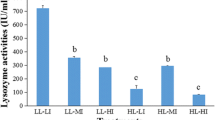
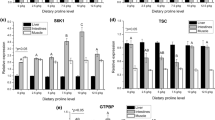
Abstract
The purpose of this research is to explore the interaction between dietary leucine and isoleucine levels on whole-body composition, plasma and liver biochemical indexes, amino acids deposition in the liver, and amino acid metabolism of blunt snout bream (Megalobrama amblycephala). The test fish (average weight: 56.00 ± 0.55 g) were fed one of six diets at random containing two leucine levels (1.70% and 2.50%) and three isoleucine levels (1.00%, 1.20%, and 1.40%) for 8 weeks. The results showed that the final weight and weight gain rate were the highest in the fish fed low-level leucine and high-level isoleucine diets (P > 0.05). Furthermore, the crude lipid content was significantly adjusted by diets with diverse levels of leucine and isoleucine (P < 0.05). In addition, interactive effects of these two branched-chain amino acids (BCAAs) were found on plasma total protein, blood ammonia, and blood urea nitrogen of test fish (P < 0.05). Additionally, the liver amino acid profiles were significantly influenced by the interactive effects of the two BCAAs (P < 0.05). Moreover, interactive effects of dietary leucine and isoleucine were significantly observed in the expressions of amino acid metabolism-related genes (P < 0.05). These findings suggested that dietary leucine and isoleucine had interaction. Meanwhile, the interaction between them was more conducive to the growth and quality improvement of blunt snout bream when the dietary leucine level was 1.70% and isoleucine level was 1.40%.



Similar content being viewed by others
Data availability
The data sets supporting the results of this article are included within the article and its additional files.
Code availability
Not applicable.
References
Ahmad I, Ahmed I, Fatma S, Peres H (2021) Role of branched-chain amino acids on growth, physiology and metabolism of different fish species: a review. Aquac Nutr 27:1270–1289. https://doi.org/10.1111/anu.13267
Alam S, Teshima S-I, Yaniharto D, Koshio S, Ishikawa M (2002) Influence of different dietary amino acid patterns on growth and body composition of juvenile Japanese flounder, Paralichthys olivaceus. Aquaculture 210:359–369. https://doi.org/10.1016/s0044-8486(01)00892-4
AOAC (1990) Official methods of analysis: 15th edition. TrAC - Trends Analyt Chem 9(4):VI
Balasubramanian MN, Shan J, Kilberg MS (2013) Dynamic changes in genomic histone association and modification during activation of the ASNS and ATF3 genes by amino acid limitation. Biochem J 449:219–229. https://doi.org/10.1042/BJ20120958
Ball RO, Urschel KL, Pencharz PB (2007) Nutritional consequences of interspecies differences in arginine and lysine metabolism. J Nutr 137:1626S-1641S. https://doi.org/10.1093/jn/137.6.1626S
Benevenga NJ, Gahl MJ, Blemings KP (1993) Role of protein synthesis in amino acid catabolism. J Nutr 123:332–336. https://doi.org/10.1093/jn/123.suppl_2.332
Biasato I, Chemello G, Caimi C, Bellezza Oddon S, Capucchio MT, Colombino E, Schiavone A, Ceccotti C, Terova G, Gasco L (2022) Taurine supplementation in plant-based diets for juvenile rainbow trout (Oncorhynchus mykiss): effects on growth performance, whole body composition, and histomorphological features. Anim Feed Sci Technol 289:115314. https://doi.org/10.1016/j.anifeedsci.2022.115314
