Abstract
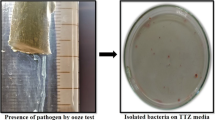
In the present investigation, optimal media compositions for enhancing the callus induction and subsequent plant regeneration were worked out in sugarcane (Saccharum spp. complex). The field grown somaclones were characterized for yield and quality contributing traits in comparison to donor variety CoJ 88. The MS + 2,4-D (4.0 mg/l) + kin (0.5 mg/l) + sucrose (30 g/l) was found to be the best for the callus induction among the nine media compositions used. The highest shoot regeneration was 34.65% on MS + BAP (0.5 mg/l) followed by 31.07% on MS + IAA (2.0 mg/l) + kin (0.5 mg/l) and the least 11.80% on MS + BAP (2.0 mg/l). The shoot elongation was optimum on MS + IBA (5.0 mg/l). The rooting was carried on MS + NAA (3.0 mg/l) + IBA (2.0 mg/l) + sucrose (70 g/l). The somaclones, on field evaluation, exhibited huge variability for characters viz., tiller number at 120 days of field transfer, number of millable canes, cane height, number of nodes and internodal length at time of harvest and also for HR Brix at 240 and 300 days after field transfer. In R0 generation, the variations induced for the characters such as stalk height after 120 days, HR Brix after 240 and 300 days of field transfer were negatively skewed with most of somaclones having values higher than the population mean values thereby offering adequate scope for improvement cane and quality. Elite somaclones marked for each trait were reported. Out of 228 somaclones inoculated with Cf 08 pathotype of red rot in R1, three were resistant, four were moderately resistant and 221 somaclones showed variable degree of susceptibility, whereas, to Cf 03 pathotype, fourteen were resistant, nineteen were moderately resistant and 195 showed variable degree of susceptibility. Three somaclones exhibited moderate resistance (MR) to both the pathotypes, whereas, donor variety CoJ 88 showed susceptible reaction. These somaclones will further enhance the germplasm for sugarcane breeding.









Similar content being viewed by others
Abbreviations
- BAP:
-
6-benzylaminopurine
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- IAA:
-
Indole-3-aceticacid
- IBA:
-
Indole-3-butyricacid
- kin:
-
Kinetin (6-furfuryl aminopurine)
- NAA:
-
Naphthaleneacetic acid
References
Brown PTH (1991) The spectrum of molecular changes associated with somaclonal variation. Int Assoc Plant Tissue Cult Newslett 66:14–25
Cheema AS, Singh H, Gosal SS (1992) Response of different genotypes to callus induction and plant regeneration in sugarcane. Crop Improv 19:6–12
Dhumale BD, Ingola GL, Durge DV (1994) Variation for morphological and quality attributes in clones of callus regenerants in sugarcane cv. CoC 671. Indian J Genet 54:317–320
Fitch MMM, Moore PH (1993) Long term culture of embryogenic sugarcane callus. Plant Cell Tissue Organ Cult 32:335–343
Gallo-Meagher M, Irvine JE (1996) Herbicide resistant transgenic sugarcane plants containing bar gene. Crop Sci 36:1367–1374
Gill NK, Gill R, Gosal SS (2002) Somatic embryogenesis and plant regeneration in some sugarcane cultivars of sugarcane. Crop Improv 29:28–34
Gosal SS, Thind KS, Dhaliwal HS (1998) Micropropagation of sugarcane-an efficient protocol for commercial plant production. Crop Improv 25:1–5
Heinz DJ, Krishnamurthy M, Nickell LG, Maretzki A (1977) Cell, tissue and organ culture in sugarcane improvement. In: Reinert J, Bajaj YPS (eds) Applied and fundamental aspects of plant cell, tissue and organ culture. Springer-Verlag, Berlin, Heidelberg, New York, pp 3–17
Heinz DJ, Mee GWP (1969) Plant differentiation from callus tissue of Saccharum spp. Crop Sci 9:346–348
Ho WJ, Vasil IK (1983) Somatic embryogenesis in sugarcane (Saccharum oficinarum L.): the morphology and physiology of callus formation and the ontogeny of somatic embryos. Protoplasma 118:169–180
Irvine JE (1984) The frequency of the marker changes in sugarcane plants regenerated from callus cultures. Plant Cell Tissue Organ Cult 3:202–209
Kaur A (1999) Induction of somaclonal variation in sugarcane (Saccharum oficinarum L.). M.Sc. Thesis, Punjab Agricultural University, Ludhiana, India
Kaur A (2004) Genetic transformation in sugarcane varieties. Ph.D. Dissertation, Punjab Agricultural University, Ludhiana, India
Kaur A, Gosal SS, Gill Raman, Thind KS (2001) Induction of plant regeneration and somaclonal variation for some agronomic traits in sugarcane (Saccharum oficinarum L.). Crop Improv 28:167–172
Kendall GM, Stuart A (1968) The advanced theory of statistics. Charles Griffin and Company Limited, London
Larkin PJ (1982) Sugarcane tissue and protoplast culture. Plant Cell Tissue Organ Cult 2:149–164
Larkin PJ, Scowcroft WR (1981) Somaclonal variation—a novel source of variability from cell cultures for plant improvement. Theor Appl Genet 60:197–214
Liu MC, Chen WH (1974) Histological studies on the origin and process of plantlets differention in sugarcane callus mass. Proc Soc Sugarcane Technol 15:118-121
Liu MC, Chen WH (1978) Tissue and cell culture an aid to sugarcane breeding. II. Performance and yield potential of callus derived lines. Euphytica 27:273–282
Maheswaran M, Rangasamy SRS (1989) Influence of genotypes and culture media on callus induction and plant regeneration in Oryza spp. J Genet, Plant Breed 43:99–106
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Plant Physiol 15:473–497
Orshinsky KK, McGregor LJ, Johnson GIE, Hull P, Kartha KK (1990) Improved embryoid induction and green shoot regeneration from wheat anthers cultured on medium with maltose. Plant Cell Rep 9:365–369
Pang HH (1987) Studies on induction frequency of plantlets derived from pollen in O sativa L spontana Roschevicz. Acta Agro Sin 13:255–256
Preece JE, Compton ME (1999) Problems with explant exuclation in micropropagation. In: Bajaj YPS (ed) Biotechnology in agriculture and foresty. Springer-Verlag, Berlin, pp 168–189
Samad MA, Begum S (2000) Somaclonal variation of nonirradiated and irradiated calli of sugarcane (Saccharum oficinarum L.). Plant Tissue Cult 10:25–29
Sarao NK, Gill MS, Gosal SS (2003) Induction of haploids/double haploids through pollen culture in an Indica × Japonica rice cross. Crop Improv 30:120–124
Singh A, Lal M, Singh MP, Lal K, Singh SB (2000) Variation for red rot resistance in somaclones of sugarcane. Sugar Tech 2:56–58
Srinivasan KV, Bhat NR (1961) Red rot of sugarcane-criteria for grading resistance. J Indian Bot Soc 40:565–577
Taylor PWJ, Ko HL, Adkins SW, Rathus C, Birch RG (1992) Establishment of embryogenic callus and high protoplast yielding suspension cultures of sugarcane (Saccharum spp hybrids). Plant Cell Tissue Organ Cult 28:69–78
Vasil IK (1987) Improvement of cereals and grass crops through in vitro techniques. SABRAO J 19:127–128
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, G., Sandhu, S.K., Meeta, M. et al. In vitro induction and characterization of somaclonal variation for red rot and other agronomic traits in sugarcane. Euphytica 160, 35–47 (2008). https://doi.org/10.1007/s10681-007-9531-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-007-9531-2