:623–635. https://doi.org/10.1111/j.1439-0426.2010.01556.x
Cejko BI, Horváth Á, Kollár T et al (2018) Optimisation of sodium and potassium concentrations and pH in the artificial seminal plasma of common carp Cyprinus carpio L. Fish Physiol Biochem 44:1435–1442. https://doi.org/10.1007/s10695-018-0491-3
Chantzaropoulos A, Nathanailides C, Kokokiris L et al (2015) A brief exposure to low pH prior to refrigerated storage reduces the motility and viability of goldfish sperm (Carassius auratus, Linnaeus, 1758). J Appl Ichthyol 31:89–93. https://doi.org/10.1111/jai.12735
Contreras P, Dumorné K, Ulloa-Rodríguez P et al (2020) Effects of short-term storage on sperm function in fish semen: a review. Rev Aquac 12:1373–1389. https://doi.org/10.1111/raq.12387
Cosson J (2019) Fish Sperm Physiology: Structure, Factors Regulating Motility, and Motility Evaluation. In: Biological Research in Aquatic Science. IntechOpen. https://doi.org/10.5772/intechopen.85139
Dietrich MA, Judycka S, Żarski D et al (2021) Proteomic analysis of pikeperch seminal plasma provides novel insight into the testicular development of domesticated fish stocks. Animal 15. https://doi.org/10.1016/j.animal.2021.100279
Domingos JA, Budd AM, Banh QQ et al (2018) Sex-specific dmrt1 and cyp19a1 methylation and alternative splicing in gonads of the protandrous hermaphrodite barramundi. PLoS ONE 13:1–19. https://doi.org/10.1371/journal.pone.0204182
Domingos JA, Goldsbury JA, Bastos Gomes G et al (2021) Genotype by environment interactions of harvest growth traits for barramundi (Lates calcarifer) commercially farmed in marine vs. freshwater conditions. Aquaculture 532:735989. https://doi.org/10.1016/j.aquaculture.2020.735989
Domingos JA, Smith-Keune C, Jerry DR (2014) Fate of genetic diversity within and between generations and implications for DNA parentage analysis in selective breeding of mass spawners: a case study of commercially farmed barramundi, Lates calcarifer. Aquaculture 424–425:174–182. https://doi.org/10.1016/j.aquaculture.2014.01.004
Domingos JA, Smith-Keune C, Robinson N et al (2013) Heritability of harvest growth traits and genotype – environment interactions in barramundi, Lates calcarifer (Bloch). Aquaculture 402–403:66–75. https://doi.org/10.1016/j.aquaculture.2013.03.029
Dreanno C, Cosson J, Suquet M et al (1999) Effects of osmolality, morphology perturbations and intracellular nucleotide content during the movement of sea bass (Dicentrarchus labrax) spermatozoa. Reproduction 116:113–125. https://doi.org/10.1530/jrf.0.1160113
FRDC (2020) Fisheries Research and Development Corporation Annual Report 2019–20. Canberra, Australia
Frost LA, Evans BS, Jerry DR (2006) Loss of genetic diversity due to hatchery culture practices in barramundi (Lates calcarifer). Aquaculture 261:1056–1064. https://doi.org/10.1016/j.aquaculture.2006.09.004
Guppy JL, Marc AF, Jerry DR (2022) Maturation and spawning performance of hormonally-induced precocious female barramundi (Lates calcarifer) and implications of their use in selective breeding. Aquaculture 552:737991. https://doi.org/10.1016/j.aquaculture.2022.737991
Haque MA, Hossain MI, Aftabuddin S et al (2021) First onboard fertilization of Asian seabass, Lates calcarifer: Effects of egg stocking density on the fertilization, hatching and survival rate. Sci African 12:1–7. https://doi.org/10.1016/j.sciaf.2021.e00841
Harvey B, Kelley RN (1984) Control of spermatozoan motility in a euryhaline teleost acclimated to different salinities. Can J Zool 62:2674–2677. https://doi.org/10.1139/z84-389
Hulak M, Rodina M, Linhart O (2008) Characteristics of stripped and testicular Northern pike (Esox lucius) sperm: spermatozoa motility and velocity. Aquat Living Resour 21:207–212. https://doi.org/10.1051/alr:2008022
Inaba K, Dreanno C, Cosson J (2003) Control of flatfish sperm motility by CO2 and carbonic anhydrase. Cell Motil Cytoskeleton 55:174–187. https://doi.org/10.1002/cm.10119
Islam MS, Akhter T (2012) Tale of fish sperm and factors affecting sperm motility: a review. Adv Life Sci 1:11–19. https://doi.org/10.5923/j.als.20110101.03
Jerry DR, Jones DB, Lillehammer M et al (2022) Predicted strong genetic gains from the application of genomic selection to improve growth related traits in barramundi (Lates calcarifer). Aquaculture 549. https://doi.org/10.1016/j.aquaculture.2021.737761
Jing R, Huang C, Bai C et al (2009) Optimization of activation, collection, dilution, and storage methods for zebrafish sperm. Aquaculture 290:165–171. https://doi.org/10.1016/j.aquaculture.2009.02.027