Biswas D, Duffley L, Pulinilkunnil T (2019) Role of branched-chain amino acid-catabolizing enzymes in intertissue signaling, metabolic remodeling, and energy homeostasis. FASEB J 33:8711–8731. https://doi.org/10.1096/fj.201802842RR
Brosnan JT, Brosnan ME (2006) Branched-chain amino acids: enzyme and substrate regulation. J Nutr 136:207S-211S. https://doi.org/10.1093/jn/136.1.207S
Cemin HS, Tokach MD, Woodworth JC, Dritz SS, DeRouchey JM, Goodband RD (2019) Branched-chain amino acid interactions in growing pig diets. Transl Animal Sci 3:1246–1253. https://doi.org/10.1093/tas/txz087
Chance RE, Mertz ET, Halver JE (1964) Nutrition of salmonoid fishes. Xii. Isoleucine, leucine, valine and phenylalanine requirements of chinook salmon and inter-relations between isoleucine and leucine for growth. J Nutr 83:177–185. https://doi.org/10.1093/jn/83.3.177
Cheng CR, Wang JT, Han T, Li XY, Jiang YD, Wen XB (2016) Effects of different dietary amino acid patterns on growth performance and body composition of juvenile giant croaker Nibea japonica. Aquac Res 47:3942–3951. https://doi.org/10.1111/are.12844
Chevalier P, Aschkenasy A (1977) Hematological and immunological effects of excess dietary leucine in the young rat. Am J Clin Nutr 30:1645–1654. https://doi.org/10.1093/ajcn/30.10.1645
Chew SF, Ip YK (2014) Excretory nitrogen metabolism and defence against ammonia toxicity in air-breathing fishes. J Fish Biol 84:603–638. https://doi.org/10.1111/jfb.12279
Chikhou FH, Moloney AP, Allen P, Quirke JF, Austin FH, Roche JF (1993) Long-term effects of cimaterol in Friesian steers: I. Growth, feed efficiency, and selected carcass traits. J Anim Sci 71:906–913. https://doi.org/10.2527/1993.714906x
Choo PS, Smith TK, Cho CY, Ferguson HW (1991) Dietary excesses of leucine influence growth and body composition of rainbow trout. J Nutr 121:1932–1939. https://doi.org/10.1093/jn/121.12.1932
Colvin LB, Brand CW (2009) The protein requirement of penaeid shrimp at various life-cycle stages in controlled environment systems. Proc Annual Meet - World Mariculture Soc 8:821–840. https://doi.org/10.1111/j.1749-7345.1977.tb00164.x
Coma J, Carrion D, Zimmerman DR (1995) Use of plasma urea nitrogen as a rapid response criterion to determine the lysine requirement of pigs. J Anim Sci 73:472–481. https://doi.org/10.2527/1995.732472x
Conceicao LE, Aragao C, Dias J, Costas B, Terova G, Martins C, Tort L (2012) Dietary nitrogen and fish welfare. Fish Physiol Biochem 38:119–141. https://doi.org/10.1007/s10695-011-9592-y
Couch JR, Trammell JH, Cooper J, Farr FM, Abbott WW (1972) The effect of feeding a sub-optimum level of lysine to female chicks on the amino acid content of selected tissues and organs. Br Poult Sci 13:425–434. https://doi.org/10.1080/00071667208415970
D’Mello JP, Lewis D (1970) Amino acid interactions in chick nutrition. 2. Interrelationships between leucine, isoleucine and valine. Br Poult Sci 11:313–323. https://doi.org/10.1080/00071667008415821
Dong Y, Yu M, Wu Y, Xia T, Wang L, Song K, Zhang C, Lu K, Rahimnejad S (2022) Hydroxytyrosol promotes the mitochondrial function through activating mitophagy. Antioxidants 11. https://doi.org/10.3390/antiox11050893
Dong YZ, Li L, Espe M, Lu KL, Rahimnejad S (2020) Hydroxytyrosol attenuates hepatic fat accumulation via activating mitochondrial biogenesis and autophagy through the AMPK pathway. J Agric Food Chem 68:9377–9386. https://doi.org/10.1021/acs.jafc.0c03310
