Abstract
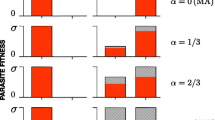
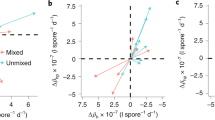
Evolutionary studies often estimate fitness components with the aim to make predictions about the outcome of selection. Depending on the system and the question, different fitness components are used, but their usefulness for predicting the outcome of selection is rarely tested. Here we estimate host fitness components in different ways with the aim to test how well they agree with each other and how well they predict host fitness at the population level in the presence of the parasite. We use a Daphnia magna-microparasite system to study the competitive ability of host clones in the absence and presence of the parasite, the infection intensity of the parasite in individuals of twelve host clones (an estimate of both host resistance and parasite reproductive success), and parasite persistence in small host populations (an estimate of R 0 of the parasite). Analysis of host competitive ability and parasite persistence reveals strong host genotype effects, while none are found for infection intensity. Host competitive ability further shows a genotype-specific change upon infection, which is correlated with the relative persistence of the parasite in the competing hosts. Hosts in which the parasite persists better suffer a competitive disadvantage in the parasite’s presence. This suggests that in this system, parasite-mediated selection can be predicted by parasite persistence, but not by parasite infection intensity.




Similar content being viewed by others
References
Anderson RM, May RM (1981) The population dynamics of microparasites and their invertebrate hosts. Phil Trans R Soc B 291:451–524
Anderson RM, May RM (1982) Coevolution of hosts and parasites. Parasitology 85:411–426
Bull JJ (1994) Virulence. Evolution 48:1423–1437
Capaul M, Ebert D (2003) Parasite-mediated selection in experimental Daphnia magna populations. Evolution 57:249–260
Carius HJ, Little TJ, Ebert D (2001) Genetic variation in a host-parasite association: potential for coevolution and frequency-dependent selection. Evolution 55:1136–1145
Clarke BC (1979) Evolution of genetic diversity. Proc R Soc B 205:453–474
de Roode JC, Yates AJ, Altizer S (2008) Virulence-transmission trade-offs and population divergence in virulence in a naturally occurring butterfly parasite. Proc Natl Acad Sci USA 105:7489–7494
Decaestecker E, Vergote A, Ebert D, Meester LD (2003) Evidence for strong host clone-parasite species interactions in the Daphnia microparasite system. Evolution 57:784–792
Decaestecker E, Declerck S, Meester LD, Ebert D (2005) Ecological implications of parasites in natural Daphnia populations. Oecologia 144:382–390
Dieckmann U (ed) (2002) Adaptive dynamics of infectious disease. Cambridge University Press, Cambridge
Duffy MA (2007) Selective predation, parasitism, and trophic cascades in a bluegill–Daphnia–parasite system. Oecologia 153:453–460
Duffy MA, Sivars-Becker L (2007) Rapid evolution and ecological host-parasite dynamics. Ecol Lett 10:44–53
Duncan AB, Mitchell SE, Little TJ (2006) Parasite-mediated selection and the role of sex and diapause in Daphnia. J Evol Biol 19:1183–1189
Ebert D (2005) Ecology, epidemiology, and evolution of parasitism in Daphnia [Internet]. National Library of Medicine (US), National Center for Biotechnology Information, Bethesda, MD. Available from http://www.ncbi.nlm.nih.gov/books/NBK2036. (Accessed Feb 2011)
Ebert D (2008) Host-parasite coevolution: insights from the Daphnia-parasite model system. Curr Opin Microbiol 11:290–301
Ebert D, Bull JJ (2003) Challenging the trade-off model for the evolution of virulence: is virulence management feasible? Trends Microbiol 11:15–20
Ebert D, Zschokke-Rohringer CD, Carius HJ (1998) Within- and between-population variation for resistance of Daphnia magna to the bacterial endoparasite Pasteuria ramosa. Proc R Soc B 265:2127–2134
Ebert D, Lipsitch M, Mangin KL (2000) The effect of parasites on host population density and extinction: experimental epidemiology with Daphnia and six microparasites. Am Nat 156:459–477
Ebert D, Haag C, Kirkpatrick M, Riek M, Hottinger J, Pajunen VI (2002) A selective advantage to immigrant genes in a Daphnia metapopulation. Science 295:485–488
Fels D (2005) The effect of food on microparasite transmission in the waterflea Daphnia magna. Oikos 109:360–366
Fels D, Lee VA, Ebert D (2004) The impact of microparasites on the vertical distribution of Daphnia magna. Arch Hydrobiol 161:65–80
Frank SA (1996) Models of parasite virulence. Quart Rev Biol 71:37–78
