Abstract
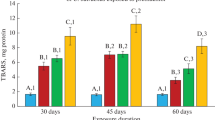
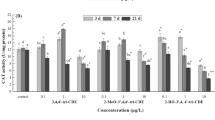
The toxicological evaluations of crude oil, xylene, toluene and benzene were carried out against juvenile stages of Clarias gariepinus. On the basis of LC50 value, benzene (0.017 ml/l) was found to be the most toxic followed by xylene (0.086 ml/l), toluene (0.398 ml/l) and crude oil (2.219 ml/l) was the least toxic. The results of the lipid peroxidation assay showed that the level of malonaldehyde (MDA) in liver and gills of fish exposed to all the test chemicals increased significantly (P < 0.05) when compared to control. Measurement of activities of antioxidant enzymes, superoxide dismutase (SOD), catalase (CAT) and glutathione-S-transferase (GST) in liver and gill of fishes exposed to sublethal concentration of the test chemicals over a 28-day period was found to decrease significantly (P < 0.05) when compared to control. The observed reduction in the activities of antioxidant defense enzymes such as SOD, CAT and GST in conjunction with an increase in MDA levels in the gill and liver tissues of test animals exposed to aromatic hydrocarbon compounds were recommended as a good battery of biomarkers for early detection of pollution during biomonitoring programmes.
Similar content being viewed by others
References
Achuba, F. I., & Osakwe, S. A. (2003). Petroleum-induced free radical toxicity in African catfish (Clarias gariepinus). Fish physiology and Biochemistry, 29, 97–103.
Avci, A., Kacmaz, M., & Durak, I. (2005). Peroxidation in muscle and liver tissues from fish in a contaminated river due to a petroleum refinery industry. Ecotoxicology and Environmental Safety, 60, 101–105.
Bocchettii, R., Fattorini, D., Gambi, M. C., & Regoli, F. (2004). Trace metal concentrations and susceptibility to oxidative stress in the polychaete Sabella spallanzanii (Gmelin) (Sabellidae): Potential role of antioxidants in revealing stressful environmental conditions in the Mediterranean. Archives of Environmental Contamination and Toxicology, 46, 353–361.
Brown, P. J., Long, S. M., Spurgeon, D. J., Svendsen, C., & Hankard, P. K. (2004). Toxicological and biochemical responses of the earthworm Lumbricus rubellus to pyrene, a non-carcinogenic polycyclic aromatic hydrocarbon. Chemosphere, 57, 1675–1681.
Calvert, J. G., Atkinson, R., Becker, K. H., Kamens, R. M., Seinfeld, J. H., Wallington, T. J., et al. (2000). The mechanism of atmospheric oxidation of aromatic hydrocarbons. New York: Oxford University Press.
Catala, A. (2009). Lipid peroxidation of membrane phospholipids generates hydroxyl-alkenals and oxidized phospholipids active in physiological and/or pathological conditions. Chemistry and Physics of Lipids, 157, 1–11.
Cheung, C. C. C., Zheng, G. J., Li, A. M. Y., Richardson, B. J., & Lam, P. K. S. (2001). Relationship between tissue concentrations of polycyclic aromatic hydrocarbons and antioxidative responses of marine mussels, Perna viridis. Aquatic Toxicology, 52, 189–203.
Cohen, G., Dembiec, D., & Marcus, J. (1970). Measurement of catalase activity in tissue extracts. Analytical Biochemistry, 34, 30–38.
Dorval, J., Leblond, V. S., & Hontela, A. (2003). Oxidative stress and loss of cortisol secretion in adrenocortical cells of rainbow trout (Oncorhynchus mykiss) exposed in vitro to endosulfan, an organochlorine pesticide. Aquatic Toxicology, 63, 229–241.
Fatima, R. A., & Ahmad, M. (2005). Certain antioxidant enzymes of Allium cepa as biomarkers for the detection of toxic heavy metals in wastewater. Science of the Total Environment, 346, 256–273.
Finney, D. Y. (1971). Probit analysis (3rd ed.). London: Cambridge University Press.
Fournier, D., Bride, J. M., Poirie, M., Berge, J. B., & Plapp, F. W. (1992). Insect glutathione-S-transferase: Biochemical characteristics of the major forms of houseflies susceptible and resistant to insecticides. Journal of Biological Chemistry, 267, 1840–1845.
Gorinstein, S., Moncheva, S., Katrich, E., Toledo, F., Arancibia, P., Goshev, I., et al. (2003). Antioxidants in the black messel (Mytilus galloprovincialis) as an indicator of black sea coastal pollution. Marine Pollution Bulletin, 46, 1317–1325.
