Abstract
The quality of surface water could be influenced by both anthropogenic and natural factors. This study was designed to determine the impact of informal settlement and wastewater treatment plants on helminth egg contamination of urban rivers and the risks associated with everyday use. We also ascertained the accumulation of these eggs in the river sediments. The study was carried out in two rivers in the eThekwini Municipality of South Africa. Grab samples were taken at different points over a 10-month period. Ascaris spp., hookworm, Toxocara spp., Trichuris spp. and Taenia spp. were the helminth eggs detected in both the water column and sediments, with mean Ascaris spp. eggs of 0–6.3 (± 5.1)/L in the water and 0–6.8 (± 5.2)/kg in sediment samples. The helminth egg concentrations showed seasonal variation, probably due to changes in infection levels of the populations or natural factors, such as rainfall. The informal settlements had a greater impact than treated wastewater. For every 10,000 recreational users of the rivers 19 to 58 may be infected under undisturbed conditions, increasing to 29–88 individuals when the riverbed is disturbed. The risk from agricultural use of the rivers was above the tolerable risk values applicable for wastewater reuse, recommended by the World Health Organization. This calls for a re-evaluation of the policies governing surface water quality assessment, where the inclusion of helminth eggs and sediment monitoring will be critical.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Surface water quality is an important factor affecting not only human health but the entire ecological system (Wang et al. 2013). This is most important in urban areas where rivers are impacted by several anthropogenic and natural factors. For example, anthropogenic activities such as industrial, agricultural and chemical spills and dam construction are major contributors to the quality of surface water (Qadir et al. 2008). These are cumulative in nature over time and space (Gazzaz et al. 2012a). Additionally, natural processes such as erosion and climatic conditions may also affect surface water quality (Zhang et al. 2010). River quality is not only an indication of the health of the river but may also reflect the health of the surrounding landscapes (Zhou et al. 2012).
Pollution from other sources such as wastewater treatment plants has an impact on river ecosystems (Bernhardt and Palmer 2007; Grant et al. 2012), affecting their everyday use. Wastewater effluents may still contain a complex mixture of contaminants such as pharmaceuticals and personal care products (Kuster et al. 2008; Ginebreda et al. 2010), microorganisms (Gazzaz et al. 2012b; Jagals 1997) and nutrients (Aristi et al. 2015). A decline in the performance of wastewater treatment plants in South Africa has been reported; in 2010, 53% of these plants were within compliance limits (Dungeni et al. 2010). However, by 2019, an average of 67% of South Africa’s sewage systems were not functioning properly (Herbig and Meissner 2019). Therefore, the discharge of effluents into surface water may result in contamination and subsequent public health issues. In addition, informal settlements, sometimes referred to as slums, may contribute to the pollution of surface water within urban areas (Abia et al. 2018; Bandyopadhyay and De 2017). The impact from these informal settlements adds to the pollution of urban surface water by wastewater treatment plant effluents.
The potential contamination of surface water from all these sources highlights the importance of water quality monitoring. However, these water quality monitoring approaches mainly focus on bacteria, such as E. coli and a few other coliforms (Dalla Vecchia et al. 2015; Kirschner et al. 2017; Egbueri 2019). In monitoring surface water quality, less attention has been paid to helminth egg contamination, except in instances when contamination with wastewater is suspected (Amoah et al. 2016; Fuhrimann et al. 2017). Except for wastewater reuse guidelines, no other water guideline (drinking or recreational) considers helminths as a major health threat. However, exposure to wastewater or faecally contaminated surface water has been shown to exhibit high risk of helminth infections (Amoah et al. 2016; Fuhrimann et al. 2017; Ribas et al. 2017; Mather et al. 2020). This is therefore a major gap in ensuring the protection of public health, especially in areas where helminth infections are high. For instance, although the total number of people infected with soil-transmitted helminths (STHs) in South Africa is unknown (Molvik et al. 2017), it is estimated that over 3 million children require treatment (WHO 2014). Therefore, rivers within urban areas with poor sanitation and in areas receiving wastewater effluents could be impacted by faecal contamination and in turn act as a transmission route. The risk of infection associated with wastewater or faecally contaminated wastewater during irrigation practices has been studied extensively (Fuhrimann et al. 2016; Amoah et al. 2018; Msoffe 2019); however, within urban settings, the use of the surface water goes beyond irrigation to include, potentially, recreation and other domestic uses. Additionally, the water quality monitoring relies on the water column alone without assessing the impact of river sediments (Jamieson et al. 2004; Bai and Lung 2005; Characklis et al. 2005; Fries et al. 2008). Microorganisms in the aquatic environment have the tendency to either settle, depending on their settleability, or become attached to fine suspended sediment particles (Gao et al. 2011; Abia et al. 2015). Therefore, under disturbed conditions, these may result in an increase in their concentrations in the water column leading to potentially higher risks of infections.
In the present study, we highlight the importance of the inclusion of STH analysis to water quality monitoring especially in areas with poor sanitation and wastewater effluent influence. We also show that the addition of sediment samples in the river or surface water quality monitoring may give a much more efficient estimation of potential risks. The use of the quantitative microbial risk assessment approach makes it possible to determine the potential risks of helminth infections for populations exposed to contaminated rivers. Our study therefore contributes to the design of appropriate water quality monitoring approaches and estimation of health.