Grech K, Watt K, Read AF (2006) Host–parasite interactions for virulence and resistance in a malaria model system. J Evol Biol 19:1620–1630
Green J (1974) Parasites and epibionts of Cladocera. Trans Zool Soc Lond 32:417–515
Haag CR, Ebert D (2004) Parasite-mediated selection in experimental metapopulations of Daphnia magna. Proc R Soc B 271:2149–2155
Haag CR, Sakwinska O, Ebert D (2003) Test of synergistic interaction between infection and inbreeding in Daphnia magna. Evolution 57:777–783
Hamilton WD (1980) Sex versus non-sex versus parasite. Oikos 35:282–290
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387
Hartl DL (1987) A primer of population genetics. Sinauer, Sunderland
Hochberg ME (1998) Establishing genetic correlations involving parasite virulence. Evolution 52:1865–1868
Jensen KH, Little TJ, Skorping A, Ebert D (2006) Empirical support for optimal virulence in a castrating parasite. PLoS Biol 4:e197
Jungen H, Hartl DL (1979) Average fitness of populations of Drospohila melanogaster as estimated using compound-autosome strains. Evolution 33:359–370
Larsson JIR, Ebert D, Vávra J, Voronin VN (1996) Redescription of Pleistophora intestinalis Chatton, 1907, a microsporidian parasite of Daphnia magna and Daphnia pulex, with establisment of the new genus Glugoides (Microspora, Glugeidae). Europ J Protistol 32:251–261
Little TJ, Carius HJ, Sakwinska O, Ebert D (2002) Competitiveness and life-history characteristics of Daphnia with respect to susceptibility to a bacterial pathogen. J Evol Biol 15:796–802
Mackinnon MJ, Read AF (1999) Genetic relationships between parasite virulence and transmission in the rodent malaria Plasmodium chabaudi. Evolution 53:689–703
May RM, Anderson RM (1983) Epidemiology and genetics in the coevolution of parasites and hosts. Proc R Soc B 219:281–313
Mitchell SE, Read AF, Little TJ (2004) The effect of a pathogen epidemic on the genetic structure and reproductive strategy of the crustacean Daphnia magna. Ecol Lett 7:848–858
Packer C, Holt RD, Hudson PJ, Lafferty KD, Dobson AP (2003) Keeping the herds healthy and alert: Implications of predator control for infectious disease. Ecol Lett 6:797–802
Refardt D, Ebert D (2006) Quantitative PCR to detect, discriminate and quantify intracellular parasites in their host: an example from three microsporidians in Daphnia. Parasitology 133:11–18
Refardt D, Ebert D (2007) Inference of parasite local adaptation using two different fitness components. J Evol Biol 20:921–929
Refardt D, Canning EU, Mathis A, Cheney SA, Lafranchi-Tristem NJ, Ebert D (2002) Small subunit ribosomal DNA phylogeny of microsporidia that infect Daphnia (Crustacea: Cladocera). Parasitology 124:381–389
Rigby MC, Hechinger RF, Stevens L (2002) Why should parasite resistance be costly? Trends Parasitol 18:116–120
Rolff J, Joop G (2002) Estimating condition: pitfalls of using weight as a fitness correlate. Evol Ecol Res 4:931–935
Roy BA, Kirchner JW (2000) Evolutionary dynamics of pathogen resistance and tolerance. Evolution 54:51–63
SAS Institute Inc (1989) JMP, Version 8. SAS Institute Inc., Cary
Schmid-Hempel P, Ebert D (2003) On the evolutionary ecology of specific immune defence. Trends Ecol Evol 18:27–32
Schwarzenbach GA, Ward PI (2006) Responses to selection on phenoloxidase activity in yellow dung flies. Evolution 60:1612–1621
Stearns SC (1992) The evolution of life histories. Oxford University Press, Oxford
Stirnadel HA, Ebert D (1997) Prevalence, host specificity and impact on host fecundity of microparasites and epibionts in three sympatric Daphnia species. J Anim Ecol 66:212–222
Thompson JN (1994) The coevolutionary process. University of Chicago Press, Chicago
Tompkins DM, Begon M (1999) Parasites can regulate wildlife populations. Parasitol Today 15:311–313
Vijendravarma RK, Kraaijeveld AR, Godfray HCJ (2009) Experimental evolution shows Drosophila melanogaster resistance to a microsporidian pathogen has fitness costs. Evolution 63:104–114
Acknowledgments
We thank N. Basieux, J. Hottinger, S. Lass and L. Sygnarski for technical assistance, M. Kölliker for statistical advice, and H. K. Alexander for polishing the language. The manuscript benefitted from comments by J. Bull, J. Jokela, C. Kost, S. Lass, M. Zbinden, two anonymous reviewers, and the handling editor. The authors were supported by the Swiss National Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Refardt, D., Ebert, D. The impact of infection on host competition and its relationship to parasite persistence in a Daphnia microparasite system. Evol Ecol 26, 95–107 (2012). https://doi.org/10.1007/s10682-011-9487-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-011-9487-5