Duan Y, Li F, Li Y, Tang Y, Kong X, Feng Z, Anthony TG, Watford M, Hou Y, Wu G, Yin Y (2015) The role of leucine and its metabolites in protein and energy metabolism. Amino Acids 48:41–51. https://doi.org/10.1007/s00726-015-2067-1
Elliott JM (1976) The energetics of feeding, metabolism and growth of brown trout (Salmo trutta L.) in relation to body weight, water temperature and ration size. J Anim Ecol 45:923–948. https://doi.org/10.2307/3590
Farhat, Khan MA (2014) Response of fingerling stinging catfish, Heteropneustes fossilis (Bloch) to varying levels of dietary L-leucine in relation to growth, feed conversion, protein utilization, leucine retention and blood parameters. Aquac Nutr 20:291–302. https://doi.org/10.1111/anu.12077
Featherston WR (1972) Effect of diet on levels of amino acids in plasma and tissues. Poultry Sci 51:17–27
Gao Z, Luo W, Liu H, Zeng C, Liu X, Yi S, Wang W (2012) Transcriptome analysis and SSR/SNP markers information of the blunt snout bream (Megalobrama amblycephala). Plos One 7:e42637. https://doi.org/10.1371/journal.pone.0042637
Ge YP, Chen WL, Sun M, Zhang L, Liu WB, Li XF (2022) Molecular characterization of farnesoid X receptor alpha in Megalobrama amblycephala and its potential roles in high-carbohydrate diet-induced alterations of bile acid metabolism. J Steroid Biochem Mol Biol 219:106065. https://doi.org/10.1016/j.jsbmb.2022.106065
Hambraeus L, Bilmazes C, Dippel C, Scrimshaw N, Young VR (1976) Regulatory role of dietary leucine on plasma branched-chain amino acid levels in young men. J Nutr 106:230–240. https://doi.org/10.1093/jn/106.2.230
Han YZ, Han RZ, Koshio S, Ishikawa M, Yokoyama S, Gao J (2014) Interactive effects of dietary valine and leucine on two sizes of Japanese flounder Paralichthys olivaceus. Aquaculture 432:130–138. https://doi.org/10.1016/j.aquaculture.2014.05.004
Hao F, Dong S, Yang C, Li Z, Cheng Z, Zhong L, Teng L, Meng Q, Lu J, Wu F, Xie J, Teng L, Lee RJ (2019) Targeted and efficient delivery of siRNA using tunable polymeric hybrid micelles for tumor therapy. Anticancer Res 39:1169–1178. https://doi.org/10.21873/anticanres.13226
Harper AE, Benton DA, Winje ME, Elvehjem CA (1954) Leucine-isoleucine antagonism in the rat. Arch Biochem Biophys 51:523–524. https://doi.org/10.1016/0003-9861(54)90509-3
Herman MA, She P, Peroni OD, Lynch CJ, Kahn BB (2010) Adipose tissue branched chain amino acid (BCAA) metabolism modulates circulating BCAA levels. J Biol Chem 285:11348–11356. https://doi.org/10.1074/jbc.M109.075184
Huai MY, Tian LX, Liu YJ, Xu AL, Liang GY, Yang HJ (2009) Quantitative dietary threonine requirement of juvenile Pacific white shrimp, Litopenaeus vannamei (Boone) reared in low-salinity water. Aquac Res 40:904–914. https://doi.org/10.1111/j.1365-2109.2009.02181.x
Hughes SG, Rumsey GL, Nesheim MC (1983) Dietary requirements for essential branched-chain amino acids by lake trout. Trans Am Fish Soc 112:812–817. https://doi.org/10.1577/1548-8659(1983)112%3c812:Drfeba%3e2.0.Co;2
Hughes SG, Rumsey GL, Nesheim MC (1984) Effects of dietary excesses of branched-chain amino acids on the metabolism and tissue composition of lake trout (Salvelinus namaycush). Comp Biochem Physiol A Physiol 78:413–418. https://doi.org/10.1016/0300-9629(84)90569-3
Hutson SM, Islam MM, Zaganas I (2011) Interaction between glutamate dehydrogenase (GDH) and L-leucine catabolic enzymes: intersecting metabolic pathways. Neurochem Int 59:518–524. https://doi.org/10.1016/j.neuint.2011.05.001
