Abstract
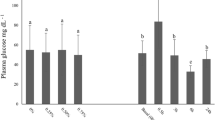
This study aimed to investigate the effects of transport stress on the physiological responses and immunity of Megalobrama amblycephala (blunt snout bream). Fish (109.67 ± 1.51 g) were sampled at nine time points: before transport (control), at 0 h, 1 h, 3 h, 6 h, 12 h, 24 h, 3 days, and 7 days after 4 h of medium-distance transportation, and four fish were sampled in each time point. The results showed that plasma cortisol, triiodothyronine (T3), complement component 3 (C3), complement component 4 (C4), immunoglobulin M (IgM) and nitrogen monoxide (NO) concentrations, and alternative complement pathway (ACH50), acid phosphatase (ACP), and myeloperoxidase (MPO) activities all reached the peak at 0 h after transportation; C4 and NO concentrations as well as ACP and MPO activities returned to the control level after 1 h, ACH50 activity as well as cortisol, T3, and IgM concentration returned to the control level after 12 h, and C3 concentration returned to the control level after 24 h respectively. Plasma glucose and total protein concentrations as well as lysozyme activity all reached the peak at 1 h after transportation, total protein concentration and lysozyme activity returned to the control level after 3 h, and glucose concentration returned to the control level after 6 h (P < 0.05). Liver heat shock protein 70 expression reached the peak at 1 h after transportation, and returned to the control level after 24 h; liver heat shock protein 90 expression reached the peak at 0 h after transportation and returned to the control level after 12 h (P < 0.05). Overall, these findings suggested that 4 h of medium-distance transportation caused stress response of blunt snout bream, and transport stress had a significant effect on plasma indicators. But the recovery of 24 h after transport could return the physiological response, immune indexes, and the expression of heat shock protein to the normal level. This also provided data support for the medium-distance transportation of blunt snout bream in the future.



Similar content being viewed by others
Data availability
All data generated during this study are included in this published article.
Code availability
Not applicable.
References
Abre DEJS, Sanabria-Ochoa IA, Gonçalves FD, Urbinati EC (2008) Stress responses of juvenile matrinxa (Brycon amazonicus) after transport in a closed system under different loading densities. Cienc Rural 38(5):1413–1417. https://doi.org/10.1590/S0103-84782008000500034
Alcorn SW, Pascho RJ, Murray A, Shearer KD (2003) Effects of ration level on immune functions in chinook salmon (oncorhynchus tshawytscha). Aquaculture 217(1–4):529–545. https://doi.org/10.1016/S0044-8486(97)00264-0
Alexander C, Sahu N, Pal A, Akhtar M (2011) Haemato-immunological and stress responses of Labeo rohita (Hamilton) fingerlings: effect of rearing temperature and dietary gelatinized carbohydrate. J Anim Physiol an N 95(5):653–663. https://doi.org/10.1111/j.1439-0396.2010.01096.x
Asadi F, Asadian AH, Shahriari A, Pourkabir M (2009) Serum lipid, free fatty acid, and proteins in juvenile sturgeons: Acipenser persicus and Acipenser stellatus. Comp Clin Pathol 18(3):287–289. https://doi.org/10.1007/s00580-008-0797-0
Barcellos LJG, Woehl VM, Wassermann GF, Quevedo RM, Ittzés I, Krieger MH (2001) Plasma levels of cortisol and glucose in response to capture and tank transference in Rhamdia quelen (Quoy & Gaimard), a South American catfish. Aquac Res 32(2):121–123. https://doi.org/10.1046/j.1365-2109.2001.00539.x
