Abstract
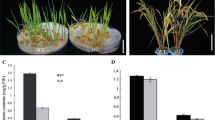
As the primary organ for photosynthesis, the leaf color is crucial for plant growth and development. In this study, we isolated a yellow green leaf 14 (ygl14) mutant from Xinong 1B, an indica restorer line of rice (Oryza sativa L.), which was treated with ethyl methane sulfonate. The mutant not only showed a yellow-green leaf phenotype for the entire life cycle, the flag leaf is significantly longer and the total leaf area is increased, which resulted in an increase in photosynthetic area. Leaf pigment (chlorophyll and carotenoids) contents were significantly lower in ygl14 than those in the wild type (WT). Compared with WT, stomatal conductance, photosynthetic rate, and transpiration rate of ygl14 were significantly reduced, whereas the intercellular CO2 concentration was significantly increased. Anatomical observation revealed that a portion of the chloroplasts in mesophyll cells of ygl14 were structurally impaired compared with those of WT. YGL14 was fine-mapped to a 70.7 kb region on chromosome 5, covered includes 15 open reading frames, no other yellow-green leaf related mutants have ever been reported within this region in rice. Quantitative real-time PCR analysis showed that the expression levels of genes associated with chlorophyll metabolism and photosynthesis were significantly altered in the ygl14 mutant. These results provide a foundation for cloning and functional analysis of the yellow-green leaf gene YGL14.






Similar content being viewed by others
References
Afsar Awan M, Konzak CF, Rutger JN, Nilan RA (1980) Mutagenic effects of sodium azide in rice. Crop Sci 20(5):663–668
Bansal U, Saini R, Kaur A (1999) Genetic variability in leaf area and chlorophyll content of aromatic rice. Int Rice Res Notes 24:21
Beale SI (2005) Green genes gleaned. Trends Plant Sci 10(7):309–312
Fang T, Yao S, Zhang J, Aziz A (2010) Viscous flow over a shrinking sheet with a second order slip flow model. Commun Nonlinear Sci Numer Simul 15(7):1831–1842
Fromme P, Melkozernov A, Jordan P, Krauss N (2003) Structure and function of photosystem I: interaction with its soluble electron carriers and external antenna systems. FEBS Lett 555:40–44
Huang J, Qin F, Zang G, Kang Z, Zou H, Hu F, Yue C, Li X, Wang G (2013) Mutation of OsDET1 increases chlorophyll content in rice. Plant Sci 210:241–249
Huq E, Al-Sady B, Hudson M, Kim C, Apel K, Quail PH (2004) Phytochrome-interacting factor l is a critical bHLH regulator of chlorophyll biosynthesis. Science 305:1937–1942
Jiang H, Li M, Liang N, Yan H, Wei Y, Xu X, Liu J, Xu Z, Chen F, Wu J (2007) Molecular cloning and function analysis of the stay green gene in rice. Plant J 52:197–209
Krushnir S, Babiychuk E, Storozhenko S, Davey MW, Papenbrock J, Rycke RD, Engler G, Stephan UW, Lange H, Kispal G, Lill R, Montagu MV (2001) A mutation of the mitochondrial ABC transporter Sta1 leads to dwarfism and chlorosis in the Arabidopsis mutant starik. Plant Cell 13:89–100
Kusaba M, Ito H, Morita R, Iida S, Sato Y, Fujimoto M, Kawasaki S, Tanaka R, Hirochika H, Nishimura M, Tanaka A (2007) Rice NON-YELLOW COLORING1 is involved in light-harvesting complex II and grana degradation during leaf senescence. Plant Cell 19:1362–1375
Kusumi K, Sakata C, Nakamura T, Kawasaki S, Yoshimura A, Iba K (2011) A plastid protein NUS1 is essential for build-up of the genetic system for early chloroplast development under cold stress conditions. Plant J 68:1039–1050
Lee S, Kim JH, Yoo ES, Lee CH, Hirochika H, An G (2005) Differential regulation of chlorophyll a oxygenase genes in rice. Plant Mol Biol 57:805–818
Leister D (2003) Chloroplast research in the genomic age. Trends Genet 9:47–56
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Liu F, Zhang L, Luo Y, Xu M, Fan Y, Wang L (2016) Interactions of Oryza sativa OsCONTINUOUS VASCULAR RING-LIKE 1 (OsCOLE1) and OsCOLE1-INTERACTING PROTEIN reveal a novel intracellular auxin transport mechanism. New Phytol 212:96–107
Michelmore RW, Paran I, Kesseli RV (1991) Identification of markers linked to disease-resistance genes by bulked segregant analysis: a rapid method to detect markers in specific genomic regions by using segregating populations. Proc Natl Acad Sci USA 88:9828–9832