Jia XY, He C, Jiang WB, Wen C, Gao F, Jiang GZ, Li XF, Chi C, Liu WB, Zhang DD (2021) Identification of potential pathways whereby dietary L-tryptophan ameliorates the glucose metabolic disorder of blunt snout bream through long non-coding RNAs. Aquaculture 545:737256. https://doi.org/10.1016/j.aquaculture.2021.737256
Khan MA, Abidi SF (2007) Dietary isoleucine requirement of fingerling Indian major carp, Labeo rohita (Hamilton). Aquac Nutr 13:424–430. https://doi.org/10.1111/j.1365-2095.2007.00493.x
Kilberg MS, Pan YX, Chen H, Leung-Pineda V (2005) Nutritional control of gene expression: how mammalian cells respond to amino acid limitation. Annu Rev Nutr 25:59–85. https://doi.org/10.1146/annurev.nutr.24.012003.132145
Kilberg MS, Shan J, Su N (2009) ATF4-dependent transcription mediates signaling of amino acid limitation. Trends Endocrinol Metab 20:436–443. https://doi.org/10.1016/j.tem.2009.05.008
Lei J, Feng D, Zhang Y, Dahanayaka S, Li X, Yao K, Wang J, Wu Z, Dai Z, Wu G (2012) Regulation of leucine catabolism by metabolic fuels in mammary epithelial cells. Amino Acids 43:2179–2189. https://doi.org/10.1007/s00726-012-1302-2
Lemaire P, Drai P, Mathieu A, Lemaire S, Carriere S, Giudicelli J, Lafaurie M (1991) Changes with different diets in plasma enzymes (Got, Gpt, Ldh, Alp) and plasma-lipids (cholesterol, triglycerides) of sea-bass (Dicentrarchus-labrax). Aquaculture 93:63–75. https://doi.org/10.1016/0044-8486(91)90205-L
Li P, Yin YL, Li D, Kim SW, Wu G (2007) Amino acids and immune function. Br J Nutr 98:237–252. https://doi.org/10.1017/S000711450769936X
Li P, Mai KS, Trushenski J, Wu GY (2009) New developments in fish amino acid nutrition: towards functional and environmentally oriented aquafeeds. Amino Acids 37:43–53. https://doi.org/10.1007/s00726-008-0171-1
Liang H, Habte-Tsion HM, Ge X, Ren M, Xie J, Miao L, Zhou Q, Lin Y, Pan W (2017) Dietary arginine affects the insulin signaling pathway, glucose metabolism and lipogenesis in juvenile blunt snout bream Megalobrama amblycephala. Sci Rep 7:7864. https://doi.org/10.1038/s41598-017-06104-3
Liang H, Mokrani A, Chisomo-Kasiya H, Ji K, Ge X, Ren M, Liu B, Xi B, Sun A (2019) Dietary leucine affects glucose metabolism and lipogenesis involved in TOR/PI3K/Akt signaling pathway for juvenile blunt snout bream Megalobrama amblycephala. Fish Physiol Biochem 45:719–732. https://doi.org/10.1007/s10695-018-0594-x
Liebert F (2009) Amino acid requirement studies in Oreochromis niloticus by application of principles of the diet dilution technique. J Anim Physiol Anim Nutr 93:787–793. https://doi.org/10.1111/j.1439-0396.2008.00869.x
Liu FJ, Liu YJ, Tian LX, Li XF, Zhang ZH, Yang HJ, Du ZY (2014) Quantitative dietary isoleucine requirement of juvenile Pacific white shrimp, Litopenaeus vannamei (Boone) reared in low-salinity water. Aquacult Int 22:1481–1497. https://doi.org/10.1007/s10499-014-9761-y
Liu Z, Zhao X, Jiang X, Zou S (2018) Transcription of blunt snout bream (Megalobrama amblycephala) HIF3alpha and its localization in the nucleus under both normoxic and hypoxic conditions. Biochem Biophys Res Commun 500:443–449. https://doi.org/10.1016/j.bbrc.2018.04.099
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Loewith R, Hall MN (2011) Target of rapamycin (TOR) in nutrient signaling and growth control. Genetics 189:1177–1201. https://doi.org/10.1534/genetics.111.133363
Lopez-Cervantes J, Sanchez-Machado DI, Rosas-Rodriguez JA (2006) Analysis of free amino acids in fermented shrimp waste by high-performance liquid chromatography. J Chromatogr A 1105:106–110. https://doi.org/10.1016/j.chroma.2005.08.040
