Abstract
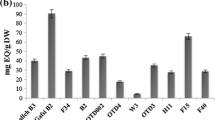
Genetic improvement of sweet potato [Ipomoea batatas (L.) Lam.] is based on recurrent cycles of phenotypic selection and recombination of parents. Metabolite profiling can be used to differentiate chemotypes to assist selection of parents. Chlorogenic acids (CQAs) and coumarins are natural fungicides and insect repellents present in the storage root periderm which can contribute to reduction of post-harvest losses and insect damage. The objectives of this study were: (i) to develop a rapid and reliable analytical technique for screening breeding lines, (ii) to correlate their CQAs and coumarins contents with periderm colour and biotic stress susceptibility, and (iii) to assess variation among clones. A first experiment measured the variation in scopolin, scopoletin, chlorogenic acid (CGA) and three dicaffeoylquinic acids (3,4-, 4,5-, 3,5-diCQA) in 296 breeding lines periderm. CGA (mean of 0.95 µg/mg FW) and 3,5-diCQA (0.86 µg/mg) contents were significantly higher than scopolin (0.50 µg/mg), scopoletin (0.09 µg/mg), 3,4-diCQA (0.09 µg/mg), and 4,5-diCQA (0.16 µg/mg). CGA was significantly and positively correlated with all compounds but especially with 3,5-diCQA (+ 0.812**). All compounds were in significantly higher content in purple-red periderm breeding lines. A second experiment estimated the range of variation among five clones for 39 selected breeding lines. The results confirmed the first experiment. Furthermore, for all six compounds the coefficient of variation in the second experiment were low indicating that they are genetically controlled. CQAs appear to be more interesting compounds compared to coumarins for selecting breeding lines and chemotype assisted breeding should be a useful tool to reduce post-harvest losses and insect damage.





Similar content being viewed by others
References
Alam A, Subhan N, Hossain H, Hossain M, Reza HM, Rahman M, Ullah MO (2016) Hydroxycinnamic acid derivatives: a potential class of natural compounds for the management of lipid metabolism and obesity. Nut and Metab 13:27. https://doi.org/10.1186/s12986-016-0080-3
Al-Rifai A, Aqel A, Saleem MA (2020) Development and validation of a rapid and efficient method for simultaneous determination of scopolin and scopoletin in convolvulus species by ultra-high-performance liquid chromatography-tandem mass spectrometry. Cur Pharm Anal 16(5):494–503. https://doi.org/10.2174/1573412915666190206122916
Anyanga MO, Muyinza H, Talwana H, Hall DR, Farman DI, Ssemakula GN, Mwanga ROM, Stevenson PC (2013) Resistance to the weevils Cylas puncticollis and Cylas brunneus conferred by sweetpotato root surface compounds. J Agric Food Chem 61:8141–8147. https://doi.org/10.1021/jf4024992
Anyanga MO, Yada B, Yencho GC, Ssemakula GN, Alajo A, Farman DI, Mwanga ROM, Stevenson PC (2017) Segregation of hydroxycinnamic acid esters mediating sweetpotato weevil resistance in storage roots of sweetpotato. Frontiers Plant Sci. https://doi.org/10.3389/fpls.2017.01011
