Abstract
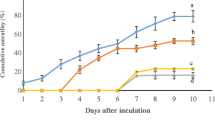
In this study, we evaluated to reveal the effects of aqueous methanolic extract of celery (Apium graveolens) on the growth performance, immune responses, and resistance against Vibrio anguillarum in European seabass (Dicentrarchus labrax). For this purpose, twenty fish (initial mean weight of 4.80 ± 0.06 g) were placed into twelve tanks (400 L) in triplicate and fish were fed with control (C) and three different levels (0.01, 0.05, and 0.1 g/kg) of A. graveolens (AG) extract-containing diets (AG0.01, AG0.05, and AG0.1) for 30 days. Blood and tissue (kidney, spleen, and intestine) samples were taken from the fish every 10 days during the study to determine the immune responses of the fish. Respiratory burst activity (RBA) was significantly decreased in the AG0.1 group compared to all other groups on the 10th day of the study (P < 0.05). Significance was noticed in the RBA of fish in all AG groups compared to the C group (P < 0.05) on the 30th day of the experiment Lysozyme activity (LYS) was raised on the 10th day of the study in all celery groups compared to the C group (P < 0.05). No differences in the myeloperoxidase activity (MPO) were observed among the experimental groups (P > 0.05). The final mean weight (FMW) was not affected in any experimental groups (P > 0.05). However, in the AG0.05 group, the specific growth rate (SGR) increased, and the feed conversion ratio (FCR) decreased compared to other groups (P < 0.05). IL-1β in the kidney was highly elevated in the AG0.01 group on the 20th day of the study (P < 0.05). Similar results were observed on IL-6, IL-8, and TNF-α expression in the kidney (P < 0.05). Anti-inflammatory responses (IL-10 and TGF-β) also increased in all experimental groups and tissues compared to the C group (P < 0.05). COX-2 was upregulated on the 20th day of the study in all tissues (P < 0.05). At the end of the feeding trial, the survival rate of the AG0.1 group in fish infected with Vibrio anguillarum infection was higher than the C group. Dietary celery extract did not affect growth performance directly but increased innate immune responses and a high survival rate. Overall, compared to the control group, the growth, immunity, and resistance of European seabass fed with a diet containing 0.05 g/kg celery aqueous methanolic extract has been improved, and this could be used as an immunostimulant feed additive.












Similar content being viewed by others
Data availability
All data generated and analyzed during this study are presented in this article.
Code availability
Not applicable.
References
Abdel-Tawwab M, Khalifa E, Diab AM, Khallaf MA, Abdel-Razek N, Khalil RH (2020) Dietary garlic and chitosan alleviated zearalenone toxic effects on performance, immunity, and challenge of European sea bass, Dicentrarchus labrax, to Vibrio alginolyticus infection. Aquac Int 28(2):493–510. https://doi.org/10.1007/s10499-019-00477-0
Afzali S, Wong W (2019) Effects of dietary supplementation of Sonneratia alba extract on immune protection and disease resistance in goldfish against Aphanomyces invadans. Trop Biomed 36(1):274–288
Ahmad A, Abdullah SRS, Hasan HA, Othman AR, Ismail NI (2021) Aquaculture industry: supply and demand, best practices, effluent and its current issues and treatment technology. J Environ Manage 287:112271. https://doi.org/10.1016/j.jenvman.2021.112271