Habig, W. H., & Jakoby, W. B. (1981). Bioassays for differentiation of glutathione-S-transferases. Methods in Enzymology, 77, 398–405.
Hamed, R. R., Elawa, S. E., & Farid, N. M. (1999). Evaluation of detoxification enzyme levels in Egyptian catfish, Clarias lazera, exposed to dimethoate. Bulletin of Environmental Contamination and Toxicology, 63, 789–796.
Hermes-Lima, M., Willmore, W. G., & Storey, K. B. (1995). Quantification of lipid peroxidation in tissue extracts based on Fe (III) xylenol orange complex formation. Free Radical Biology &Medicine, 19, 271–280.
Irwing, R. J. (1971). Environmental contaminants encyclopedia entry for BTEX and BTEX compounds (p. 36). Colorado: National Park Service.
Jifa, W., Zhiming, Y., Xiuxian, S., & You, W. (2006). Response of integrated biomarkers of fish (Lateolabrax japonicus) exposed to benzo(a)pyrene and sodium dodecylbenzene sulfonate. Ecotoxicology and Environmental Safety, 65, 230–236.
Kelly, S. A., Havrilla, C. M., Brady, T. C., Abramo, K. H., & Levin, E. D. (1998). Oxidative stress in toxicology: Established mammalian and emerging piscine model systems. Environmental Health Perspectives, 106, 375–384.
Leaver, M. J., & George, S. G. (1998). A piscine glutathione-S-transferase which efficiently conjugates the end-products of lipid peroxidation. Marine Environmental Research, 46, 71–74.
Manduzio, H., Monsinjon, T., Rocher, B., Leboulenger, F., & Galap, C. (2003). Characterisation of an inducible isoform of the Cu/Zn superoxide dismutase in the blue mussel Mytilus edulis. Aquatic Toxicology, 64, 73–83.
Oruc, E. O., Sevgiler, Y., & Ulner, N. (2004). Tissue-specific oxidative stress responses responses in fish exposed to 2,4-D and azinphosmethyl. Comparative Biochemistry and Physiology C, 137, 43–51.
Regoli, F., Gorbi, S., Frenzilli, G., Nigro, M., Corsi, I., Focardi, S., & Winston, G. W. (2002). Oxidative stress in ecotoxicology: From the analysis of individual antioxidants to a more integrated approach. Marine Environmental Research, 54(3–5), 419–423.
Srivastava, S. K., Hu, X., Xia, H., Awasthi, S., Amin, S., & Singh, S. V. (1999). Metabolic fate of glutathione conjugate of benzo(a)pyrene-(7R, 8S)-diol(9S, 10R)-epoxide in human liver. Archives of Biochemistry and Biophysics, 371, 340–344.
Stohs, S. J., Bagchi, D., Hassoun, E., & Bagchi, M. (2001). Oxidative mechanisms in the toxicity of chromium and cadmium ions. Journal of Environmental Pathology, Toxicology and Oncology, 19, 201–213.
Storey, K. B. (1996). Oxidative stress: Animal adaptations in nature. Brazilian. Journal of Medicine and Biological Research, 29, 1715–1733.
Sun, M., & Zigman, S. (1978). An improved spectrophotometric assay for superoxide dismutase based on epinephrine autoxidation. Analytical Biochemistry, 90(1), 81–89.
Valavanidis, A., Vlahogianni, T., Dassenakis, M., & Scoullos, M. (2006). Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicology and Environmental Safety, 64, 178–189.
Wu, R. S. S., Siu, W. H. L., & Shin, P. K. S. (2005). Induction, adaptation and recovery of biological responses: Implications for environmental monitoring. Marine Pollution Bulletin, 51, 623–634.
Yagi, K. (1998). Simple procedure for specific enzyme of lipid hydroperoxides in serum or plasma. Methods in Molecular Biology, 108, 107–110.
Zhang, J. F., Liu, H., Sun, Y. Y., Wang, X. R., Wu, J. C., & Xue, Y. Q. (2005). Responses of the antioxidants defenses of the goldfish, Carasisius auratus, exposed to 2,4-dichlorophenol. Environmental Toxicology Pharmacology, 19, 185–190.
Zielinski, S., & Portner, H.-O. (2000). Oxidative stress and antioxidative defense in cephalopods: A function of metabolic rate or age? Comparative Biochemistry and Physiology B, 125, 147–160.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Otitoloju, A., Olagoke, O. Lipid peroxidation and antioxidant defense enzymes in Clarias gariepinus as useful biomarkers for monitoring exposure to polycyclic aromatic hydrocarbons. Environ Monit Assess 182, 205–213 (2011). https://doi.org/10.1007/s10661-010-1870-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10661-010-1870-0