Methodology
Study area
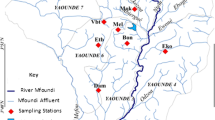
This study was performed in two catchments, the Isipingo and Palmiet rivers within the city of Durban, in the eThekweni municipality of South Africa. The Isipingo River is located about 20 km south of the central business area of the city and is approximately 27 km long (Pillay 2013). The Palmiet River is approximately 25 km and located within the northern periphery of the city of Durban. Table 1 presents more details about the sample points within the study areas.
Sampling
Surface water and sediment samples were collected monthly from January to October 2016. Grab samples were taken in triplicates of 1 L using sterilized containers, approximately 0.5 m below the water surface at each sampling point. Care was taken not to disturb the sediments. Sediment sampling was adapted from Adeyinka et al. (2019); briefly, composite sediment samples were taken from the top 5 cm at each point by using a hand-held spade to collect the sediments into a 500-mL bucket.
Laboratory analysis
The water samples were analysed for helminth eggs using a modified method based on the principle of centrifugation and flotation presented in Amoah et al. (2018). Only viable helminth eggs determined using the method referenced were counted and reported in this paper. All the pellets incubated were viewed under the microscope (×100), counted and reported per 1 kg for the sediments and 1 L for the water samples.
Statistical analysis
The concentration of eggs at the different sampling points and sites was described through descriptive statistics using Excel (2016 version, Microsoft Corporation). To determine the statistical significance or otherwise, difference in concentration of the eggs at the sampling points was determined using the Kruskal-Wallis tests and the Mann-Whitney U test used to compare the concentrations between the helminth eggs in the water column and the sediments using a 95% confidence interval (Bethea et al. 1995). Additionally, seasonal variation was determined by comparing the concentrations of the different helminth eggs over the four seasons prevalent in South Africa, thus autumn: March–May, winter: June–August, spring: September–October and summer: January–February. This was performed using the Kruskal-Wallis tests with Dunn’s multiple-comparison test afterwards. All statistical analysis was performed in Graphpad Prism 7 software (GraphPad Software, Inc. USA).
Risk assessment
The quantitative microbial risk assessment (QMRA) approach was used to assess the risks of helminth infections as described by the four steps below:
Hazard identification
For the purposes of QMRA, only Ascaris spp. have a dose-response model, which was therefore chosen as the index for the helminths.
Exposure assessment
In this study, three exposure scenarios were considered; exposure during recreation, irrigation and indirectly through consumption of irrigated vegetables.
Dose-response assessment
The exponential dose-response model (Westrell 2004; Seidu et al. 2008) given by the formula below was used:
where Pinf is the probability of infection associated with the ingestion of Ascaris spp. eggs, r the dimensionless infectivity constant for Ascaris spp. and d the dose of the eggs ingested under each scenario considered. An r value of 0.039 was used in this assessment (Navarro et al. 2008). The dose of Ascaris spp. eggs ingested per exposure was modelled by fitting a probability distribution function to the concentrations reported in this study. Increase in Ascaris spp. concentration in the water column from disturbance was also considered where it was assumed that concentrations will increase by 30–55% (Krometis et al. 2007).
The dose of Ascaris spp. eggs ingested during recreational use or during irrigation of crops was determined using the formula:
where D is the concentration (dose) ingested by the swimmer or farmer, Craw the concentration of Ascaris spp. eggs per millilitre and V the volume (mL/day) ingested by swimmer or farmer. The dose (DC) of Ascaris spp. eggs ingested by consumers was modelled with lettuce as a surrogate vegetable using the formula
where V is the volume of water caught on the lettuce in millilitres per gram of lettuce, I the mean per capita intake of lettuce in grams per person per day and c the concentration of Ascaris spp. eggs in the water used for irrigation. The different exposure scenarios and volumes ingested are presented in Table 2.
Risk characterization
Risks of infection from multiple exposures were determined using the formula;
where P1(A) is the risk of infection after multiple exposures, P1(d) the risk of infection from a single exposure to a dose d of the Ascaris spp. egg and n is the number of days of exposure to the single dose d (Sakaji and Funamizu 1998).
Results
Concentration of helminth eggs in water and sediments
Eggs of Ascaris spp., hookworm, Toxocara spp., Trichuris spp. and Taenia spp. were detected in both the water and sediments, with Ascaris spp. and hookworm the most abundant. In the Isipingo River, Ascaris spp. eggs ranged from 0 to 6.3 (± 5.1)/L in the water and 0–6.8 (± 5.2)/kg in the sediments. Similarly, for hookworm eggs, high concentrations were found in the sediments (0–6.6 (± 5.7)/L) (Table 3). The highest concentration of eggs was found at the sampling point next to the transit camp (point 3) both for the water and sediment samples. These differences in egg concentrations were statistically significant (p value ≤ 0.05).
The occurrence of helminth eggs was similar both in relation to speciation and abundance in the water and sediments from Palmiet River. Mean Ascaris spp. eggs was 10 (± 8.4)/L and 12.9 (± 8.2)/kg in the water and sediments respectively. Taenia spp. were less abundant than others in the water samples (4.0 (± 3.5)/L), and Toxocara spp. eggs in the sediment samples had a mean concentration of 4.5 (± 3.1)/kg. These differences were not statistically significant (p value ≥ 0.05). The sampling point at the beginning of the informal settlement (PA 2) recorded the highest egg concentration. For example, the mean Ascaris spp. egg concentration in the water from PA 2 was 10 (± 8.5)/L while corresponding counts were 3.4 (± 3.4)/L for the sampling point where the Palmiet River joins the Umgeni River (PA 4) (Table 4). The difference in helminth egg concentrations at the various sampling points was statistically significant (p value ≤ 0.05).