Alexander JB, Ingram GA (1992) Noncellular nonspecific defence mechanisms of fish. Annu Rev Fish Dis 2:249–279. https://doi.org/10.1016/0959-8030(92)90066-7
Almabrok AA, Amhamed ID, Mohamed GA, Bilen S, Altief TAS (2018) Effect of Tilia tomentosa methanolic extract on growth performance, digestive enzyme activity, immune system and haematological indices of common carp (Cyprinus carpio). Mar Sci Tech Bull 7(1):12–20. https://doi.org/10.33714/masteb.421047
Amoush OA, Bilen S, Sönmez AY, Elp M (2022) Antioxidant and immunostimulant responses in rainbow trout (Oncorhynchus mykiss) fed with cherry stem extract. Aquac Res 53(2):487–496. https://doi.org/10.1111/are.15594
AOAC (2000) Official methods of analysis, 17th ed. Association of Official Analytical Chemists, Arlington, USA
Azeredo R, Machado M, Kreuz E, Wuertz S, Oliva-Teles A, Enes P, Costas B (2017) The European seabass (Dicentrarchus labrax) innate immunity and gut health are modulated by dietary plant-protein inclusion and prebiotic supplementation. Fish Shellfish Immunol 60:78–87. https://doi.org/10.1016/j.fsi.2016.11.019
Bilen S, Elbeshti HTAG (2019) A new potential therapeutic remedy against Aeromonas hydrophila infection in rainbow trout (Oncorhynchus mykiss) using tetra, Cotinus coggygria. J Fish Dis 42(10):1369–1381. https://doi.org/10.1111/jfd.13061
Bilen S, Karga M, Altunoglu YC, Ulu F, Biswas G (2020) Immune responses and growth performance of the aqueous methanolic extract of Malva sylvestris in Oncorhynchus mykiss. Mar Sci Tech Bull 9(2):159–167. https://doi.org/10.33714/masteb.746951
Bilen S, Ispir S, Kenanoglu ON, Taştan Y, Güney K, Terzi E (2021) Effects of Greek juniper (Juniperus excelsa) extract on immune responses and disease resistance against Yersinia ruckeri in rainbow trout (Oncorhynchus mykiss). J Fish Dis 44(6):729–738. https://doi.org/10.1111/jfd.13293
Cabello FC, Godfrey HP, Tomova A, Ivanova L, Dölz H, Millanao A, Buschmann AH (2013) Antimicrobial use in aquaculture re-examined: its relevance to antimicrobial resistance and to animal and human health. Environ Microbiol 15(7):1917–1942. https://doi.org/10.1111/1462-2920.12134
Campoverde C, Milne DJ, Estévez A, Duncan N, Secombes CJ, Andree KB (2017) Ontogeny and modulation after PAMPs stimulation of β-defensin, hepcidin, and piscidin antimicrobial peptides in meagre (Argyrosomus regius). Fish Shellfish Immunol 69:200–210. https://doi.org/10.1016/j.fsi.2017.08.026
Cañada-Cañada F, de la Peña AM, Espinosa-Mansilla A (2009) Analysis of antibiotics in fish samples. Anal Bioanal Chem 395(4):987–1008. https://doi.org/10.1007/s00216-009-2872-z
Capkin E, Terzi E, Altinok I (2015) Occurrence of antibiotic resistance genes in culturable bacteria isolated from Turkish trout farms and their local aquatic environment. Dis Aquat Org 114(2):127–137. https://doi.org/10.3354/dao02852
Castro R, Piazzon MC, Noya M, Leiro JM, Lamas J (2008) Isolation and molecular cloning of a fish myeloperoxidase. Mol Immunol 45(2):428–437. https://doi.org/10.1016/j.molimm.2007.05.028
Ellis AE (1990) Lysozyme assays. In: Stolen JS, Fletcher TC, Anderson DP, Roberson BS, Muiswinkel WB (eds) Techniques in Fish Immunology. SOS Publication, pp 101–103
FAO (2022) Dicentrarchus labrax. Cultured Aquatic Species Information Programme. Text by Bagni M. Fisheries and Aquaculture Division [online]. Rome. Updated 2005–04–06 [Cited Friday, June 17th 2022]
