Abstract
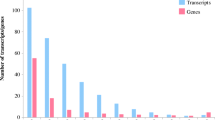
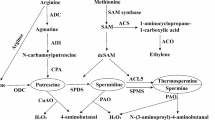
Camelina [Camelina sativa (L.) Crtz.] of the Brassicaceae family is an annual oilseed crop that has received increasing attention as a biofuel feedstock because of its excellent adaptability to low fertility drylands. Even though moisture is one of the critical factors affecting plant growth and seed yield, genetic studies on response to water deficit in camelina have been limited. This study aimed at identifying camelina genes responding to water deficit and validating their expression pattern. Camelina plants were subjected to 1.9 kPa (well-watered), 4.6 kPa (3 days after water stop), 73.2 kPa (17 days after water stop), and 2.0 kPa (rehydrated) soil water regimes. The transcriptome of test and control plants was analyzed using RNA sequences for de novo sequence assembly followed by gene annotation, which was performed against 39 plant species of the Phytozome database (http://www.phytozome.net). Six metabolic novel genes that responded to water deficit in camelina were selected for further analysis: PYRIMIDINE 4, CYP89A9, FASCICLIN-like arabinogalactan, eukaryotic aspartyl protease family protein, photosystem II light harvesting complex gene 2.3, and adenosylmethionine decarboxylase family protein. The expression pattern of these genes obtained by real-time reverse transcription PCR and quantitative reverse transcription PCR analysis were similar to that obtained by transcriptome analysis. The up-regulated genes are known to function in protecting cells against water deficit, whereas the genes with reduced expression were found to be suppressed by lower soil water causing a decrease in their function. The information obtained from the molecular validation of the changes in expression patterns of these six metabolic genes presents insight as to which genes of the camelina may account for actively responding to soil water deficit, which will be target loci for investigating the allelic variations or SNP screening in the camelina germplasm for future breeding program.



Similar content being viewed by others
References
Abe H, Yamaguchi-Shinozaki K, Urao T, Iwasaki T, Hosokawa D, Shinozaki K (1997) Role of arabidopsis MYC and MYB homologs in drought- and abscisic acid-regulated gene expression. Plant Cell 9(10):1859–1868. https://doi.org/10.1105/tpc.9.10.1859
Alberte RS, Thornber JP, Fiscus EL (1977) Water stress effects on the content and organization of chlorophyll in mesophyll and bundle sheath chloroplasts of maize. Plant Physiol 59(3):351–353
Alcázar R, Altabella T, Marco F, Bortolotti C, Reymond M, Koncz C, Carrasco P, Tiburcio AF (2010) Polyamines: molecules with regulatory functions in plant abiotic stress tolerance. Planta 231(6):1237–1249
Barber J (2002) Photosystem II: a multisubunit membrane protein that oxidises water. Curr Opin Struct Biol 12(4):523–530
Berti M (2016) Camelina uses, genetics, genomics, production, and management. Ind Crops Prod 94:690–710
Boyer JS (1982) Plant productivity and environment. Science 218(4571):443–448. https://doi.org/10.1126/science.218.4571.443
Chao WS, Wang H, Horvath DP, Anderson JV (2019) Selection of endogenous reference genes for qRT-PCR analysis in Camelina sativa and identification of Flowering locus C allele-specific markers to differentiate summer- and winter-biotypes. Ind Crops Prod 129:495–502
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103(4):551–560
Christ B, Süssenbacher I, Moser S, Bichsel N, Egert A, Müller T, Kräutler B, Hörtensteiner S (2013) Cytochrome P450 CYP89A9 is involved in the formation of major chlorophyll catabolites during leaf senescence in Arabidopsis. Plant Cell 25(5):1868–1880
Farooq M, Wahid A, Kobayashi N, Fujita D, Basra SMA (2009) Plant drought stress: effects, mechanisms and management. In: Lichtfouse E, Navarrete M, Debaeke P, Véronique S, Alberola C (eds) Sustainable agriculture. Springer, Netherlands, pp 153–188. https://doi.org/10.1007/978-90-481-2666-8_12
Fröhlich A, Rice B (2005) Evaluation of Camelina sativa oil as a feedstock for biodiesel production. Ind Crops Prod 21(1):25–31
Giardi M, Cona A, Geiken B, Kučera T, Masojidek J, Mattoo A (1996) Long-term drought stress induces structural and functional reorganization of photosystem II. Planta 199(1):118–125