Variation in helminth egg concentration over the study period
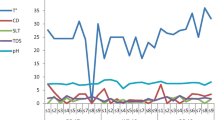
In the Isipingo River, mean Ascaris spp. egg concentrations in the water increased steadily from February to April, then dropped to the lowest of 0.5 (± 1)/L in June. Then again, from a mean concentration of 1.5 (± 3)/L in July, the concentration increased to 4 (± 4.8)/L in October. In contrast, Ascaris spp. eggs in the sediments saw a steady increase from 0 in March to 7 (± 5.8)/kg in June and declined to 0 in October. Similar trends were found for the other helminths. These variations in the concentrations were statistically significant (p value ≤ 0.05).
There was an observed difference in helminth egg concentrations considering the different seasons within the study area. In the water samples from the Isipingo River, the observed difference was statistically significant (p value ≤ 0.05). Spring had the highest concentrations for almost all the helminths identified; Ascaris spp. (3.9 ± 0.2 eggs/L), hookworm (3.4 ± 1.2 eggs/L), Trichuris spp. (2.8 ± 0.4 eggs/L) and Taenia spp. (3.9 ± 0.2 eggs/L). The only exception was observed for Toxocara spp., where the highest concentrations were observed in summer (3 ± 0.7 eggs/L). However, the difference in egg concentrations in the sediments did not exhibit any statistically significant differences. No one season stood out in terms of egg concentrations.
In the Palmiet River, the differences in the concentrations over the months were much clearer than in the Isipingo River. For instance, as shown in Figs. 1 and 2, the concentrations of Ascaris spp., hookworm and Trichuris spp. eggs (respectively) were higher in the months of March to June. In addition, the overall concentration of these eggs in the Palmiet River was higher than in the Isipingo River as can be seen by comparing results in Figs. 1 and 2. Water samples from the Palmiet River did not show any statistical difference in relation to seasonal variation in egg concentrations. However, mean egg concentrations were higher in summer for Ascaris spp. (8.8 ± 3.2 eggs/L), Trichuris spp. (5.4 ± 2.3 eggs/L) and Taenia spp. (3.8 ± 0.4 eggs/L). Hookworm and Toxocara spp. concentrations were highest in spring (3.8 ± 1.8 and 4.3 ± 0.4 eggs/L respectively). However, in the sediment samples analysed, the differences in egg concentrations between the four seasons were statistically significant (p value ≤ 0.05). In these sediments, the highest concentrations were observed for the seasons of autumn (Ascaris spp. (10.7 ± 1.4 eggs/kg), hookworm (6.1 ± 2.3 eggs/kg) and Taenia spp. (6.2 ± 3.9 eggs/kg)) and winter (Trichuris spp. (6.0 ± 1.8 eggs/kg) and Toxocara spp. (5.5 ± 1.8 eggs/kg)).
Risk of Ascaris spp. infection after ingestion of eggs
Infection with helminths is associated with exposure to the water either through intentional or accidental ingestion. Recreational use (swimming or bathing) of the Palmiet River may result in median risks of 19 to 58 out of 10,000 people being infected under normal conditions. However, with disturbance of the sediments, the risks increased to between 29 to 88 out of 10,000; the difference in risk from under the normal and disturbed conditions was statistically significant (p value ≤ 0.05). The highest risks as expected were observed at points with high contamination as described above (Table 5). The risks of Ascaris spp. infections for swimmers at the Isipingo River were much lower than those for the Palmiet River. For one-time exposures, the risks were in the range from 2 out of 100, 000 to 33 out of 10,000 under normal conditions and 6 out of 100,000 to 50 out of 10,000 when the sediments are disturbed with increase for annual exposures (Table 5).
Similar difference in risks between the two rivers was observed for irrigational use of the water by farmers; in the Palmiet River, one time use of the water results in risks of infections ranging from 4 to 12 out of 10,000 farmers infected under normal water conditions, with an increase when the sediments are disturbed (Table 6).
Consumption of lettuce irrigated with water from the Palmiet River may lead to 8 out of 10,000 to 23 out of 10,000 of the consumers infected under normal water conditions. Corresponding risks from the Isipingo River were 11 out of 100,000 to 14 out of 10,000 consumers (Table 7). Annual consumption of the lettuce increases the risks significantly.
Discussion
The dominance of Ascaris spp. and hookworm eggs in the water and sediments could be attributed to the human infections reported in South Africa (Appleton et al. 2009; Mkhize-Kwitshana and Mabaso 2014; Molvik et al. 2017). Additionally, our previous studies in the area gave a similar profile of helminth eggs in wastewater and sludge (Amoah et al. 2018). Therefore, the type and concentration of the helminths reported in this study are corroborated by these reports from clinical infections and wastewater/sludge analysis. The concentrations in the water column could also be influenced by natural events such as rainfall. For instance, rainfall may result in the increase of pathogen concentrations in water columns due to resuspension of the sediments (Muirhead et al. 2004; Griffith et al. 2010). Krometis et al. (2007) reported an increase by 30–55% of the presence of different indicator organisms under disturbed conditions. With settling velocities of 0.65 m/h for Ascaris spp. eggs and 1.53 m/h for Trichuris spp. eggs (David and Lindquist 1982; Dryden et al. 2005), most of these eggs may be in the sediments under normal river conditions with an expected increase in the water column following sediment disturbance. This could account for the significantly higher egg concentrations in the sediments from the Palmiet River than the water, for Ascaris spp., hookworm and Taenia spp., which may be due to the impact of the informal settlements in this study area.