Barton BA, Iwama GK (1991) Physiological changes in fish from stress in aquaculture with emphasis on the response and effects of corticosteroids. Annu Rev Fish Dis 1:3–26. https://doi.org/10.1016/0959-8030(91)90019-G
Bosworth BG, Small BC, Mischke C (2004) Effects of transport water temperature, aerator type, and oxygen level on channel catfish Ictalurus punctatus fillet quality. J World Aquacult Soc 35(3):412–419. https://doi.org/10.1111/j.1749-7345.2004.tb00105.x
Couto A, Enes P, Peres H, Oliva-Teles A (2008) Effect of water temperature and dietary starch on growth and metabolic utilization of diets in gilthead sea bream (Sparus aurata) juveniles. Comp Biochem Phys A 151(1):45–90. https://doi.org/10.1016/j.cbpa.2008.05.013
Cuesta A, Meseguer J, Esteban MA (2004) Total serum immunoglobulin M levels are affected by immunomodulators in seabream (Sparus aurata L). Vet Immunol Immunop 101(3–4):203–210
Cunha MAD, Barros FM, Garcia L, Veeck APL, Heinzmann BM, Loro VL, Emanuelli
Baldisserotto TB (2010) Essential oil of Lippia alba: a new anesthetic for silver catfish. Rhamdia Quelen Aquaculture 306(1–4):403–406. https://doi.org/10.1016/j.aquaculture.2010.06.014
Dang VT, Speck P, Benkendorff K (2012) Influence of elevated temperatures on the immune response of abalone. Haliotis Rubra Fish Shellfish Immun 32(5):732–740. https://doi.org/10.1016/j.fsi.2012.01.022
Delaney MA, Klesius PH (2004) Hypoxic conditions induce Hsp70 production in blood, brain and head kidney of juvenile Nile tilapia Oreochromis niloticus (L). Aquaculture 236(1–4):633–644. https://doi.org/10.1016/j.aquaculture.2004.02.025
Dhanasiri AK, Fernandes JM, Kiron V (2013a) Acclimation of zebrafish to transport stress. Zebrafish 10(1):87–98. https://doi.org/10.1089/zeb.2012.0843
Dhanasiri AK, Fernandes JM, Kiron V (2013b) Liver transcriptome changes in zebrafish during acclimation to transport-associated stress. PLoS One 8(6):1–12. https://doi.org/10.1371/journal.pone.0065028
Dobšikova R, Svobodova Z, Blahova J, Modra H, Velišek J (2009) The effect of transport on biochemical and haematological indices of common carp (Cyprinus carpio L). Czech J Anim Sci 54(11):510–518
Dominguez M, Takemura A, Tsuchiya M, Nakamura S (2004) Impact of different environmental factors on the circulating immunoglobulin levels in the Nile tilapia. Oreochromis Niloticus Aquac 241(1–4):491–500. https://doi.org/10.1016/j.aquaculture.2004.06.027
El-Boshy ME, El-Ashram AM, Abdelhamid FM, Gadalla HA (2010) Immunomodulatory effect of dietary Saccharomyces cerevisiae, beta-glucan and laminaran in mercuric chloride treated Nile tilapia (Oreochromis niloticus) and experimentally infected with Aeromonas hydrophila. Fish Shellfish Immun 28(5–6):802–808. https://doi.org/10.1016/j.fsi.2010.01.017
Ellis AE (1999) Immunity to bacteria in fish. Fish Shellfish Immun 9(4):291–308. https://doi.org/10.1006/fsim.1998.0192
Farcy E, Serpentini A, Fiévet B, Lebel J (2007) Identification of cDNAs encoding HSP70 and HSP90 in the abalone Haliotis tuberculata: transcriptional induction in response to thermal stress in hemocyte primary culture. Comp Biochem Phys B 7:540–550. https://doi.org/10.1016/j.cbpb.2006.12.006
Frei B (1999) Molecular and biological mechanisms of antioxidant action. Faseb J 13(9):963–964. https://doi.org/10.1096/fasebj.13.9.963
