Abstract
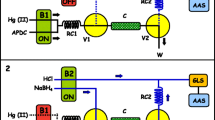
Amberlite XAD-4 resin functionalized with β-nitroso-α-naphthol was applied to an online system for the preconcentration and determination of uranium. U (VI) ions were retained on the minicolumn at an appropriate pH and then desorbed with acid solution. The amount of uranium in the eluate was measured spectrophotometrically at 650 nm using Arsenazo III as a colorimetric reagent. The limit of detection and the preconcentration factor were 1.8 μg L − 1 and 10, respectively. The chemical and flow variables affecting the preconcentration were studied. The influence of several ions on the system was also investigated. The method was successfully applied for the evaluation of uranium in water and in effluent samples.
Similar content being viewed by others
References
Ayata, S., Kaynak, I., & Merdivan, M. (2009). Solid phase extractive preconcentration of silver from aqueous samples. Environmental Monitoring and Assessment, 153, 333–338.
Bezerra, M. A., Arruda, M. A. Z., & Ferreira, S. L. C. (2005). Cloud point extraction as procedure of separation and preconcentration for metal determination using spectroanalytical techniques: A review. Applied Spectroscopy Reviews, 40, 269–299.
Bozkurt, S. S., & Merdivan, M. (2009). Solid phase extraction of gold(III) on silica gel modified with benzoylthiourea prior to its determination by flame atomic absorption spectrometry. Environmental Monitoring and Assessment, 158, 15–21.
Camel, V. (2003). Solid phase extraction of trace elements. Spectrochimica Acta Part B, 58, 1177–1233.
Demirel, N., Merdivan, M., Pirinccioglu, N., & Hamamci, C. (2003). Thorium(IV) and uranium(VI) sorption studies on octacarboxymethyl-C-methylcalix[4]resorcinarene impregnated on a polymeric support. Analytica Chimica Acta, 485, 213–219.
Fang, Z.-L. (1993). Flow injection separation and preconcentration. New York: Wiley.
Ferreira, S. L. C., Bezerra, M. A., Santos, W. N. L., & Neto, B. B. (2003). Application of Doehlert designs for optimisation of an on-line preconcentration system for copper determination by flame atomic absorption spectrometry. Talanta, 61, 293–303.
Gama, E. M., Lima, A. S., & Lemos, V. A. (2006). Preconcentration system for cadmium and lead determination in environmental samples using polyurethane foam/Me-BTANC. Journal of Hazardous Materials, 136, 757–762.
Gladis, J. M., & Rao, T. P. (2002). Quinoline-8-ol-immobilized Amberlite XAD-4: Synthesis, characterization, and uranyl ion uptake properties suitable for analytical applications. Analytical and Bioanalytical Chemistry, 373, 867–872.
Jain, V. K., Handa, A., Sait, S. S., Shrivastav, P., & Agrawal, Y. K. (2001). Preconcentration, separation and trace determination of lanthanum(III), cerium(III), thorium(IV) and uranium(VI) on polymer supported o-vanillinsemicarbazone. Analytica Chimica Acta, 429, 237–246.
Kumar, M., Rathore, D. P. S., & Singh, A. K. (2001). Pyrogallol immobilized amberlite XAD-2: A newly designed collector for enrichment of metal ions prior to their determination by flame atomic absorption spectrometry. Microchimica Acta, 137, 127–134.
Lemos, V. A., Gama, E. M., & Lima, A. S. (2006). On-line preconcentration and determination of cadmium, cobalt and nickel in food samples by flame atomic absorption spectrometry using a new functionalized resin. Microchim Acta, 153, 179–186.
Lemos, V. A., Novaes, G. S., Carvalho, A. L., Gama, E. M., & Santos, A. G. (2009). Determination of copper in biological samples by flame atomic absorption spectrometry after precipitation with Me-BTAP. Environmental Monitoring and Assessment, 148, 245–253.
Lemos, V. A., Santos, E. S., & Gama, E. M. (2007). A comparative study of two sorbents for copper in a flow injection preconcentration system. Separation and Purification Technology, 56, 212–219.
Marczenko, Z. (1976). Spectrophotometric determination of elements. New York: Wiley.
Metilda, P., Sanghamitra, K., Gladis, J. M., Naidu, G. R. K., & Rao, T. P. (2005). Amberlite XAD-4 functionalized with succinic acid for the solid phase extractive preconcentration and separation of uranium(VI). Talanta, 65, 192–200.
Prabhakaran, D., & Subramanian, M. S. (2004). Selective extraction of U (VI), Th (IV), and La(III) from acidic matrix solutions and environmental samples using chemically modified Amberlite XAD-16 resin. Analytical and Bioanalytical Chemistry, 379, 519–525.
Skoog, D. A., West, D. M., & Holler, F. J. (1996). Fundamentals of analytical chemistry. Orlando: Saunders College Publishing.
Soylak, M., Narin, I., Bezerra, M. A., & Ferreira, S. L. C. (2005). Factorial design in the optimization of preconcentration procedure for lead determination by FAAS. Talanta, 65, 895–899.
Tewari, P. J., & Singh, A. K. (2000). Thiosalicylic acid-immobilized Amberlite XAD-2: Metal sorption behaviour and applications in estimation of metal ions by flame atomic absorption spectrometry. Analyst, 125, 2350–2355.
Tokalıoglu, S., Yılmaz, V., & Kartal, S. (2009). Solid phase extraction of Cu(II), Ni(II), Pb(II), Cd(II) and Mn(II) ions with 1-(2-thiazolylazo)-2-naphthol loaded Amberlite XAD-1180. Environmental Monitoring and Assessment, 152, 369–377.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lemos, V.A., Gama, E.M. An online preconcentration system for the determination of uranium in water and effluent samples. Environ Monit Assess 171, 163–169 (2010). https://doi.org/10.1007/s10661-009-1268-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10661-009-1268-z