The difference in the helminth egg concentration at the different sampling points could be attributed to the various activities along these rivers. For instance, in the Palmiet River, the highest concentrations were found at points directly influenced by informal settlements. Collectively, the two sampling points within the informal settlements had an average of 5.8 (± 1.9) eggs/L and 7.4 (± 3.0) eggs/kg for water and sediments respectively. Comparatively, the first sampling point (PA 1) had no helminth eggs, probably due to the absence of direct human contact or impact with the river at this section. PA 4 had low concentration (3.5 (± 1.1) eggs/L for water and 4.3 (± 1.3) eggs/kg for the sediments) compared to the two points within the settlement. This sampling point is located about 1 km away from the centre of the settlement (downstream) and may be the reason for the lesser helminth egg concentrations. Helminth infection is strongly correlated with the socio-economic status of the population (Stolk et al. 2016). Inhabitants of these informal settlements are usually migrants who moved to the cities in search of jobs; they live in make-shift accommodations. Therefore, these informal settlements are characterized by poor socio-economic and housing conditions with poor sanitation. Linked with the lack of proper sanitation, some of the inhabitants especially the children (who are the most vulnerable group) defecate near these rivers contributing to the high helminth egg concentrations reported at these points. The impact of open defecation on surface water contamination has been observed by other studies (Semwal and Akolkar 2006; Vijay et al. 2011). The impact of the informal settlements on the river water quality was higher than that of the wastewater treatment although these were not on the same river. Based on calculations, the wastewater effluents contributed 2.3 (± 0.4) eggs/L and 2.8 (± 0.6) eggs/kg for the water and sediments, respectively, to the concentration of the helminth eggs in the river. Additionally, well-functioning wastewater treatment plants are expected to reduce the concentration of these parasites as well as other pathogens before discharge, which may have also contributed to the lesser impact from these plants in the study. For instance, in our assessment of the wastewater treatment plant discharging into the Isipingo River, we observed removal efficiency between 72 and 100% for helminth eggs (Amoah et al. 2018). Therefore, the influence will be lesser compared to the informal settlements where open defecation may result in the direct deposition of the eggs into the river without treatment. A study in Argentina reported an average A. lumbricoides concentration of 5 eggs/L in the Arias-Arenales River (Kundu et al. 2014). The concentrations reported in some of the sections of the two rivers we studied were similar to the Argentinian study; however, concentrations in the Palmiet River were higher especially within the informal settlement than the report from Argentina.
The variations in the helminth egg concentration over the 10-month study period may just reflect a normal variation between grab samples or be influenced by environmental factors. For instance, during the months of May–July, the concentrations were higher in the sediments than the surface water. These months are characterized by lower rainfall levels, resulting in slower flow rate of the rivers which may aid egg settling. With rainfall, the flow rate increases as well as dislodgement of the eggs into the water column. This was seen in both rivers, but more evident in the Palmiet River, which is the most influenced by an informal settlement. Seasonality has been associated with different diseases (Pafčo et al. 2017; Martinez 2018; Mayengue et al. 2020; Poulin 2020). It has been reported that each disease has its own window of occurrence which may vary from one geographical location to another (Martinez 2018). In this study, the observed seasonal variation in helminth egg concentrations, although not statistically significant in some instances, may be an indication of this seasonal dependent infection dynamics. The infection dynamics could be as a result of an increased exposure to these parasites in the water, resulting in infections, or it could be that the increased infections resulted in increased occurrence in the rivers. Considering that most of these are STHs that require the soil in their life cycle to become infectious, the former scenario (increased exposure) could be the most likely reason. Although there is lack of information on seasonal impact on helminth infections in humans, in sheep and other livestock, an increase in intestinal nematode infections has been observed (Waller et al. 2004; Ahmed 2017).
The determined risks from recreational use of these rivers resulted in lower likelihood of ascariasis as expected. For instance, 19 to 58 and a maximum of 33 people out of 10,000 are at risk of infection for the recreational use of the Palmiet and Isipingo rivers, respectively. Although these numbers are low, this reflects no disturbance conditions and therefore does not factor in the increase in egg concentrations in the surface water during events such as storms and rainfall. With appropriate incorporation of the effect of disturbance, these risks increase to levels that call for public health concern (refer to the section on “Risk of Ascaris spp. infection after ingestion of eggs”). Annual exposure to the water will result in increased risks of infection (Tables 5, 6, and 7), therefore creating major public health concerns. There is a lack of studies on the risks of helminth infections from recreational use of contaminated rivers. However, the Argentinian study referenced earlier (Kundu et al. 2014) reported that accidental ingestion of water during recreation by children resulted in risks of 1.31 × 10−4; in adults the risks were lower (6.47 × 10−5), as well as secondary recreators (6.50 × 10−6). Therefore, our risk estimates from recreational exposure are corroborated by that study.