AOAC (2002) Guidelines for single laboratory validation of chemicals methods for dietary supplements and botanicals. http://www.aoac.org/imis15_prod/AOAC_Docs/StandardsDevelopment/SLV_Guidelines_Dietary_Supplements.pdf. Accessed March 2020
Beninger CW, Abou-Zaid MM, Kistner ALE, Hallett RH, Iqbal MJ, Grodzinski B, Hall C (2004) A flavanone and two phenolic acids from Chrysanthemum morifoliumwith phytotoxic and insect growth regulating activity. J Chem Ecol 30(3):589–606. https://doi.org/10.1023/b:joec.0000018631.67394.e5
Beyer SF, Beesley A, Rohmann PFW, Schultheiss H, Conrath U, Langenbach CJG (2019) The Arabidopsis non-host defence-associated coumarin scopoletin protects soybean from Asian soybean rust. Plant J 99:397–413. https://doi.org/10.1111/tpj.14426
La Bonte DR, Villordon AQ, Clark CA, Wilson PW, Stoddard CS (2008) Murasaki-29. Sweetpotato HortSci 43(6):1895–1896. https://doi.org/10.1111/plb.12947
CABI (2020) Cylas formicarius, sweet potato weevil. Invasive Species Compendium https://www.cabi.org/isc/datasheet/17408
Chen J, Stout MJ, Beuzelin J, Smith TP, LaBonte D, Murray JM, Davis JA (2019) Host preference of sweetpotato weevil, Cylas formicarius elegantulus (Summers): an example of Hopkins’ host-selection principle. Arthro-Plant Inter 13:779–786. https://doi.org/10.1007/s11829-019-09704-0
CIP-AVRDC-IBPGR (1991) Descriptors for Sweet Potato. Huamán Z., editor. CIP, AVRDC, IBPGR International Board for Plant Genetic Resources (IPGRI), Rome, Italy. 134p
Clark CA, Ferrin DM, Smith TP, Holmes GJ (eds) (2013) Compendium of sweetpotato diseases, pests, and disorders, 2nd edn. APS Press, The American Phytopathological Society. St. Paul, MO, US, p 160
Clifford MN, Jaganath IB, Ludwig IA, Crozier A (2017) Chlorogenic acids and the acyl-quinic acids: discovery, biosynthesis, bioavailability and bioactivity. Nat Prod Rep Review 34:1391–1421. https://doi.org/10.1039/c7np00030h
Data ES, Nottingham SF, Kays SJ (1996) Effect of Sweetpotato latex on sweetpotato weevil feeding and oviposition. J Eco Entom 89:544–549. https://doi.org/10.1093/jee/89.2.544
De Oliveira PES, Conserva LM, Brito AC, Lemos RPL (2005) Coumarin derivatives from Esenbeckia grandiflora and its larvicidal activity against Aedes aegypti. Pharm Biol 43(1):53–57. https://doi.org/10.1080/13880200590903363
Döll S, Kuhlmann M, Rutten T, Mette MF, Scharfenberg S, Petridis A, Berreth DC, Mock HP (2018) Accumulation of the coumarin scopolin under abiotic stress conditions is mediated by the Arabidopsis thaliana THO/TREX complex. Plant J 93:431–444. https://doi.org/10.1111/tpj.13797
Drapal M, Rossel G, Heider B, Fraser PD (2019) Metabolic diversity in sweet potato (Ipomoea batatas, Lam.) leaves and storage roots. Hort Res 6:2. https://doi.org/10.1038/s41438-018-0075-5
FAOSTAT (2020) www.fao.org. Crop production statistics. Accessed Oct. 16th 2020.