Frieri M, Kumar K, Boutin A (2017) Antibiotic resistance. J Infect Public Health 10(4):369–378. https://doi.org/10.1016/j.jiph.2016.08.007
Fu X, Ding Z, Fan J, Wang H, Zhou F, Cui L, Boxiang C, Wang W, Liu H (2016) Characterization, promoter analysis and expression of the interleukin-6 gene in blunt snout bream, Megalobrama amblycephala. Fish Physiol Biochem 42(6):1527–1540. https://doi.org/10.1007/s10695-016-0238-y
Guardiola FA, Porcino C, Cerezuela R, Cuesta A, Faggio C, Esteban MA (2016) Impact of date palm fruits extracts and probiotic enriched diet on antioxidant status, innate immune response and immune-related gene expression of European seabass (Dicentrarchus labrax). Fish Shellfish Immunol 52:298–308. https://doi.org/10.1016/j.fsi.2016.03.152
Habiba MM, Hussein EE, Ashry AM, El-Zayat AM, Hassan AM, El-Shehawi AM, Sewilam H, Van Doan H, Dawood MA (2021) Dietary cinnamon successfully enhanced the growth performance, growth hormone, antibacterial capacity, and immunity of European sea bass (Dicentrarchus labrax). Animals 11(7):2128. https://doi.org/10.3390/ani11072128
Hassanen NH, Eissa A, Hafez S, Mosa EA (2015) Antioxidant and antimicrobial activity of celery (Apium graveolens) and coriander (Coriandrum sativum) herb and seed essential oils. Int J Curr Microbiol Appl Sci 4(3):284–296
Jørgensen JB, Lunde H, Jensen L, Whitehead AS, Robertsen B (2000) Serum amyloid A transcription in Atlantic salmon (Salmo salar L.) hepatocytes is enhanced by stimulation with macrophage factors, recombinant human IL-1β, IL-6 and TNFα or bacterial lipopolysaccharide. Dev Comp Immunol 24(6–7):553–563. https://doi.org/10.1016/S0145-305X(00)00022-7
Karga M, Kenanoğlu ON, Bilen S (2020) Investigation of antibacterial activity of two different medicinal plants extracts against fish pathogens. J Agric Prod 1(1):5–7. https://doi.org/10.29329/agripro.2020.341.2
Kumar V, Bossier P (2018) Importance of plant-derived compounds and/or natural products in aquaculture. Aquafeed Adv Process Formulation 10(3):28–31
Lakwani MA, Kenanoğlu ON, Taştan Y, Bilen S (2022) Effects of black mustard (Brassica nigra) seed oil on growth performance, digestive enzyme activities and immune responses in rainbow trout (Oncorhynchus mykiss). Aquac Res 53(1):300–313. https://doi.org/10.1111/are.15577
Lee M, Lee H, Ryu P (2001) Public health risks: chemical and antibiotic residues-review. Asian Australas J Anim Sci 14(3):402–413. https://doi.org/10.5713/ajas.2001.402
Lees M, Thomas F (2008) Confirming the origin of wild and farmed fish. In Ø Lie (Ed.), Improving farmed fish quality and safety (pp. 565–584), Elsevier Publishing. https://doi.org/10.1533/9781845694920.3.565
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Lobo G, Pereira LF, Gonçalves JFM, Peixoto MJ, Ozório ROA (2018) Effect of dietary seaweed supplementation on growth performance, antioxidant and immune responses in European seabass (Dicentrarchus labrax) subjected to rearing temperature and salinity oscillations. Int Aquat Res 10(4):321–331. https://doi.org/10.1007/s40071-018-0208-3
Magnadóttir B (2006) Innate immunity of fish (overview). Fish Shellfish Immunol 20(2):137–151. https://doi.org/10.1016/j.fsi.2004.09.006
Mehrabi Z, Firouzbakhsh F, Rahimi-Mianji G, Paknejad H (2020) Immunity and growth improvement of rainbow trout (Oncorhynchus mykiss) fed dietary nettle (Urtica dioica) against experimental challenge with Saprolegnia parasitica. Fish Shellfish Immunol 104:74–82. https://doi.org/10.1016/j.fsi.2020.05.050