In contrast to recreational use, a lot of attention has been placed on the risks of helminth infections due to agricultural use of wastewater or faecally contaminated surface water. To protect public health, the WHO recommended that wastewater used for unrestricted agriculture should have ≤ 1 helminth egg per litre (WHO 2006). However, the surface water which is influenced by wastewater contains eggs above this recommended levels. Therefore, we observed a high risk of infection for farmers using the Palmiet River, which was higher than the tolerable risk (10−3 per person per year) value recommended by the WHO (Mara et al. 2007), as well as risks estimated for the Arias-Arenales River in Argentina (10−4). However, the risks of infections for the farmers using the Isipingo River were much lower than the tolerable risk values from WHO. Similar risks were observed for consumers of lettuce irrigated with the river water, also higher than the WHO tolerable risks figures for consumers (WHO 2006) when the Palmiet River is considered. This was determined with the assumption that no further reduction in concentrations will occur from the point of harvest to consumption. However, Amoah et al. (2011) reported that washing or disinfection of the vegetables with bleach or vinegar could potentially reduce risks of infections. In addition, cessation of irrigation for some days before harvesting has been recommended (Keraita et al. 2007). However, this approach may not be applicable, especially under dry conditions, where without irrigation for a few days, produce loss may occur. Several studies have looked at the risks of helminth infections for farmers and consumers using wastewater (Barker et al. 2014; Seidu et al. 2008). The reports from these studies indicate that the concentration of the helminth eggs in the irrigation water is the key factor in ascertaining the level of risks. These were the considerations used in developing the WHO wastewater reuse guidelines mentioned above. Additionally, these eggs may accumulate in the soil after each irrigation activity (Seidu et al. 2008) and survive for longer periods of time (Zdybel et al. 2015: Gaspard et al. 1995), increasing the risks of infection further.
Conclusion
The microbial quality of the two rivers studied was found to be poor with high concentrations of helminth eggs, especially in the sediments. It is therefore important that assessment of river water quality includes sediment analysis to give a better assessment. The high concentration of the helminth eggs highlight the importance of helminth analysis in addition to the routine indicator organism analysed. This is especially very critical in urban settings where faecal contamination of surface water is common, as shown in this study. This is because daily use of these urban rivers may increase helminth infections in these areas for exposed populations. Additionally, there has been a lot of focus on the role wastewater treatment plants play in surface water contamination; however, our study has shown that informal settlements or slums with poor sanitation may have a much higher impact. It is therefore imperative that for a long-term solution to urban pollution of rivers, we should also focus on improving sanitation coverage in our inner cities, especially slums and informal settlements, in addition to improvements in wastewater treatment.
References
Abia, A. L. K., Ubomba-Jaswa, E., & Momba, M. N. B. (2015). Impact of seasonal variation on Escherichia coli concentrations in the riverbed sediments in the Apies River, South Africa. Science of the Total Environment, 537, 462–469.
Abia, A. L. K., Alisoltani, A., Keshri, J., & Ubomba-Jaswa, E. (2018). Metagenomic analysis of the bacterial communities and their functional profiles in water and sediments of the Apies River, South Africa, as a function of land use. Science of the Total Environment, 616–617, 326–334.
Adeyinka, G. C., Moodley, B., Birungi, G., & Ndungu, P. (2019). Evaluation of organochlorinated pesticide (OCP) residues in soil, sediment and water from the Msunduzi River in South Africa. Environmental Earth Sciences, 78(6), 223.
Ahmed, M. A. A. (2017). Epidemiology and seasonal dynamics of internal parasite infections in small ruminants at Ukulinga Research Farm, South Africa. Journal of Veterinary Medicine and Animal Production, 7(1).
Amoah, P., Keraita, B., Akple, M., Drechsel, P., Abaidoo, R. C., & Konradsen, F. (2011). Low-cost options for reducing consumer health risks from farm to fork where crops are irrigated with polluted water in West Africa. International Water Management Institute Research Report, 141, 44–47.
Amoah, I. D., Abubakari, A., Stenström, T. A., Abaidoo, R. C., & Seidu, R. (2016). Contribution of wastewater irrigation to soil transmitted helminths infection among vegetable farmers in Kumasi, Ghana. PLoS Neglected Tropical Diseases, 10(12), e0005161. https://doi.org/10.1371/journal.pntd.0005161.
Amoah, I. D., Reddy, P., Seidu, R., & Stenström, T. A. (2018). Removal of soil-transmitted helminth egg in selected centralized and decentralized wastewater treatment plants in South Africa and Lesotho: health implications for direct and indirect exposure to the effluents. Environmental Science and Pollution Research, 25(13), 12883–12895.
Appleton, C. C., Mosala, T. I., Levin, J., & Olsen, A. (2009). Geohelminth infection and reinfection after chemotherapy among slum-dwelling children in Durban, South Africa. Annals of Tropical Medicine and Parasitology, 103(3), 249–261.
Aristi, I., Von Schiller, D., Arroita, M., Barceló, D., Ponsatí, L., Garćia-Galán, M. J., Sabater, S., Elosegi, A., & Acuña, V. (2015). Mixed effects of effluents from a wastewater treatment plant on river ecosystem metabolism: subsidy or stress? Freshwater Biology, 60(7), 1398–1410.
Bai, S., & Lung, W. (2005). Modeling sediment impact on the transport of fecal bacteria. Water Research, 39(20), 5232–5240.
Bandyopadhyay, S., De, S. K. (2017). Impact of slums and rural clusters on the Haora River. In: Human interference on river health. Advances in Asian Human-Environmental Research. Springer. Switzerland. https://doi.org/10.1007/978-3-319-41018-0.