Gnonlonfin GJB, Sanni A, Brimer L (2012) Review Scopoletin – a coumarin phytoalexin with medicinal properties. Crit Rev Plant Sci 31(1):47–56. https://doi.org/10.1080/07352689.2011.616039
Harrison HF, Michell TR, Peterson JK, Wechter WP, Majetich GF, Snook ME (2008) Contents of caffeoylquinic acid compounds in the storage roots of sixteen sweetpotato genotypes and their potential biological activity. J Amer Soc Hort Sci. 133(4): 492–500. https://doi.org/10.21273/JASHS.133.4.492
Jackson D, Harrison H, Ryan-Bohac J (2012) Insect resistance in sweetpotato plant introduction accessions. J Econ Entom 105:651–658. https://doi.org/10.1603/EC11307
Kundu A, Vadassery J (2019) Chlorogenic acid-mediated chemical defence of plants against insect herbivores. Plant Bio 21:185–189. https://doi.org/10.1111/plb.12947
Lebot V (1986) Evaluation of local and introduced cultivars of sweet potato (Ipomoea batatas (L.) Lam.) in Vanuatu. J South Pac Agric 11(3):25–31
Lebot V, Michalet S, Legendre L (2016) Identification and quantification of phenolic compounds responsible for the antioxidant activity of sweet potatoes with different flesh colours using high performance thin layer chromatography (HPTLC). J Food Comp Anal 49:94–101. https://doi.org/10.1016/j.jfca.2016.04.009
Lee HU, Chung MN, Lee JS, Ahn SH, Kim JM, Han SK, Yang JW, Song YS, Shin HM, Moon JY, Choi KH, Lim SH, Choi IH (2018) A Sweetpotato variety resistant to fusarium wilt, ‘Yeseumi.’ Kor J Breed Sci 50(2):131–138. https://doi.org/10.9787/KJBS.2018,50(2),pp.131
Leiss KA, Maltese F, Choi YH, Verpoorte R, Klinkhamer PGL (2009) Identification of chlorogenic acid as a resistance factor for thrips in chrysanthemum. Plant Physio 150:1567–1575. https://doi.org/10.1104/pp.109.138131
Li J, Wu J (2016) Scopolin. a glycoside form of the phytoalexin scopoletin. is likely involved in the resistance of Nicotania attenuata against Alternaria alternata. J Plant Patho. 98 (3):641–644. https://www.jstor.org/stable/44280514
Martínez G, Santiago MR, Del Rio JM, Laurade la Canal MP (2017) Chlorogenic acid is a fungicide active against phytopathogenic fungi. Pest Biochem Physio 140:30–35. https://doi.org/10.1016/j.pestbp.2017.05.012
Mwanga ROM, Niringiye C, Alajo A, Kigozi B, Namukula J, Mpembe I, Tumwegamire S, Gibson RW, Yencho GC (2011) ‘NASPOT 11’, a Sweetpotato Cultivar Bred by a Participatory Plant-breeding Approach in Uganda. HortSci 46(2):317–321. https://doi.org/https://doi.org/10.21273/HORTSCI.46.2.317
Mwanga ROM, Andrade MI, Carrey EE, Low JW, Yencho GC, Grüneberg WJ (2017) Sweetpotato (Ipomoea batatas L.). In: Campos. H. and Caligari. P.D.S. (eds) Genetic Improvement of Tropical Crops. Springer International Publishing. https://doi.org/10.1007/978-3-319-59819-2_6
Nelson S (2008) Java Black Rot of Okinawan Sweetpotato. Cooperative Extension Service, University of Hawaii, UH-CTAHR, Plant Dis PD 55:1–6
Nelson S (2009) Rhizopus Soft Rot of Sweetpotato. Cooperative Extension Service, University of Hawaii, UH-CTAHR, Plant Dis PD 68:1–6
Nielsen LW, Yen DE (1966) Resistance in sweetpotato to the scurf and black rot pathogens. NZ J Agric Res 9(4):1032–1041. https://doi.org/10.1080/00288233.1966.10429363
Nottingham SF, Son KC, Wilson DD, Severson RF, Kays SJ (1989) Feeding ovoposition preferences of sweet potato weevil Cylas formicarius elegantulus (Summers) on storage roots of sweet potato cultivars with differing surface chemistries. J Chem Ecol 15(3):895–903. https://doi.org/10.1007/BF01015185
Orsák M, Hamouz K, Lachman J, Kasal P (2019) Chlorogenic acid content in potato tubers with colored flesh as affected by a genotype location and longterm storage. Plant Soil Environ. 65: 355–360. https://doi.org/10.17221/195/2019-PSE