Millet S, Aluwe M, Ampe B, De Campeneere S (2015) Interaction between amino acids on the performances of individually housed piglets. J Anim Physiol Anim Nutr 99:230–236. https://doi.org/10.1111/jpn.12227
Nishimura J, Masaki T, Arakawa M, Seike M, Yoshimatsu H (2010) Isoleucine prevents the accumulation of tissue triglycerides and upregulates the expression of PPARα and uncoupling protein in diet-induced obese mice. J Nutr 140:496–500. https://doi.org/10.3945/jn.109.108977
Perham RN (1991) Domains, motifs, and linkers in 2-oxo acid dehydrogenase multienzyme complexes: a paradigm in the design of a multifunctional protein. Biochemistry 30:8501–8512. https://doi.org/10.1021/bi00099a001
Reed LJ, Hackert ML (1990) Structure-function relationships in dihydrolipoamide acyltransferases. J Biol Chem 265:8971–8974. https://doi.org/10.1016/S0021-9258(19)38795-2
Ren MC, Habte-Tsion HM, Liu B, Miao LH, Ge XP, Xie J, Liang HL, Zhou QL, Pan LK (2015) Dietary leucine level affects growth performance, whole body composition, plasma parameters and relative expression of TOR and TNF-alpha in juvenile blunt snout bream, Megalobrama amblycephala. Aquaculture 448:162–168. https://doi.org/10.1016/j.aquaculture.2015.06.008
Ren M, Habte-Tsion HM, Liu B, Miao L, Ge X, Xie J, Zhou Q (2017) Dietary isoleucine requirement of juvenile blunt snout bream, Megalobrama amblycephala. Aquac Nutr 23:322–330. https://doi.org/10.1111/anu.12396
Roux PP, Topisirovic I (2012) Regulation of mRNA translation by signaling pathways. Cold Spring Harb Perspect Biol 4. https://doi.org/10.1101/cshperspect.a012252
Sarma D, Akhtar MS, Das P, Das P, Shahi N, Ciji A, Mahanta PC, Yengkokpam S, Debnath D (2013) Nutritional quality in terms of amino acid and fatty acid of five coldwater fish species: implications to human health. National Acad Sci Letters-India 36:385–391. https://doi.org/10.1007/s40009-013-0151-1
Subhadra B, Lochmann R, Rawles S, Chen RG (2006) Effect of fish-meal replacement with poultry by-product meal on the growth, tissue composition and hematological parameters of largemouth bass (Micropterus salmoides) fed diets containing different lipids. Aquaculture 260:221–231. https://doi.org/10.1016/j.aquaculture.2006.06.029
Suryawan A, Hawes JW, Harris RA, Shimomura Y, Jenkins AE, Hutson SM (1998) A molecular model of human branched-chain amino acid metabolism. Am J Clin Nutr 68:72–81. https://doi.org/10.1093/ajcn/68.1.72
Tacon AGJ, Metian M (2013) Fish matters: importance of aquatic foods in human nutrition and global food supply. Rev Fish Sci 21:22–38. https://doi.org/10.1080/10641262.2012.753405
Teodoro GF, Vianna D, Torres-Leal FL, Pantaleao LC, Matos-Neto EM, Donato J Jr, Tirapegui J (2012) Leucine is essential for attenuating fetal growth restriction caused by a protein-restricted diet in rats. J Nutr 142:924–930. https://doi.org/10.3945/jn.111.146266
Wang BK, Liu WB, Xu C, Cao XF, Zhong XQ, Shi HJ, Li XF (2017a) Dietary carbohydrate levels and lipid sources modulate the growth performance, fatty acid profiles and intermediary metabolism of blunt snout bream Megalobrama amblycephala in an interactive pattern. Aquaculture 481:140–153. https://doi.org/10.1016/j.aquaculture.2017.08.034