Legendre M, Alavi SMH, Dzyuba B et al (2016) Adaptations of semen characteristics and sperm motility to harsh salinity: extreme situations encountered by the euryhaline tilapia Sarotherodon melanotheron heudelotii (Dumeril, 1859). Theriogenology 86:1251–1267. https://doi.org/10.1016/j.theriogenology.2016.04.066
Leung LKP (1987) Cryopreservation of spermatozoa of the barramundi, Lates calcarifer (Teleostei: Centropomidae). Aquaculture 64:243–247. https://doi.org/10.1016/0044-8486(87)90329-2
Linhart O, Walford J, Sivaloganathan B, Lam TJ (1999) Effects of osmolality and ions on the motility of stripped and testicular sperm of freshwater- and seawater-acclimated tilapia, Oreochromis mossambicus. J Fish Biol 55:1344–1358. https://doi.org/10.1006/jfbi.1999.1133
Liu QH, Li J, Xiao ZZ et al (2007) Use of computer-assisted sperm analysis (CASA) to evaluate the quality of cryopreserved sperm in red seabream (Pagrus major). Aquaculture 263:20–25. https://doi.org/10.1016/j.aquaculture.2006.11.017
Loughnan SR, Domingos JA, Smith-Keune C et al (2013) Broodstock contribution after mass spawning and size grading in barramundi (Lates calcarifer, Bloch). Aquaculture 404–405:139–149. https://doi.org/10.1016/j.aquaculture.2013.04.014
Marc AF, Guppy JL, Bauer P et al (2021) Validation of advanced tools to evaluate sperm function in barramundi (Lates calcarifer). Aquaculture 531:735802. https://doi.org/10.1016/j.aquaculture.2020.735802
Matthews JL, Murphy JM, Carmichael C et al (2018) Changes to extender, cryoprotective medium, and in vitro fertilization improve zebrafish sperm cryopreservation. Zebrafish 15:279–290. https://doi.org/10.1089/zeb.2017.1521
Mayorga IC, Astilleros JM, Fernández-Díaz L (2019) Precipitation of CaCO3 polymorphs from aqueous solutions: the role of pH and sulphate groups. Minerals 9:1–16. https://doi.org/10.3390/min9030178
Moore R (1979) Natural Sex Inversion in the Giant Perch (Lates calcarifer). Mar Freshw Res 30:803. https://doi.org/10.1071/MF9790803
Morisawa S, Morisawa M (1988) Induction of potential for sperm motility by bicarbonate and pH in rainbow trout and chum salmon. J Exp Biol 136:13–22. https://doi.org/10.1242/jeb.136.1.13
Nishigaki T, José O, González-Cota AL et al (2014) Intracellular pH in sperm physiology. Biochem Biophys Res Commun 450:1149–1158. https://doi.org/10.1016/j.bbrc.2014.05.100
Ohta H, Ikeda K, Izawa T (1997) Increases in concentrations of potassium and bicarbonate ions promote acquisition of motility in vitro by Japanese eel spermatozoa. J Exp Zool 277:171–180. https://doi.org/10.1002/(SICI)1097-010X(19970201)277:2%3c171::AID-JEZ9%3e3.0.CO;2-M
Ohta H, Kagawa H, Tanaka H, Unuma T (2001) Control by the environmental concentration of ions of the potential for motility in Japanese eel spermatozoa. Aquaculture 198:339–351. https://doi.org/10.1016/S0044-8486(00)00597-4
Palmer PJ, Blackshaw AW, Garrett RN (1993) Successful fertility experiments with cryopreserved spermatozoa of barramundi, Lates calcarifer (Bloch), using dimethylsulfoxide and glycerol as cryoprotectants. Reprod Fertil Dev 5:285–293. https://doi.org/10.1071/RD9930285
Pérez L, Asturiano JF, Yoshida M, Gallego V (2022) Ionic control of sperm motility and trials for the improvement of pufferfish (Takifugu alboplumbeus) sperm extenders. Aquaculture 554. https://doi.org/10.1016/j.aquaculture.2022.738146
Robinson NA, Jerry DR (2009) Development of a genetic management and improvement strategy for Australian cultured barramundi. Final report to the Australian Seafood CRC. Project No. 2008/758. Flinders University
Robinson NA, Schipp G, Bosmans J, Jerry DR (2010) Modelling selective breeding in protandrous, batch-reared Asian sea bass (Lates calcarifer, Bloch) using walkback selection. Aquac Res 41:643–655. https://doi.org/10.1111/j.1365-2109.2010.02584.x
Savage J (2016) Australian fisheries and aquaculture statistics 2015. Canberra, Australia
Suquet M, Dreanno C, Fauvel C et al (2000) Cryopreservation of sperm in marine fish. Aquac Res 31:231–243. https://doi.org/10.1046/j.1365-2109.2000.00445.x
Tanaka S, Zhang H, Yamada Y et al (2002) Inhibitory effect of sodium bicarbonate on the motility of sperm of Japanese eel. J Fish Biol 60:1134–1141. https://doi.org/10.1006/jfbi.2002.1924
Ulloa-Rodríguez P, Figueroa E, Díaz R et al (2017) Mitochondria in teleost spermatozoa. Mitochondrion 34:49–55. https://doi.org/10.1016/j.mito.2017.01.001
Vílchez MC, Morini M, Peñaranda DS et al (2016) Sodium affects the sperm motility in the European eel. Comp Biochem Physiol - Part A Mol Integr Physiol 198:51–58. https://doi.org/10.1016/j.cbpa.2016.04.008