Barker, S. F., Amoah, P., & Drechsel, P. (2014). A probabilistic model of gastroenteritis risks associated with consumption of street food salads in Kumasi, Ghana: evaluation of methods to estimate pathogen dose from water, produce or food quality. Science of the Total Environment, 487, 130–142.
Bernhardt, E. S., & Palmer, M. A. (2007). Restoring streams in an urbanized word. Freshwater Biology, 52(4), 738–751.
Bethea, R. M., Duran, B. S., & Boullion, T. L. (1995). Statistical methods for engineers and scientists (3rd ed.). New York: Marcel Dekker.
Characklis, G. W., Dilts, M. J., Simmons III, O. D., Likirdopulos, C. A., Krometis, L. H., & Sobsey, M. D. (2005). Microbial partitioning to settleable particles in stormwater. Water Research, 39(9), 1773–1782.
Dalla Vecchia, A., Rigotto, C., Staggemeier, R., Soliman, M. C., De Souza, F. G., Henzel, A., Santos, E. L., Do Nascimento, C. A., de Quevedo, D. M., Fleck, J. D., & Heinzelmann, L. S. (2015). Surface water quality in the Sinos River basin, in southern Brazil: tracking microbiological contamination and correlation with physicochemical parameters. Environmental Science and Pollution Research, 22(13), 9899–9911.
David, E. D., & Lindquist, W. D. (1982). Determination of the specific gravity of certain helminth eggs using sucrose density gradient centrifugation. Journal of Parasitology, 68(5), 916–919.
Dorevitch, S., Pathi, S., Huang, Y., Li, H., Michalek, A. M., Pratap, P., Wroblewski, M., Liu, L., Scheff, P. A., & Li, A. (2011). Water ingestion during water recreation. Water Research, 45, 2020–2028.
Dryden, M. W., Payne, P. A., Ridley, R., & Smith, V. (2005). Comparison of common fecal flotation techniques for the recovery of parasite eggs and oocysts. Veterinary Therapeutics, 6(1), 15–29.
Dungeni, M., Van DerMerwe, R. R., & Momba, M. N. B. (2010). Abundance of pathogenic bacteria and viral indicators in chlorinated effluents produced by four wastewater treatment plants in the Gauteng Province, South Africa. Water SA, 36(5), 607–614.
Egbueri, J. C. (2019). Water quality appraisal of selected farm provinces using integrated hydrogeochemical, multivariate statistical, and microbiological technique. Modeling Earth Systems and Environment, 5(3), 997–1013.
Fries, J. S., Characklis, G. W., & Noble, R. T. (2008). Sediment–water exchange of Vibrio sp. and fecal indicator bacteria: Implications for persistence and transport in the Neuse River estuary, North Carolina, USA. Water Research, 42(4–5), 941–950.
Fuhrimann, S., Pham-Duc, P., Cissé, G., Tram, N. T., Ha, H. T., Ngoc, P., Nguyen-Viet, H., Vuong, T. A., Utzinger, J., Schindler, C., & Winkler, M. S. (2016). Microbial contamination along the main open wastewater and storm water channel of Hanoi, Vietnam, and potential health risks for urban farmers. Science of the Total Environment, 566, 1014–1022.
Fuhrimann, S., Nauta, M., Pham-Duc, P., Tram, N. T., Nguyen-Viet, H., Utzinger, J., Cissé, G., & Winkler, M. S. (2017). Disease burden due to gastrointestinal infections among people living along the major wastewater system in Hanoi, Vietnam. Advances in Water Resources, 108, 439–449. https://doi.org/10.1016/j.advwatres.2016.12.010.
Gao, G., Falconer, R. A., & Lin, B. (2011). Numerical modelling of sediment–bacteria interaction processes in surface waters. Water Research, 45(5), 1951–1960.
Gaspard, P. G., Wiart, J., & Shwartzbrod, J. (1995). Urban sludge reuse in agriculture: waste treatment and parasitological risk. Bioresource Technology, 52, 37–40.
Gazzaz, N. M., Yusoff, M. K., Ramli, M. F., Aris, A. Z., & Juahir, H. (2012a). Characterization of spatial patterns in river water quality using chemometric pattern recognition techniques. Marine Pollution Bulletin, 64(4), 688–698.
Gazzaz, N. M., Yusoff, M. K., Aris, A. Z., Juahir, H., & Ramli, M. F. (2012b). Artificial neural network modeling of the water quality index for Kinta River (Malaysia) using water quality variables as predictors. Marine Environmental Research, 64(11), 2409–2420.
Ginebreda, A., Munoz, I., Lopez de Alda, M., Brix, R., Lopez-Doval, J., & Barcelo, D. (2010). Environmental risk assessment of pharmaceuticals in rivers: relationships between hazard indexes and aquatic macroinvertebrate diversity indexes in the Llobregat River (NE Spain). Environment International, 36(2), 153–162.
Grant, S. B., Saphores, J. D., Feldman, D. L., Hamilton, A. J., Fletcher, T. D., Cook, P. L. M., Stewardson, M., Sanders, B. F., Levin, L. A., Ambrose, R. F., Deletic, A., Brown, R., Jiang, S. C., Rosso, D., Cooper, W. J., & Marusic, I. (2012). Taking the “waste” out of “wastewater” for human water security and ecosystem sustainability. Science, 337(6095), 681–686.