Gao Q, Zhao J, Song L, Qiu L, Yu Y, Zhang H, Ni D (2008) Molecular cloning, characterization and expression of heat shock protein 90 gene in the haemocytes of bay scallop Argopecten irradians. Fish Shellfish Immun 24(4):379–385. https://doi.org/10.1016/j.fsi.2007.08.008
Gomes LC, Araujo-Limrao CRM, Oubach D, Chippari-Gomeansd A, Lopes N, Urbinati EC (2003) Effect of fish density during transportation on stress and mortality of juvenile Tambaqui Colossoma macropomum. J World Aquacult Soc 34(1):76–84. https://doi.org/10.1111/j.1749-7345.2003.tb00041.x
Harmon TS (2009) Methods for reducing stressors and maintaining water quality associated with live fish transport in tanks: a review of the basics. Rev Aquac 1(1):58–66. https://doi.org/10.1111/j.1753-5131.2008.01003.x
Hasan M, Bart AN (2007) Effects of capture, loading density and transport stress on the mortality, physiological responses, bacterial density and growth of rohu Labeo rohita fingerlings. Fish Physiol Biochem 33(3):241–248. https://doi.org/10.1007/s10695-007-9136-7
Hohlenwerger JC, Copatti CE, Sena AC, Couto RD, Baldisserotto B, Heinzmann BM, Caron BO, Schmidt D (2016) Could the essential oil of Lippia alba provide a readily available and cost-effective anaesthetic for Nile tilapia (Oreochromis niloticus)? Mar Freshw Behav Physiol 49(2):119–126. https://doi.org/10.1080/10236244.2015.1123869
Hou YY, Han XD (2001) Effects of temperature and steroid hormones on immunoglobulin M (IgM) in immature rainbow trout, Oncorhynchus mykiss. J Nanjing Univ (Nat Sci) 5:563–568
Ivanina A, Taylor C, Sokolova I (2009) Effects of elevated temperature and cadmium exposure on stress protein response in eastern oysters Crassostrea virginica (Gmelin). Aquat Toxicol 91(3):245–254. https://doi.org/10.1016/j.aquatox.2008.11.016
Iversen M, Finstad B, Nilssen KJ (1998) Recovery from loading and transport stress in Atlantic salmon (Salmosalar L) smolts. Aquaculture 168(1–4):387–394. https://doi.org/10.1016/S0044-8486(98)00364-0
Iwama GK, Thomas PT, Forsyth RB, Vijayan MM (1998) Heat shock protein expression in fish. Rev Fish Biol Fisher 8(1):35–56. https://doi.org/10.1023/A:1008812500650
Iwama GK, Vijayan MM, Forsyth RB, Ackerman PA (1999) Heat shock proteins and physiological stress in fish. Am Zool 39(6):901–909. https://doi.org/10.1093/icb/39.6.901
King HR (2009) Fish transport in the aquaculture sector: an overview of the road transport of Atlantic salmon in Tasmania. J Vet Behav 4(4):163–168. https://doi.org/10.1016/j.jveb.2008.09.034
Kingsley GR (1939) The determination of serum total protein, albumin and globulin by the biuret reaction. J Biol Chem 131(1):197–200. https://doi.org/10.1016/S0021-9258(18)73494-7
Leadon DP (1994) Studies of the effects of transporting horses: better to arrive than to travel. Equine Vet J 26(5):346–347. https://doi.org/10.1111/j.2042-3306.1994.tb04400.x
Levin BA, Bolotovskiy AA (2015) Discovery of latitudinal gradient of triiodothyronine concentrations in ectotherms as revealed from a cyprinid fish, the common roach Rutilus rutilus. Biochem Syst Ecol 62:128–136. https://doi.org/10.1016/j.bse.2015.08.007
Li XF, Liu WB, Lu KL, Xu WN, Wang Y (2012) Dietary carbohydrate/lipid ratios affect stress, oxidative status and non-specific immune responses of fingerling blunt snout bream. Megalobrama Amblycephala Fish Shellfish Immun 33(2):316–323. https://doi.org/10.1016/j.fsi.2012.05.007
