Abstract
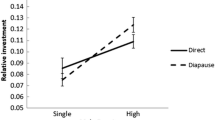
Differences in reproductive success (RS) between different groups of individuals are of interest to researchers studying natural and sexual selection. Since it is often not feasible to quantify RS in the wild, researchers make use of proxies instead. One such proxy is clutch size. However, research on species providing parental care (mainly birds and mammals) has learned that a large clutch size does not guarantee a large number of offspring. In contrast, much less is known on the link between clutch size and RS for species lacking parental care, such as many reptiles and insects. Here, we ask whether clutch size provides a satisfactory estimate of RS for a polymorphic insect. Our study species is a damselfly showing two distinct female morphs for which RS (estimated by clutch size) has been studied to evaluate the evolutionary role of sexual conflict. However, in this system not only among family variation in offspring viability, but also differences between female morphs, may affect how clutch size relates to offspring number and quality. To evaluate the use of clutch size as estimate of RS, we examined how clutch size correlated with subsequent success measures of developing offspring by rearing damselfly from eggs to adults under two laboratory food treatments. In both treatments, we detected that clutch size correlated well with offspring number early in larval life, but that this relation is reduced by among family variation in survival in later developmental stages. Clutch size was moderately correlated with the number of offspring that successfully metamorphosed to winged adults. Patterns did not differ between female morphs and the nature of the correlation could not be explained from offspring quantity-quality trade-offs.



Similar content being viewed by others
References
Abbott J, Svensson EI (2005) Phenotypic and genetic variation in emergence and developmental time of a trimorphic damselfly. J Evol Biol 18:1464–1470
Arnqvist G, Rowe L (2005) Sexual conflict. Princeton University Press, Princeton
Askew RR (2004) The dragonflies of Europe, 2nd edn. Harley Books, Essex
Banks MJ, Thompson DJ (1987) Lifetime reproductive success of females of the damselfly Coenagrion puella. J Anim Ecol 56:815–832
Chowdhury SH, Corbet PS (1989) Feeding-related behaviour in larvae of Enallagma cyathigerum (Charpentier) (Zygoptera: Coenagrionidae). Odonatologica 18:285–288
Clutton-Brock TH (1988) Reproductive success: studies of individual variation in contrasting breeding systems. University of Chicago Press, Chicago
Cooper G, Holland PWH, Miller PL (1996) Captive breeding of Ischnura elegans (Vanderlinden): observations on longevity, copulation and oviposition (Zygoptera: Coenagrionidae). Odonatologica 25:261–273
Corbet PS (1999) Dragonflies: behaviour and ecology of odonata. Harley Books, Essex
De Block M, Stoks R (2004) Cannibalism-mediated life history plasticity to combined time and food stress. Oikos 106:587–597
De Block M, Stoks R (2005) Fitness effects from egg to reproduction: bridging the life history transition. Ecology 86:185–197
De Block M, McPeeck MA, Stoks R (2008) Life-history evolution when Lestes damselflies invaded vernal ponds. Evolution 62:485–493
Desouhant E, Debouzie D, Ploye H et al (2000) Clutch size manipulations in the chestnut weevil, Curculio elephas: fitness of oviposition strategies. Oecologia 122:493–499
Dijkstra KDW, Lewington R (2006) Field guide to the dragonflies and damselflies of Britain and Europe. British Wildlife Publishing, UK
Dijkstra C, Bult A, Bijlsma S et al (1990) Brood size manipulations in the kestrel (Falco tinnunculus): effects on offspring and parent survival. J Anim Ecol 59:269–285
Doerksen GP (1980) Notes on the reproductive behaviour of Enallagma cyathigerum (Charpentier) (Zygoptera: Coenagrionidae). Odonatologica 9:293–296
Fincke OM (1984) Sperm competition in the damselfly Enallagma hageni Walsh (Odonata: Coenagrionidae): benefits of multiple mating to males and females. Behav Ecol Sociobiol 14:235–240
Fincke OM, Hadrys H (2001) Unpredictable offspring survivorship in the damselfly, Megaloprepus coerulatus, shapes parental behaviour, constrains sexual selection, and challenges traditional fitness estimates. Evolution 55:762–772
Fox CW, Martin JD, Thakar MS (1996) Clutch size manipulations in two seed beetles: consequences for progeny fitness. Oecologia 108:88–94
Gavrilets S, Waxman D (2002) Sympatric speciation by sexual conflict. Proc Natl Acad Sci USA 99:10533–10538