Vílchez MC, Morini M, Peñaranda DS et al (2017) Role of potassium and pH on the initiation of sperm motility in the European eel. Comp Biochem Physiol -Part A Mol Integr Physiol 203:210–219. https://doi.org/10.1016/j.cbpa.2016.09.024
Viveiros ATM, Leal MC, França TS et al (2016) Osmolality and composition of the activating solution affects motility of fresh and frozen Prochilodus lineatus sperm differently. Anim Reprod Sci 173:73–79. https://doi.org/10.1016/j.anireprosci.2016.08.014
Vuthiphandchai V, Boonthai T, Nimrat S (2021) Chilled storage of Asian seabass, Lates calcarifer Bloch semen: Effects of ions, extenders and storage periods on sperm quality and fertilization ability. J Appl Ichthyol 37:593–600. https://doi.org/10.1111/jai.14218
Williams-Smith DL, Bray RC, Barber MJ et al (1977) Changes in apparent pH on freezing aqueous buffer solutions and their relevance to biochemical electron-paramagnetic-resonance spectroscopy. Biochem J 167:593–600. https://doi.org/10.1042/bj1670593
Yang H, Hazlewood L, Walter RB, Tiersch TR (2006) Effect of osmotic immobilization on refrigerated storage and cryopreservation of sperm from a viviparous fish, the green swordtail Xiphophorus helleri. Cryobiology 52:209–218. https://doi.org/10.1016/j.cryobiol.2005.11.002
Acknowledgements
The authors thank Mainstream Aquaculture Group Pty Ltd for financial support and access to broodstock, technical staff from the Marine and Aquaculture Research Facility for their additional support, and Dr Diana Mendez (JCU Doctoral Cohort Studies Program) for her additional guidance in the writing process.
Funding
This study was funded by a James Cook University Large Partnership Grant to DP in collaboration with Mainstream Aquaculture Group Pty Ltd. AM was supported by JCU Postgraduate Research and Advance Queensland industry PhD scholarships. JG was supported by the Australian Postgraduate Awards scholarship.
Author information
Authors and Affiliations
Contributions
AM and DP conceptualized the project. AM, JG, HM, DR, and DP contributed to the development of the methodology. AM and JG conducted the experiment. AM, JG, and HM processed the samples. AM, JG, and DJ managed project administration. DJ, DR, and DP supervised the project. AM analyzed the data and generated the visuals. AM wrote the manuscript. All authors read, reviewed, and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All activities relating to the care and use of animals were conducted in compliance with the Australian code for the care and use of animals for scientific purposes (https://www.nhmrc.gov.au/about-us/publications/australian-code-care-and-use-animals-scientific-purposes) in line with state legislation and university policy. Experiments and animal protocols were approved by the James Cook University Animal Ethics Committee (Ethics Permit: A2406).
Consent to participate
All authors gave their consent to participate.
Consent for publication
All authors gave their consent to publish this manuscript.
Competing interests
The authors declare no competing interests.
Conflict of interests
The authors declare no competing interests.
Disclaimer
The funding sources provided financial support for the experiments described but had no role in the design of the study or collection, analysis, and interpretation of data or in writing the manuscript.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Donna Rudd and Damien B. B. P. Paris the work was jointly conducted in the laboratories of these two senior authors
Key points
– The biochemical composition of seminal plasma was characterized for the first time in barramundi.
– Increasing osmolality in the non-activating medium to levels mimicking seminal plasma was key to maintaining viability during short-term chilled storage of barramundi spermatozoa.
– Substitution of NaHCO3 with HEPES buffering agent improved sperm motility after storage in a non-activating medium.
– An optimized non-activating medium containing 185 mM Na+/5 mM K+, HEPES buffer at pH 7.4 and 400 mOsm/kg was found to be essential for optimal viability and motility of barramundi spermatozoa after short-term chilled storage for up to 48 h, thereby facilitating further development of advanced reproductive technologies for the species.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Marc, A.F., Guppy, J.L., Marshall, H. et al. Optimization of a non-activating medium for short-term chilled storage of barramundi (Lates calcarifer) testicular spermatozoa. Fish Physiol Biochem 49, 559–576 (2023). https://doi.org/10.1007/s10695-023-01191-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01191-8