Padda MS, Picha DH (2008) Quantification of phenolic acids and antioxidant activity in sweetpotato genotypes. Sci Hort 119:17–20. https://doi.org/10.1016/j.scienta.2008.07.008
Peterson JK, Harrison HF, Jackson MD, Snook ME (2003) Biological activities and contents of scopolin and scopoletin in sweetpotato clones. HortSci. 38(6) 1129–1133. https://doi.org/10.21273/HORTSCI.38.6.1129
Peterson JK, Harrison HF, Snook ME, Jackson DM (2005) Chlorogenic acid content in sweetpotato germplasm: possible roles in disease and pest resistance. Allelopathy J 16:239–250
Prats E, Bazzalo ME, León A, Jorrin JV (2006) Fungitoxic effect of scopolin and related coumarins on Sclerotinia sclerotiorum. A way to overcome sunflower head rot. Euphytica 147:451–460. https://doi.org/10.1007/s10681-005-9045-8
Sharma A, Shahzad B, Rehman A, Bhardwaj R, Landi M, Zheng B (2019) Review Response of Phenylpropanoid Pathway and the Role of Polyphenols in Plants under Abiotic Stress. Molecules 24:2452. https://doi.org/10.3390/molecules24132452
Shimizu BI, Miyagawa H, Ueno T, Sakata K, Watanabe K, Ogawa K (2005) Morning Glory Systemically Accumulates Scopoletin and Scopolin after Interaction with Fusarium oxysporum. Verlag der Zeitschrift für Naturforschung 60:83–90. https://doi.org/10.1515/znc-2005-1-216
Siwinska J, Kadzinski L, Banasiuk R, Gwizdek-Wisniewska A, Olry A, Banecki B, Lojkowska E, Ihnatowicz A (2014) Identification of QTLs affecting scopolin and scopoletin biosynthesis in Arabidopsis thaliana. BMC Plant Bio 14:280. https://doi.org/10.1186/s12870-014-0280-9
Snook MEE, Data ES, Kays SJ (1994) Characterization and quantification of hexadecyl. octadecyl and eicosyl esters of P-coumaric acid in the vine and root latex of sweetpotato (Ipomoea batatas (L.) Lam.). J Agric Food Chem 42:2589–2595. https://doi.org/10.1021/jf00047a041
Son KC, Severson RF, Arrendale RF, Kays SJ (1990a) Isolation and characterization of pentacyclic triterpene ovipositional stimulant for the sweet potato weevil from Ipomoea batatas (L.) Lam. J Agric Food Chem 38:134–137. https://doi.org/10.1021/jf00091a028
Son KC, Severson RF, Arrendale RF, Kays SJ (1990) Surface chemical differences between sweet potato lines with varying levels of resistance to the sweetpotato weevil. J Amer Soc Hort Sci 115(4):696–699. https://doi.org/10.21273/JASHS.115.4.696
Soni B, Tseng TMP, Yue ZM (2019) Identification and Quantification of Allelochemicals from Selected Sweet Potato (Ipomoea batatas (L.) Lam.) Cultivars. Amer J Plant Sci 10:2354–2365. https://doi.org/10.4236/ajps.2019.1012163
Stange RR, Midland SL, Holmes JJ, Sims JJ, Mayer RT (2001) Constituents from the periderm and outer cortex of Ipomoea batatas with antifungal activity against Rhizopus stolonifera. Post Harv Bio Techn 23:85–92. https://doi.org/10.1016/S0925-5214(01)00105-3
Stevenson PC, Muyinza H, Hall DR, Porter EA, Farman DI, Talwana H, Mwanga ROM (2009) Chemical basis for resistance in sweetpotato Ipomoea batatas to the sweetpotato weevil Cylas puncticollis. Pure Appl Chem 81(1):141–151. https://doi.org/10.1351/PAC-CON-08-02-10
Sung WS, Lee DG (2010) Antifungal action of chlorogenic acid against pathogenic fungi mediated by membrane disruption. Pure Appl Chem. 82(1):219–226. https://doi.org/10.1351/PAC-CON-09-01-08
Tanaka M, Ishiguro K, Oki T, Okuno S (2017) Functional components in sweetpotato and their genetic improvement. Breed Sci 67:52–61. https://doi.org/10.1270/jsbbs.16125