Griffith, J. F., Schiff, K. C., Lyon, G. S., & Fuhrman, J. A. (2010). Microbiological water quality at non-human influenced reference beaches in southern California during wet weather. Marine Pollution Bulletin, 60, 500–508.
Hamilton, A. J., Stagnitti, F., Premier, R., Boland, A.-M., & Hale, G. (2006). Quantitative microbial risk assessment models for consumption of raw vegetables irrigated with reclaimed water. Journal of Applied and Environmental Microbiology, 72(5), 3284–3290.
Herbig, F. J., & Meissner, R. (2019). Talking dirty-effluent and sewage irreverence in South Africa: a conservation crime perspective. Cogent Social Sciences, 5(1), 1701359.
Jagals, P. (1997). Storm water runoff from typical developed and developing South African urban developments: definitely not for swimming. Water Science and Technology, 35(11–12), 133–140.
Jamieson, R., Gordon, R., Joy, D., & Lee, H. (2004). Assessing microbial pollution of rural surface waters: a review of current watershed scale modeling approaches. Agricultural Water Management, 70(1), 1–17.
Keraita, B., Konradsen, F., Drechsel, P., & Abaidoo, R. C. (2007). Effect of low-cost irrigation methods on microbial contamination of lettuce irrigated with untreated wastewater. Tropical Medicine and International Health, 12(sup 2), 15–22.
Kirschner, A. K. T., Reischer, G. H., Jakwerth, S., Savio, D., Ixenmaier, S., Toth, E., Sommer, R., Mach, R. L., Linke, R., Eiler, A., & Kolarevic, S. (2017). Multiparametric monitoring of microbial faecal pollution reveals the dominance of human contamination along the whole Danube River. Water Research, 124, 543–555.
Krometis, L.-A. H., Characklis, G. W., Simmons, O. D., Dilts, M. J., Likirdopulos, C. A., & Sobsey, M. D. (2007). Intra-storm variability in microbial partitioning and microbial loading rates. Water Research, 41(2), 506–516.
Kundu, A., Poma, H.R., Jenkins, M.W., Rajal, V. B., Wuertz, S. (2014). QMRA of intestinal nematode infection via multimedia exposure pathways. International Congress on Environmental Modelling and Software. 70. https://scholarsarchive.byu.edu/iemssconference/2014/Stream-H/70.
Kuster, M., Lopez de Alda, M. J., Hernando, M. D., Petrovic, M., Martın-Alonso, J., & Barcelo, D. (2008). Analysis and occurrence of pharmaceuticals, estrogens, progestogens and polar pesticides in sewage treatment plant effluents, river water and drinking water in the Llobregat river basin (Barcelona, Spain). Journal of Hydrology, 358, 112–123.
Mara, D. D., Sleigh, P. A., Blumenthal, U. J., & Carr, R. M. (2007). Health risks in wastewater irrigation: comparing estimates from quantitative microbial risk analyses and epidemiological studies. Journal of Water and Health, 5(1), 39–50.
Martinez, M. E. (2018). The calendar of epidemics: seasonal cycles of infectious diseases. PLoS Pathogens, 14(11), e1007327.
Mather, W., Hutchings, P., Budge, S., & Jeffrey, P. (2020). Association between water and sanitation service levels and soil-transmitted helminth infection risk factors: a cross-sectional study in rural Rwanda. Transactions of the Royal Society of Tropical Medicine and Hygiene, 114(5), 332–338.
Mayengue, P. I., Batsimba, D. K., Niama, R. F., Ottia, R. I., Malonga-Massanga, A., Fila-Fila, G. P. U., Ahombo, G., Kobawila, S. C., & Parra, H. J. (2020). Variation of prevalence of malaria, parasite density and the multiplicity of Plasmodium falciparum infection throughout the year at three different health centers in Brazzaville, Republic of Congo. BMC Infectious Diseases, 20(1), 1–10.
Mkhize-Kwitshana, Z. L., & Mabaso, M. L. H. (2014). The neglected triple disease burden and interaction of helminths, HIV and tuberculosis: An opportunity for integrated action in South Africa. South African Medical Journal, 104(4), 258–259.
Molvik, M., Helland, E., Zulu, S. G., Kleppa, E., Lillebo, K., Gundersen, S. G., Kvalsvig, J. D., Taylor, M., Kjetland, E. F., & Vennervald, B. J. (2017). Coinfection with Schistosoma haematobium and soil-transmitted helminths in rural South Africa. South African Journal of Science, 113(3/4). https://doi.org/10.17159/sajs.2017/20160251.
Msoffe, V. T. (2019). Contamination health risks of parasitic Helminths with reused wastewater for vegetable irrigation in Mbeya, Tanzania (Doctoral dissertation, Sokoine University of Agriculture).
Muirhead, R. W., Davies-Colley, R. J., Donnison, A. M., & Nagels, J. W. (2004). Faecal bacteria yields in artificial flood events: quantifying in-stream stores. Water Research, 38(5), 1215–1224.
Navarro, I., Jiménez, B., Cifuentes, E., & Lucario, S. (2008). A quantitative microbial risk assessment of helminth ova in reusing sludge for agricultural production in developing countries. WIT Transactions on Information and Communication Technologies, 39, 65–74.