Wang CC, Liu WB, Huang YY, Wang X, Li XF, Zhang DD, Jiang GZ (2020) Dietary DHA affects muscle fiber development by activating AMPK/Sirt1 pathway in blunt snout bream (Megalobrama amblycephala). Aquaculture 518. https://doi.org/10.1016/j.aquaculture.2019.734835
Wang L, Han Y, Jiang Z, Sun M, Si B, Chen F, Bao N (2017b) Interactive effects of dietary leucine and isoleucine on growth, blood parameters, and amino acid profile of Japanese flounder Paralichthys olivaceus. Fish Physiol Biochem 43:1265–1278. https://doi.org/10.1007/s10695-017-0370-3
Wilson RP, Poe WE, Robinson EH (1980) Leucine, isoleucine, valine and histidine requirements of fingerling channel catfish. J Nutr 110:627–633. https://doi.org/10.1093/jn/110.4.627
Xiao W, Li DY, Zhu JL, Zou ZY, Yue YR, Yang H (2018) Dietary valine requirement of juvenile Nile tilapia, Oreochromis niloticus. Aquac Nutr 24:315–323. https://doi.org/10.1111/anu.12562
Yuan XY, Liu MY, Cheng HH, Huang YY, Dai YJ, Liu WB, Jiang GZ (2019) Replacing fish meal with cottonseed meal protein hydrolysate affects amino acid metabolism via AMPK/SIRT1 and TOR signaling pathway of Megalobrama amblycephala. Aquaculture 510:225–233. https://doi.org/10.1016/j.aquaculture.2019.05.056
Zhang J, Wei XL, Chen LP, Chen N, Li YH, Wang WM, Wang HL (2013) Sequence analysis and expression differentiation of chemokine receptor CXCR4b among three populations of Megalobrama amblycephala. Dev Comp Immunol 40:195–201. https://doi.org/10.1016/j.dci.2013.01.011
Zhou QL, Habte-Tsion HM, Ge X, Xie J, Ren M, Liu B, Miao L, Pan L (2018) Graded replacing fishmeal with canola meal in diets affects growth and target of rapamycin pathway gene expression of juvenile blunt snout bream, Megalobrama amblycephala. Aquac Nutr 24:300–309. https://doi.org/10.1111/anu.12560
Funding
This research was supported by the Development Plan Project in Key Areas of Guangdong Province (Project No: 2020B0202010001), the Jiangsu Natural Science Foundation for basic research (BK20201325), and the earmarked fund for CARS (CARS-45–12).
Author information
Authors and Affiliations
Contributions
Mang-mang Wang, Guang-zhen Jiang, and Wen-bin Liu provided ideas and schemes for this experiment; Mang-mang Wang and Yang-yang Huang completed the feeding trial; Mang-mang Wang, Yi-lin Zhang, and Jing-Wei Fan assisted sampling. Mang-mang Wang finished data analysis and manuscript writing; Kang Xiao, Xi Wang, Hui-xing Guo, Xiang-fei Li, and Guang-zhen Jiang polished the manuscript.
Corresponding author
Ethics declarations
Ethics approval
All tests relating to animals were carried out by the Guidance of the Care and Use of Laboratory Animals in China. This research got permission from the Animal Care and Use Committee of Nanjing Agricultural University (Nanjing, China) (permit number: SYXK (Su) 2011–0036).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Interaction between leucine and isoleucine affects the bioavailability of leucine and isoleucine in blunt snout bream.
• The interaction between leucine and isoleucine will affect their ratio in feed.
• The best combinations of leucine and isoleucine in dietary of blunt snout bream would be LL-HI.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Mm., Huang, Yy., Liu, Wb. et al. Interactive effects of dietary leucine and isoleucine affect amino acid profile and metabolism through AKT/TOR signaling pathways in blunt snout bream (Megalobrama amblycephala). Fish Physiol Biochem 50, 385–401 (2024). https://doi.org/10.1007/s10695-022-01161-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01161-6