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the 2-ΔΔCt method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Ming JH, Xie J, Xu P, Ge XP, Liu WB, Ye JY (2012) Effects of emodin and vitamin C on growth performance, biochemical parameters and two HSP70s mRNA expression of Wuchang bream (Megalobrama amblycephala Yih) under high temperature stress. Fish Shellfish Immun 32(5):651–661. https://doi.org/10.1016/j.fsi.2012.01.008
Montero D, Tort L, Izquierdo L, Vergara JM (1998) Depletion of serum alternative complement pathway activity in gilthead seabream caused by a-tocopherol and n-3 HUFA dietary deficiencies. Fish Physiol Biochem 18:399–407. https://doi.org/10.1023/A:1007734720630
Obach A, Quentel C, Laurencin FB (1993) Effects of alpha-tocopherol and dietary oxidized fish oil on the immune response of sea bass Dicentrarchus labrax. Dis Aquat Organ 15(3):175–185. https://doi.org/10.3354/dao015175
Olsen SH, Tobiassen T, Akse L, Evensen TH, Midling KØ (2013) Capture induced stress and live storage of Atlantic cod (Gadus morhua) caught by trawl: consequences for the flesh quality. Fish Res 147:446–453. https://doi.org/10.1016/j.fishres.2013.03.009
Ortuño J, Cuesta A, Rodrıguez A, Esteban M, Meseguer J (2002) Oral administration of yeast, Saccharomyces cerevisiae, enhances the cellular innate immune response of gilthead seabream (Sparus aurata L). Vet Immunol Immunop 85(1–2):41–50. https://doi.org/10.1016/S0165-2427(01)00406-8
Pakhira C, Nagesh TS, Abraham TJ, Dash G, Behera S (2015) Stress responses in rohu, Labeo rohita transported at different densities. Aquac Rep 2(2):39–45. https://doi.org/10.1016/j.aqrep.2015.06.002
Reddy PS, Corley RB (1999) The contribution of ER quality control to the biologic functions of secretory IgM. Immunol Today 20(12):582–588. https://doi.org/10.1016/S0167-5699(99)01542-X
Refaey MM, Tian X, Tang R, Li D (2017) Changes in physiological responses, muscular composition and flesh quality of channel catfish Ictalurus punctatus suffering from transport stress. Aquaculture 478(1):9–15. https://doi.org/10.1016/j.aquaculture.2017.01.026
Ruane NM, Carballo EC, Komen J (2002) Increased stocking density influences the acute physiological stress response of common carp Cyprinus carpio (L). Aquac Res 33(10):777–784. https://doi.org/10.1046/j.1365-2109.2002.00717.x
Sakai DK (1992) Repertoire of complement in immunological defense mechanisms of fish. Annu Rev Fish Dis 2:223–247. https://doi.org/10.1016/0959-8030(92)90065-6
Sampaio FD, Freire CA (2016) An overview of stress physiology of fish transport changes in water quality as a function of transport duration. Fish Fish 17(4):1055–1072. https://doi.org/10.1111/faf.12158
Tafalla C, Figueras A, Nonoa B (1999) Role of nitric oxide on the replication of viral haemorthagic septicemia virus (VHSV). Vet Immunol Immunop 72(3–4):249–256. https://doi.org/10.1016/S0165-2427(99)00109-9
Tang HG, Wu TX, Zhao ZY, Pan XD (2008) Effects of fish protein hydrolysate on growth performance and humoral immune response in large yellow croaker (Pseudosciaena crocea R). J Zhejiang Univ Sci B 9(9):684–690. https://doi.org/10.1631/jzus.B0820088
Teixeira RR, Souza RC, Sena AC, Baldisserotto B, Heinzmann BM, Couto RD, Copatti CE (2016) Essential oil of Aloysia triphylla in Nile tilapia: anaesthesia, stress parameters and sensory evaluation of fillets. Aquac Res 48(7):3383–3392. https://doi.org/10.1111/are.13165
Todd KJ, Eales JG (2002) The effect of handling and blood removal on plasma levels and hepatic deiodination of thyroid hormones in adult male and female rainbow trout. Oncorhynchus Mykiss Can J Zool 80(2):372–375. https://doi.org/10.1139/z01-228