Pafčo, B., Benavides, J. A., Pšenková-Profousová, I., Modrý, D., Červená, B., Shutt, K. A., Hasegawa, H., Fuh, T., Todd, A. F., & Petrželková, K. J. (2017). Do habituation, host traits and seasonality have an impact on protist and helminth infections of wild western lowland gorillas? Parasitology Research, 116(12), 3401–3410.
Pillay, R. K. (2013). An integrated study of the Isipingo River and estuary: water and sediment quality, estuary-nearshore material fluxes, anthropogenic impacts and management. M.Sc Dissertation. University of KwaZulu-Natal. South Africa.
Poulin, R. (2020). Meta-analysis of seasonal dynamics of parasite infections in aquatic ecosystems. International Journal for Parasitology, 50(6–7), 501–510.
Qadir, A., Malik, R. N., & Husain, S. Z. (2008). Spatio-temporal variations in water quality of Nullah Aik-tributary of the river Chenab, Pakistan. Environmental Monitoring and Assessment, 140(1–3), 43–59.
Ribas, A., Jollivet, C., Morand, S., Thongmalayvong, B., Somphavong, S., Siew, C. C., Ting, P. J., Suputtamongkol, S., Saensombath, V., Sanguankiat, S., & Tan, B. H. (2017). Intestinal parasitic infections and environmental water contamination in a rural village of northern Lao PDR. The Korean Journal of Parasitology, 55(5), 523–532.
Sakaji, R. H., & Funamizu, N. (1998). Microbial risk assessment and its role in the development of wastewater reclamation policy. In T. Asano (Ed.), Wastewater reclamation and reuse (Vol. 10, pp. 705–756). Boca Raton: CRC Press.
Sant’Ana, A. S., Franco, B. D. G. M., & Schaffner, D. W. (2014). Risk of infection with Salmonella and listeria monocytogenes due to consumption of ready-to-eat leafy vegetables in Brazil. Food Control, 42, 1–8.
Seidu, R., Heistad, A., Amoah, P., Drechsel, P., Jenssen, P. D., & Stenström, T. A. (2008). Quantification of the health risk associated with wastewater reuse in Accra, Ghana: a contribution toward local guidelines. Journal of Water and Health, 6(4), 461–471.
Semwal, N., & Akolkar, P. (2006). Water quality assessment of sacred Himalayan rivers of Uttaranchal. Current Science, 91(4), 486–496.
Stolk, W. A., Kulik, M. C., le Rutte, E. A., Jacobson, J., Richardus, J. H., & de Vlasm, S. J. (2016). Between-country inequalities in the neglected tropical disease burden in 1990 and 2010, with projections for 2020. PLoS Neglected Tropical Diseases, 10(5), e0004560. https://doi.org/10.1371/journal.pntd.0004560.
Vijay, R., Khobragade, P., & Mohapatra, P. K. (2011). Assessment of groundwater quality in Puri City, India: an impact of anthropogenic activities. Environmental Monitoring and Assessment, 177, 409–418. https://doi.org/10.1007/s10661-010-1643-9.
Waller, P. J., Rudby-Martin, L., Ljungström, B. L., & Rydzik, A. (2004). The epidemiology of abomasal nematodes of sheep in Sweden, with particular reference to over-winter survival strategies. Veterinary Parasitology, 122(3), 207–220.
Wang, Y., Wang, P., Bai, Y., Tian, Z., Li, J., Shao, X., Mustavich, L. F., & Li, B.-L. (2013). Assessment of surface water quality via multivariate statistical techniques: a case study of the Songhua River Harbin region, China. Journal of Hydro-Environmental Research, 7(1), 30–40.
Westrell, T. (2004). Microbial risk assessment and its implications for risk management in urban water systems. PhD Thesis, Department of Water and Environmental Studies, Linköpings University, Sweden.
WHO-World Health Organization. (2006). Guidelines for the safe use of wastewater, excreta and greywater (Vol. 4). Geneva: World Health Organization.
WHO-World Health Organization. (2014). Neglected tropical diseases: PCT databank: soil transmitted helminthiases [database on the Internet]. c2014. Available on 7th November, 2019 from: http://www.who.int/neglected_diseases/preventive_chemotherapy/sth/en/.
Zdybel, J., Cencek, T., Karamon, J., & Kłapec, T. (2015). Effectiveness of selected stages of waste water treatment in elimination of eggs of intestinal parasites. Journal of Veterinary Research, 59, 51–57.
Zhang, Y., Dudgeon, D., Cheng, D., Thoe, W., Fok, L., Wang, Z., & Lee, J. (2010). Impacts of land use and water quality on macroinvertebrate communities in the Pearl River drainage basin, China. Hydrobiologia, 652(1), 71–88.
Zhou, T., Wu, J., & Peng, S. (2012). Assessing the effects of landscape pattern on river water quality at multiple scales: a case study of the Dongjiang River watershed, China. Ecological Indicators, 23, 166–175.
Acknowledgments
We are grateful to the Bill and Melinda Gates foundation (Grant Number: OPP1122681), the South African Research Chair Initiative (SARChI) of the Department of Science and Technology and the National Research Foundation of South Africa for funding the research. We also acknowledge the support of the Institute for Water and Wastewater Technology of the Durban University of Technology.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Amoah, I.D., Kumari, S., Reddy, P. et al. Impact of informal settlements and wastewater treatment plants on helminth egg contamination of urban rivers and risks associated with exposure. Environ Monit Assess 192, 713 (2020). https://doi.org/10.1007/s10661-020-08660-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-020-08660-0