Giermann N, Schröder M, Ritter T, Zrenner R (2002) Molecular analysis of de novo pyrimidine synthesis in solanaceous species. Plant Mol Biol 50(3):393–403
Golldack D, Lüking I, Yang O (2011) Plant tolerance to drought and salinity: stress regulating transcription factors and their functional significance in the cellular transcriptional network. Plant Cell Rep 30(8):1383–1391
Gugel R, Falk K (2006) Agronomic and seed quality evaluation of Camelina sativa in western Canada. Can J Plant Sci 86(4):1047–1058
Kanth BK, Kumari S, Choi SH, Ha H-J, Lee G-J (2015) Generation and analysis of expressed sequence tags (ESTs) of Camelina sativa to mine drought stress-responsive genes. Biochem Biophys Res Commun 467(1):83–93
Kitahata N, Saito S, Miyazawa Y, Umezawa T, Shimada Y, Min YK, Mizutani M, Hirai N, Shinozaki K, Yoshida S (2005) Chemical regulation of abscisic acid catabolism in plants by cytochrome P450 inhibitors. Bioorg Med Chem 13(14):4491–4498
Kushiro T, Okamoto M, Nakabayashi K, Yamagishi K, Kitamura S, Asami T, Hirai N, Koshiba T, Kamiya Y, Nambara E (2004) The Arabidopsis cytochrome P450 CYP707A encodes ABA 8′-hydroxylases: key enzymes in ABA catabolism. The EMBO J 23(7):1647–1656
Kwon S-T, Jeong H-J, Hasegawa PM (2010) Gene expression and response of arabidopsis AtSIZ3 mutants to temperature and drought stress. Korean J Plant Resour 23(1):25–30
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using realtime quantitative PCR and the 2−ΔΔCT method. Methods 4:402–408
Moore M (1994) Camelina comes in from the cold. Furrow 99:20–21
Nelson N, Yocum CF (2006) Structure and function of photosystems I and II. Annu Rev Plant Biol 57:521–565
Nelson DR, Koymans L, Kamataki T, Stegeman JJ, Feyereisen R, Waxman DJ, Waterman MR, Gotoh O, Coon MJ, Estabrook RW (1996) P450 superfamily: update on new sequences, gene mapping, accession numbers and nomenclature. Pharmacogenetics 6(1):1–42
Park E, Cho B-K (2014) Development of drought stress measurement method for red pepper leaves using hyperspectral short wave infrared imaging technique. Prot Hortic Plant Fact 23(23):50–55
Pilgeram AL (2007) Camelina sativa, a Montana omega-3 and fuel crop. In: Janick J, Whipkey A (eds) Issues in new crops and new uses. ASHS Press, Alexandria, pp 129–131
Putnam D, Budin J, Field L, Breene W (1993) Camelina: a promising low-input oilseed. New Crops Wiley, New York, p 314
Qi X, Bakht S, Qin B, Leggett M, Hemmings A, Mellon F, Eagles J, Werck-Reichhart D, Schaller H, Lesot A (2006) A different function for a member of an ancient and highly conserved cytochrome P450 family: from essential sterols to plant defense. Proc Natl Acad Sci 103(49):18848–18853
Schuler MA, Werck-Reichhart D (2003) Functional genomics of P450s. Annu Rev Plant Biol 54(1):629–667
Simova-Stoilova L, Vaseva I, Grigorova B, Demirevska K, Feller U (2010) Proteolytic activity and cysteine protease expression in wheat leaves under severe soil drought and recovery. Plant Physiol Biochem 48(2–3):200–206
Subburaj S, Kim AY, Lee S, Kim K-N, Suh MC, Kim G-J, Lee G-J (2016) Identification of novel stress-induced microRNAs and their targets in Camelina sativa using computational approach. Plant Biotechnol Rep 10(3):155–169
Wang W, Vinocur B, Altman A (2003) Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta 218(1):1–14
Yemets A, Boychuk YN, Shysha E, Rakhmetov D, Blume YB (2013) Establishment of in vitro culture, plant regeneration, and genetic transformation of Camelina sativa. Cytol Genet 47(3):138–144
Zhu J-K (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53(1):247–273. https://doi.org/10.1146/annurev.arplant.53.091401.143329
Acknowledgements
We are especially indebted to Drs. Byung Kwan Cho and Eunsoo Park (Department of Biosystems Machinery Engineering, Chungnam National University) for helping with implementations of soil water tension measurement and image acquiring. This research was supported by “Bio-industry Technology Development Program (No. 312033-5)”, IPET, Ministry of Agriculture, Food and Rural Affairs (MAFRA), and by “Plant Molecular Breeding Center of Next Generation BioGreen 21 Program (No. PJ01319303)”, Rural Development Administration, Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Choi, SH., Park, N., Lee, KY. et al. Novel genes in response to varying water deficit in oil crop Camelina sativa. Euphytica 215, 86 (2019). https://doi.org/10.1007/s10681-019-2402-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-019-2402-9