Mohamed GA, Amhamed ID, Almabrok AA, Barka ABA, Bilen S, Elbeshti RT (2018) Effect of celery (Apium graveolens) extract on the growth, haematology, immune response and digestive enzyme activity of common carp (Cyprinus carpio). Mar Sci Tech Bull 7(2):51–59. https://doi.org/10.33714/masteb.457721
Mu H, Shen H, Liu J, Xie F, Zhang W, Mai K (2018) High level of dietary soybean oil depresses the growth and anti-oxidative capacity and induces inflammatory response in large yellow croaker Larimichthys crocea. Fish Shellfish Immunol 77:465–473. https://doi.org/10.1016/j.fsi.2018.04.017
Muniesa A, Basurco B, Aguilera C, Furones D, Reverté C, Sanjuan-Vilaplana A, Jansen MD, Brun E, Tavornpanich S (2020) Mapping the knowledge of the main diseases affecting sea bass and sea bream in Mediterranean. Transbound Emerg Dis 67(3):1089–1100. https://doi.org/10.1111/tbed.13482
Nootash S, Sheikhzadeh N, Baradaran B, Oushani AK, Moghadam MRM, Nofouzi K, Monfaredan A, Aghebati L, Zare F, Shabanzadeh S (2013) Green tea (Camellia sinensis) administration induces expression of immune relevant genes and biochemical parameters in rainbow trout (Oncorhynchus mykiss). Fish Shellfish Immunol 35(6):1916–1923. https://doi.org/10.1016/j.fsi.2013.09.030
Okocha RC, Olatoye IO, Adedeji OB (2018) Food safety impacts of antimicrobial use and their residues in aquaculture. Public Health Rev 39:21. https://doi.org/10.1186/s40985-018-0099-2
Peixoto MJ, Ferraz R, Magnoni LJ, Pereira R, Gonçalves JF, Calduch-Giner J, Pérez-Sánchez J, Ozório ROA (2019) Protective effects of seaweed supplemented diet on antioxidant and immune responses in European seabass (Dicentrarchus labrax) subjected to bacterial infection. Sci Rep 9:16134. https://doi.org/10.1038/s41598-019-52693-6
Peixoto MJ, Salas-Leitón E, Pereira LF, Queiroz A, Magalhães F, Pereira R, Abreu H, Reis PA, Gonçalves JFM, de Almeida Ozório RO (2016) Role of dietary seaweed supplementation on growth performance, digestive capacity and immune and stress responsiveness in European seabass (Dicentrarchus labrax). Aquac Rep 3:189–197. https://doi.org/10.1016/j.aqrep.2016.03.005
Pérez Gutiérrez RM, Muñiz-Ramirez A, Garcia-Campoy AH, Mota Flores JM (2021) Evaluation of the antidiabetic potential of extracts of Urtica dioica, Apium graveolens, and Zingiber officinale in mice, zebrafish, and pancreatic β-Cell. Plants 10:1438. https://doi.org/10.3390/plants10071438
Picchietti S, Fausto AM, Randelli E, Carnevali O, Taddei AR, Buonocore F, Scapigliati G, Abelli L (2009) Early treatment with Lactobacillus delbrueckii strain induces an increase in intestinal T-cells and granulocytes and modulates immune-related genes of larval Dicentrarchus labrax (L.). Fish Shellfish Immunol 26(3):368–376. https://doi.org/10.1016/j.fsi.2008.10.008
Quade MJ, Roth JA (1997) A rapid, direct assay to measure degranulation of bovine neutrophil primary granules. Vet Immunol Immunopathol 58(3–4):239–248. https://doi.org/10.1016/S0165-2427(97)00048-2
Reverter M, Bontemps N, Lecchini D, Banaigs B, Sasal P (2014) Use of plant extracts in fish aquaculture as an alternative to chemotherapy: current status and future perspectives. Aquaculture 433:50–61. https://doi.org/10.1016/j.aquaculture.2014.05.048
Reyes-López FE, Aerts J, Vallejos-Vidal E, Ampe B, Dierckens K, Tort L, Bossier P (2018) Modulation of innate immune-related genes and glucocorticoid synthesis in gnotobiotic full-sibling European sea bass (Dicentrarchus labrax) larvae challenged with Vibrio anguillarum. Front Immunol 9:914. https://doi.org/10.3389/fimmu.2018.00914