Gillespie DOS, Russell AF, Lummaa V (2008) When fecundity does not equal fitness: evidence of an offspring quantity versus quality trade-off in pre-industrial humans. Proc R Soc B Biol Sci 275:713–722
Godfray HCJ (1987) The evolution of clutch size in invertebrates. In: Harvey PH, Partridge L (eds) Oxford surveys in evolutionary biology, vol 4. Oxford University Press, Oxford, pp 117–154
Godfray HCJ, Partridge L, Harvey PH (1991) Clutch size. Ann Rev Ecol Syst 22:409–429
Gosden TP, Svensson EI (2007) Female sexual polymorphism and fecundity consequences of male mating harassment in the wild. PLoS ONE 2:e580. doi:10.1371/journal.pone.0000580
Härdling R, Bergsten J (2006) Nonrandom mating preserves intrasexual polymorphism and stops population differentiation in sexual conflict. Am Nat 167:401–409
Hardy ICW, Griffiths NT, Godfray HCJ (1992) Clutch size in a parasitoids wasp: a manipulation experiment. J Anim Ecol 61:121–129
Hayashi TI, Vose M, Gavrilets S (2007) Genetic differentiation by sexual conflict. Evolution 61:516–529
Koskela E (1998) Offspring growth, survival and reproductive success in the bank vole: a litter size manipulation experiment. Oecologia 115:379–384
Lopez-Vaamonde C, Raine NE, Koning JW, Brown RM, Pereboom JJM, Ings TC, Ramos-Rodriguez O, Jordan WC, Bourke AFG (2009) Lifetime reproductive success and longevity of quens in an annual social insect. J Evol Biol 22:983–996
Madsen T, Shine R (1998) Quantity or quality? Determinants of maternal reproductive success in tropical pythons (Liasis fuscus). Proc R Soc B Biol Sci 265:1521–1525
Plaistow S, Siva-Jothy MT (1999) The ontogenetic switch between odonate life history stages: effects of fitness when time and food are limited. Anim Behav 58:659–667
Roff DA (1992) The evolution of life histories: theory and analysis. Chapman and Hall, New York
Sinervo B, Svensson E, Comendant T (2000) Density cycles and an offspring quantity and quality game driven by natural selection. Nature 406:985–988
Sirot LK, Brockmann HJ (2001) Costs of sexual interactions to females in Rambur’s forktail damselfly, Ischnura ramburi (Zygoptera: Coenagrionidae). Anim Behav 61:415–424
Slos S, Stoks R (2008) Predation risk induces stress proteins and reduces antioxidant defense. Funct Ecol 22:637–642
Stoks R (2001) Food stress and predator-induced stress shape developmental performance in a damselfly. Oecologia 127:222–229
Stoks R, De Block M, McPeeck MA (2005) Alternative growth and energy storage responses to mortality threats in damselflies. Ecol Lett 8:1307–1316
Svensson EI, Abbott J (2005) Evolutionary dynamics and population biology of a polymorphic insect. J Evol Biol 18:1503–1514
Svensson EI, Abbott J, Härdling R (2005) Female polymorphism, frequency dependence, and rapid evolutionary dynamics in natural populations. Am Nat 165:567–576
Svensson EI, Abbott JK, Gosden TP et al (2009) Female polymorphisms, sexual conflict and limits to speciation processes in animals. Evol Ecol 23:93–108
Van Dongen S (2006) Bayesian joint model estimation of binary outcome and time-to-event data: effects of leaf quality on pupal survival and time-to-emergence in the winter moth. - Environ Ecol Stat 13:213–228
Van Gossum H, Sanchez R, Cordero A (2003) Observations on rearing damselflies under laboratory conditions. Anim Biol 53:37–45
Van Gossum H, Sherratt TN, Cordero A (2008) The evolution of sex-limited colour polymorphisms. In: Córdoba-Aguilar A (ed) Dragonflies and Damselflies - Model Organisms for Ecological and Evolutionary Research. Oxford University Press, Oxford, pp 219–229
Van Noordwijk AJ, de Jong G (1986) Acquisition and allocation of resources—their influence on variation in lifehistory tactics. Am Nat 128:137–142
Verbeke G, Molenberghs G (1997) Linear Mixed Models. A SAS-oriented approach, Springer, New York
Zaviezo T, Mills N (2000) Factors influencing the evolution of clutch size in a gregarious parasitoid insect. J Anim Ecol 69:1047–1057
Acknowledgments
We are grateful to Tom Snijkers, Bert Van Den Branden, Tine Van Duffel, Ben Huysmans and Bie Van Linden for their help with rearing damselfly larvae. Robby Stoks and Marjan De Block kindly advised on rearing methods. The “Institute for the promotion of Innovation through Science and Technology in Flanders” (IWT-Flanders) supported this work (research grant to J.B.). H.V.G. is a postdoctoral fellow with the Fund for Scientific Research-Flanders (FWO-Flanders).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bots, J., Van Dongen, S., De Bruyn, L. et al. Clutch size and reproductive success in a female polymorphic insect. Evol Ecol 24, 1239–1253 (2010). https://doi.org/10.1007/s10682-010-9362-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-010-9362-9