Tsan MF, Gao B (2004) Cytokine function of heat shock proteins. Am J Physiol-Cell Ph 286(4):C739–C744. https://doi.org/10.1152/ajpcell.00364.2003
Wallin RP, Lundqvist A, Moré SH, Von BA, Kiessling R, Ljunggren HG (2002) Heat-shock proteins as activators of the innate immune system. Trends Immunol 23(3):130–135. https://doi.org/10.1016/S1471-4906(01)02168-8
Wang X, Wang L, Yao C, Qiu L, Zhang H, Zhi Z, Song L (2012) Alternation of immune parameters and cellular energy allocation of Chlamys farreri under ammonia-N exposure and Vibrio anguillarum challenge. Fish Shellfish Immun 32(5):741–749. https://doi.org/10.1016/j.fsi.2012.01.025
Wang BK, Liu WB, Xu C, Cao XF, Zhong XQ, Shi HJ, Li XF (2017) Dietary carbohydrate levels and lipid sources modulate the growth performance, fatty acid profiles and intermediary metabolism of blunt snout bream Megalobrama amblycephala in an interactive pattern. Aquaculture 481:140–153. https://doi.org/10.1016/j.aquaculture.2017.08.034
Weigl E, Kopecek P, Raska M, Hradilová S (1999) Heat shock proteins in immune reactions. Folia Microbiol 44(5):561–566. https://doi.org/10.1007/BF02816261
Winberg S, Lepage O (1998) Elevation of brain 5-HT activity, POMC expression, and plasma cortisol in socially subordinate rainbow trout. Am J Physiol-Re I 274(3):R645-654. https://doi.org/10.1152/ajpregu.1998.274.3.R645
Wu CX, Zhao FY, Zhang Y, Zhu YJ, Ma MS, Mao HL, Hu CY (2012) Overexpression of Hsp90 from grass carp (Ctenopharyngodon idella) increases thermal protection against heat stress. Fish Shellfish Immun 33(1):42–47. https://doi.org/10.1016/j.fsi.2012.03.033
Zhang J, Shen H, Wang X, Wu J, Xue Y (2004) Effects of chronic exposure of 2, 4-dichlorophenol on the antioxidant system in liver of freshwater fish Carassius auratus. Chemosphere 55(2):167–174. https://doi.org/10.1016/j.chemosphere.2003.10.048
Funding
This work was supported by National Modern Agriculture Industry Technology System (CARS-45–45), National Freshwater Aquatic Germplasm Resource Bank (2021DKA30470), Basic Research Business Fund Project of Henan Province (JBKY2021), and Modern Agricultural Industry Technology System of Henan Province (S2014-10).
Author information
Authors and Affiliations
Contributions
Funding acquisition and project administration: Qin Zhang; Writing-original draft preparation data curation and data validation: Bingke Wang; Methodology and writing—reviewing and editing: Yanhui Wang and Tao Jia; Resources: Jianxin Feng and Changyi Qu; Software: Xiaojun Wu and Xingli Yang. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All animal handling procedures were performed in strict accordance with the Guide for the Care and Use of Laboratory Animals Central of Henan Academy of Fishery Sciences, and the protocol was approved by the Institutional Animal Care and Use Committee of Henan Academy of Fishery Sciences.
Consent to participate
The authors agree to collaborate and publish this article.
Consent for publication
We will transfer the copyright of the article to editorial office for publishing.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, B., Wang, Y., Jia, T. et al. Changes in physiological responses and immunity of blunt snout bream Megalobrama amblycephala from transport stress. Fish Physiol Biochem 48, 1183–1192 (2022). https://doi.org/10.1007/s10695-022-01108-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01108-x