Rodgers C, Furones M (2009) Antimicrobial agents in aquaculture: practice, needs and issues. Options Méditerr Série A 86:41–59
Román L, Real F, Padilla D, El Aamri F, Déniz S, Grasso V, Acosta F (2013) Cytokine expression in head-kidney leucocytes of European sea bass (Dicentrarchus labrax L.) after incubation with the probiotic Vagococcus fluvialis L-21. Fish Shellfish Immunol 35(4):1329–1332. https://doi.org/10.1016/j.fsi.2013.07.036
Salem MOA, Salem TA, Yürüten Özdemir K, Sönmez AY, Bilen S, Güney K (2021) Antioxidant enzyme activities and immune responses in rainbow trout (Onchorhynchus mykiss) juveniles fed diets supplemented with dandelion (Taraxacum officinalis) and lichen (Usnea barbata) extracts. Fish Physiol Biochem 47(4):1053–1062. https://doi.org/10.1007/s10695-021-00962-5
Secombes C, Hardie L, Daniels G (1996) Cytokines in fish: an update. Fish Shellfish Immunol 6(4):291–304. https://doi.org/10.1006/fsim.1996.0030
Sepulcre MP, Sarropoulou E, Kotoulas G, Meseguer J, Mulero V (2007) Vibrio anguillarum evades the immune response of the bony fish sea bass (Dicentrarchus labrax L.) through the inhibition of leukocyte respiratory burst and down-regulation of apoptotic caspases. Mol Immunol 44(15):3751–3757. https://doi.org/10.1016/j.molimm.2007.03.021
Shike H, Shimizu C, Lauth X, Burns JC (2004) Organization and expression analysis of the zebrafish hepcidin gene, an antimicrobial peptide gene conserved among vertebrates. Dev Comp Immunol 28(7–8):747–754. https://doi.org/10.1016/j.dci.2003.11.009
Shivashri C, Rajarajeshwari T, Rajasekar P (2013) Hepatoprotective action of celery (Apium graveolens) leaves in acetaminophen-fed freshwater fish (Pangasius sutchi). Fish Physiol Biochem 39(5):1057–1069. https://doi.org/10.1007/s10695-012-9762-6
Siwicki AK, Anderson DP, Rumsey GL (1994) Dietary intake of immunostimulants by rainbow trout affects non-specific immunity and protection against furunculosis. Vet Immunol Immunopathol 41(1–2):125–139. https://doi.org/10.1016/0165-2427(94)90062-0
Sönmez AY, Ozdemir RC, Bilen S, Kadak AE (2019) Effect of ginseng root (Araliaceae sp.) extracts on sperm quality parameters and reproductive performance in rainbow trout (Oncorhynchus mykiss). Isr J Aquac-Bamid 71:1570. https://doi.org/10.46989/001c.20992
Sönmez AY, Bi̇len S, Taştan Y, Serag KJB, Toring CC, Romero JB, Kenanoğlu ON, Terzi E (2021) Oral administration of Sargassum polycystum extracts stimulates immune response and increases survival against Aeromonas hydrophila infection in Oncorhynchus mykiss. Fish Shellfish Immunol 117:291–298. https://doi.org/10.1016/j.fsi.2021.08.020
Srivastava PK, Pandey AK (2015) Role of immunostimulants in immune responses of fish and shellfish. Biochem Cell Arch 15(1):47–73
Sutthi N, Panase A, Chitmanat C, Sookying S, Ratworawong K, Panase P (2020) Effects of dietary leaf ethanolic extract of Apium graveolens L. on growth performance, serum biochemical indices, bacterial resistance and lysozyme activity in Labeo chrysophekadion (Bleeker, 1849). Aquac Rep 18:100551. https://doi.org/10.1016/j.aqrep.2020.100551
Takaoka O, Ji SC, Ishimaru K, Lee SW, Jeong GS, Ito J, Biswas A, Takii K (2011) Effect of rotifer enrichment with herbal extracts on growth and resistance of red sea bream, Pagrus major (Temminck & Schlegel) larvae against Vibrio anguillarum. Aquac Res 42(2):1824–1829. https://doi.org/10.1111/j.1365-2109.2010.02783.x