Taofiq O, González-Paramás AM, Barreiro MF, Ferreira ICFR (2017) Hydroxycinnamic acids and their derivatives: cosmeceutical significance challenges and future perspectives a review. Molecules 22:281. https://doi.org/10.3390/molecules22020281
Truong VD, McFeeters RFM, Thompson RT, Dean LL, Shofran B (2007) Phenolic Acid Content and Composition in Leaves and Roots of Common Commercial Sweetpotato (Ipomea batatas L.) Cultivars in the United States. J Food Sci 72(6):343–349. https://doi.org/10.1111/j.1750-3841.2007.00415.x
Valiñas MA, Lanteri ML, Have AT, Andreu AB (2015) Chlorogenic Acid Biosynthesis Appears Linked With Suberin Production in Potato Tuber (Solanum tuberosum). J Agric Food Chem 63(19):4902–13. https://doi.org/10.1021/jf505777p
Valiñas MA, Lanteri ML, Have AT, Andreu AB (2017) Chlorogenic acid, anthocyanin and flavan-3-ol biosynthesis in flesh and skin of Andean potato tubers (Solanum tuberosum subsp. andigena). Food Chem 229:837–846. https://doi.org/10.1016/j.foodchem.2017.02.150
Venugopala KN, Rashmi V, Odhav B (2013) Review on natural coumarin lead compounds for their pharmacological activity. BioMed Res Intl. https://doi.org/10.1155/2013/963248
Venugopala KN, Gleiser RM, Kasumbwe K, Aldhubiab BE, Attimarad MV, Odhav B (2014) Evaluation of Halogenated Coumarins for Antimosquito Properties. Sci World J. https://doi.org/10.1155/2014/189824
Walter WM, Schadel WE (1981) Distribution of phenols in ‘Jewel’ sweetpotato [Ipomoea batatas (L.) Lam.] roots. J Agric Food Chem 29:904–906. https://doi.org/10.1021/jf00107a003
Wianowska D, Gil M (2019) Recent advances in extraction and analysis procedures of natural chlorogenic acids. Phytochem Rev 18:273–302. https://doi.org/10.1007/s11101-018-9592-y
Wojciechowska E, Weinert CH, Egert B, Trierweiler B, Schmidt-Heydt M, Horneburg B, Graeff-Hönninger S, Kulling SE, Geisen R (2014) Chlorogenic acid, a metabolite identified by untargeted metabolome analysis in resistant tomatoes inhibits the colonization by Alternaria alternata by inhibiting alternariol biosynthesis. Euro J Plant Patho 139:735–747. https://doi.org/10.1007/s10658-014-0428-3
Zhu F, Cai YZ, Yang X, Ke J, Corke H (2010) Anthocyanins, Hydroxycinnamic Acid Derivatives, and Antioxidant Activity in Roots of Different Chinese Purple-Fleshed Sweetpotato Genotypes. J Agric Food Chem 58:7588–7596. https://doi.org/10.1021/jf101867t
Funding
This study would not have been possible without the financial support of the Agence Nationale pour la Recherche (ANR, France), through the project no. ANR-10-STRA-007, and the technical assistance of the Department of Agriculture and Rural Development (DARD) in Vanuatu who provided technical support and labour for the establishment of field experiments and their maintenance. Special thanks are due to Ms. Elisha Tekak for laboratory assistance in preparing the samples and extracts.
Author information
Authors and Affiliations
Contributions
V.L.: designed the study, performed the HPTLC experiments, analysed and interpreted the HPTLC and statistical data, wrote and reviewed drafts of the manuscript, prepared the final writing. P.L. supervised field experiments and recorded field data. L.L.: contributed to the final writing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author declares that there are no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lebot, V., Leo, P. & Legendre, L. Phenotyping chlorogenic acids and coumarins in sweet potato [Ipomoea batatas (L.) Lam.] breeding lines for enhanced tolerance to periderm pathogens. Euphytica 217, 59 (2021). https://doi.org/10.1007/s10681-021-02808-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-021-02808-w