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325
Panaud O, Chen X, McCouch SR (1996) Development of microsatellite markers and characterization of simple sequence length polymorphism (SSLP) in rice (Oryza sativa L.). Mol Gen Genet 252:597–607
Pérez-Ruiz JM, Spínola MC, Kirchsteiger K, Moreno J, Sahrawy M, Cejudo FJ (2006) Rice NTRC is a high-efficiency redox system for chloroplast protection against oxidative damage. Plant Cell 18:2356–2368
Reinbothe S, Pollmann S, Springer A, James RJ, Tichtinsky G, Reinbothe C (2005) A role of Toc33 in the protochlorophyllide-dependent plastid import pathway of NADPH: protochlorophyllide oxidoteductase (POR) A+. Plant J 42:1–12
Sugimoto H, Kusumi K, Tozawa Y, Yazaki J, Kishimoto N, Kikuchi S, Iba K (2004) The virescent-2 mutation inhibits translation of plastid transcripts for the plastid genetic system at an early stage of chloroplast differentiation. Plant Cell Physiol 45(8):985–996
Tanaka A, Tanaka R (2006) Chlorophyll metabolism. Curr Opin Plant Biol 9:248–255
Terry MJ, Kendrick RE (1999) Feedback Inhibition of chlorophyll synthesis in the phyochrome chromophore-deficient aurea and yellow-green-2 mutants of tomato. Plant Physiol 119:143–152
Wang J, Wang B, Zhou L, Xu J, Gu M, Liang G (2006) Genetic analysis and molecular mapping of a new yellow–green leaf gene ygl–2 in rice. Chin J Rice Sci 20:455–459
Wang P, Gao J, Wan C, Zhang F, Xu Z, Huang X, Sun X, Deng X (2010) Divinyl chlorophyll(ide) a can be converted to monovinyl chlorophyll(ide) a by a divinyl reductase in rice. Plant Physiol 153:994–1003
Woo HR, Chung KM, Park JH, Oh SA, Ahn T, Hong SH, Jang SK, Nam HG (2001) ORE9, an F-box protein that regulates leaf senescence in Arabidopsis. Plant Cell 13:1779–1790
Wu ZM, Zhang X, Wang JL, Wan JM (2014) Leaf chloroplast ultrastructure and photosynthetic properties of a chlorophyll-deficient mutant of rice. Photosynthetica 52:217–222
Yamatani H, Sato Y, Masuda Y, Kato Y, Morita R, Fukunaga K, Nagamura Y, Nishimura M, Sakamoto W, Tanaka A, Kusaba M (2013) NYC4, the rice ortholog of Arabidopsis THF1, is involved in the degradation of chlorophyll–protein complexes during leaf senescence. Plant J 74:652–662
Yoo SC, Cho SH, Sugimoto H, Li JJ, Kusumi K, Koh HJ, Iba K, Paek NC (2009) Rice virescent3 and stripe1 encoding the large and small subunits of ribonucleotide reductase are required for chloroplast biogenesis during early leaf development. Plant Physiol 150:388–401
Yoo JH, Park JH, Cho SH, Yoo SC, Li J, Zhang H, Kim KS, Koh HJ, Paek NC (2011) The rice bright green leaf, (bgl) locus encodes OsRopGEF10, which activates the development of small cuticular papillae on leaf surfaces. Plant Mol Biol 77:631–641
Zhang H, Li J, Yoo JH, Yoo SC, Cho SH, Koh HJ, Seo HS, Paek NC (2006) Rice Chlorina-1 and Chlorina-9 encode ChlD and ChlI subunits of Mg-chelatase, a key enzyme for chlorophyll synthesis and chloroplast development. Plant Mol Biol 62:325–337
Zhen XH, Xu JG, Shen WJ, Zhang XJ, Zhang QJ, Lu CG, Chen GX, Gao ZP (2014) Photosynthetic characteristics of flag leaves in rice white stripe mutant 6001 during senescence process. Rice Sci 21:335–342
Zhu XY, Guo S, Wang ZW, Du Q, Xing YD, Zhang TQ, Shen WQ, Sang XC, Ling YH, He GH (2016) Map-based cloning and functional analysis of YGL8, which controls leaf colour in rice (Oryza sativa). BMC Plant Biol 16:134
Acknowledgements
This work was supported by the National Key Research and Development Project (2017YFD0100201), Project of Chongqing Science and Technology Commission Grants CSTCCXLJRC201713 and CSTC2016SHMS-ZTZX0017.
Author information
Authors and Affiliations
Contributions
YHL designed the experiments and provided the resources for all experiments. JT performed the experiments, the data analysis and wrote the manuscript. TZ directed the experiment and modified the manuscript. SSX, MY and FFL helped in some experiments. XCS and GHH provided experimental materials. All authors have read, edited, and approved the current version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tan, J., Zhang, T., Xia, S. et al. Fine mapping of a novel yellow-green leaf 14 (ygl14) mutant in rice. Euphytica 215, 100 (2019). https://doi.org/10.1007/s10681-019-2424-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-019-2424-3