Talpur AD, Ikhwanuddin M (2013) Azadirachta indica (neem) leaf dietary effects on the immunity response and disease resistance of Asian seabass, Lates calcarifer challenged with Vibrio harveyi. Fish Shellfish Immunol 34(1):254–264. https://doi.org/10.1016/j.fsi.2012.11.003
Terzi E, Kucukkosker B, Bilen S, Kenanoglu ON, Corum O, Özbek M, Parug SS (2021) A novel herbal immunostimulant for rainbow trout (Oncorhynchus mykiss) against Yersinia ruckeri. Fish Shellfish Immunol 110:55–66. https://doi.org/10.1016/j.fsi.2020.12.019
Tollefson L, Miller MA (2000) Antibiotic use in food animals: controlling the human health impact. J AOAC Int 83(2):245–254. https://doi.org/10.1093/jaoac/83.2.245
Toranzo AE, Magariños B, Romalde JL (2005) A review of the main bacterial fish diseases in mariculture systems. Aquaculture 246:37–61. https://doi.org/10.1016/j.aquaculture.2005.01.002
Uney K, Terzi E, Corum DD, Ozdemir RC, Bilen S, Corum O (2021) Pharmacokinetics and pharmacokinetic/pharmacodynamic integration of enrofloxacin following single oral administration of different doses in brown trout (Salmo trutta). Animals 11(11):3086. https://doi.org/10.3390/ani11113086
Vandeputte M, Gagnaire PA, Allal F (2019) The European sea bass: a key marine fish model in the wild and in aquaculture. Anim Genet 50(3):195–206. https://doi.org/10.1111/age.12779
Woodward K (1991) Hypersensitivity in humans and exposure to veterinary drugs. Vet Hum Toxicol 33(2):168–172
Zar JH (1999) Biostatistical analysis, 4th ed. (p. 929). Prentice-Hall Inc
Acknowledgements
This research was supported by the Scientific Research Projects Coordination Unit of Yalova University with the project number 2018/AP/0012. We thank Kılıç Sea Food for providing the European seabass juveniles. Special thanks go to DSM Nutritional Products, Turkey, for providing the feed additives used in this study.
Funding
This research was supported by the Scientific Research Projects Coordination Unit of Yalova University with the project number 2018/AP/0012.
Author information
Authors and Affiliations
Contributions
DG is the project leader, managed the experiment, samplings, and analyses, wrote original draft, reviewed, and edited. BG consulted the formal analyses, collected samples, and reviewed the original draft. SB consulted the experiment and methodology, collected data, wrote original draft, reviewed, and edited. ONK analyzed and tested samples and collected data. İŞ made experimental feeds, reared fish, and collected data. ET consulted the experiment and collected data. OK reared fish and reviewed and edited original draft. SM reared fish, made analyses, and collected data.
Corresponding author
Ethics declarations
Ethics approval
The experimental protocol was approved by the local ethics committee of Kastamonu University, Kastamonu, Turkey, under protocol number KUHADYEK 2019/10.
Concent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Güroy, D., Güroy, B., Bilen, S. et al. Effect of dietary celery (Apium graveolens) on the growth performance, immune responses, and bacterial resistance against Vibrio anguillarum of European seabass (Dicentrarchus labrax). Fish Physiol Biochem 49, 75–95 (2023). https://doi.org/10.1007/s10695-022-01158-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01158-1