Abstract
Cork oak (Quercus suber) is one of the most important forest species in Portugal and in the Mediterranean basin, but in recent decades a progressive decline has been registered in the montado ecosystem. The main phytopathogenic agent involved in this process, Phytophthora cinnamomi, has a high infection potential and survival ability, and became a serious threat to this ecosystem. Extensive areas of cork oak are infected in Portugal, being imperative to find new ways to control this disease. The complex lifecycle of this pathogen and the small number of effective agrochemicals currently authorized against Phytophthora spp., with phosphonates taking advantage, justifies this work where new products with antagonism against Oomycetes or biostimulant properties were tested. This study aimed to evaluate the efficacy of BLAD (a polypeptide with antifungal, anti-oomycete and biostimulant properties), potassium phosphonate and salicylic acid in Q. suber infected with P. cinnamomi, quantifying the severity of root lesions. BLAD, potassium phosphonate and salicylic acid treatments showed lower root lesion severities than the inoculated control, and proved to be effective as they provided a slower disease evolution. The lowest lesion severity in cork oak roots observed with BLAD treatments allowed us to consider BLAD as the most promising product among the three we tested.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cork oak (Quercus suber L.) has a restricted geographic distribution, established only in the Mediterranean basin. Some economic activities such as cork production, livestock grazing and many ecosystems services are strictly connected with this species contributing to soil conservation and acting as a fire barrier (Aronson et al., 2010). Due to its environmental and economic importance in the Mediterranean basin, mainly in Portugal, where over one third of the cork oak world area is located (736,000 ha), Q. suber is one of the three forest species protected by Portuguese law (APCOR, 2020).
Increasing Q. suber mortality has been reported in Portugal during the last decades, with the affected trees showing a progressive decline, exhibiting symptoms of water and nutrient deficit, which have been attributed to a combination of factors, such as deficient soil drainage, drought, insects and pathogenic agents (Camilo-Alves et al., 2013). However, many authors have associated cork oak decline to P. cinnamomi Rands, considered as the utmost factor for this oak decline symptomatology in southern Europe, namely the Iberian Peninsula (Brasier, 1996; Brasier et al., 1993; Duque-Lazo et al., 2018) and Italy (Seddaiu et al., 2020).
Phytophthora cinnamomi is, as Phytophthora species in general, an important plant destructive pathogen, surviving for long periods of time in soil, even in the absence of hosts, affecting parts of plants in contact with the soil, and inducing root destruction (Erwin & Ribeiro, 1996; Hardham & Blackman, 2018). Fine root rotting is the primary symptom conducting to the consequent disturbance in water and nutrients absorption and loss of vigour (appearing of yellowing leaves and leaf fall) (Hardham, 2005). In cork oak trees other symptomatology less frequent can be observed such as exudations on trunks and young shoots dieback (Brasier et al., 1993). Life cycle includes exponential spread enhanced by free water in the soil, high infection rates and a high ability of survival. All these features make disease control a complex goal to achieve (Camilo-Alves et al., 2013; Erwin & Ribeiro, 1996; Hardham & Blackman, 2018). Therefore as there are extensive cork oak forest areas infected by P. cinnamomi it is imperative to establish control strategies regarding root system health.
Fungicides from the phenylamides group (e.g. metalaxyl) and from the phosphonates group (e.g. potassium phosphonate and fosetyl-Al) have been tested against Phytophthora diseases (Fernandez-Escobar et al., 1999; Rosário et al., 2021; Solla et al., 2021). However, resistance development in some Phytophthora species has been noticed, conditioning phenylamides efficacy (Drenth & Guest, 2004; Erwin & Ribeiro, 1996; FRAC, 2018). Despite the phosphonates group proven efficacy against P. cinnamomi (Fernández-Escobar et al., 1999; Gentile et al., 2009; Romero et al., 2019; González et al., 2020) and its unreported toxicity or lack of induced resistance mechanisms, any plant diseases control strategy should be based on eco-friendly and effective substances, long-lasting and easily accessible. Natural products, such as plants or fungal extracts or plant-based components, may be an optional source of new agrochemicals for plant diseases control (Kuberan et al., 2012; Nashwa & Abo-Elyousr, 2012; Sivanandhan et al., 2017). For all these reasons, and considering that the main goal is P. cinnamomi control, we tested a new product named BLAD, a multifunctional polypeptide with chelating properties and with multiple effects on fungal cell walls (FRAC, 2018), exhibiting antifungal and biostimulant properties (Monteiro et al., 2010), that have been showing optimistic results in relation to many fungi, exhibiting equal or greater efficacy than the best fungicides currently available (Monteiro et al., 2015). Salicylic acid was also tested due to its effect on defence responses in plants (An & Mou, 2011; Popova, 2013). Potassium phosphonate was used as a reference since its efficacy has already been demonstrated in Quercus spp. and Castanea sativa Mill. (Fernández-Escobar et al., 1999; González et al., 2017; Solla et al., 2021; Brandano et al., 2023).
This study aims to evaluate the efficiency of a new substance (BLAD) through the quantification of root lesion severity in young Q. suber seedlings infected by P. cinnamomi. In addition, we analysed the impact of infection in plant dry weight as well as in root tissues via histological analysis.
Materials and methods
Plant material
In this experiment, 180 cork oak seedlings from acorns with Portuguese origin obtained from a commercial nursery were used. The plants were asymptomatic and tested negative for Phytophthora spp. detection using a baiting technique adapted from Jung et al. (1996). Roots and soil from five randomly selected seedlings were flooded in plastic boxes with approximately 500 mL of distilled water, and small leaf pieces of Persea americana, and Eucalyptus globulus were used as baits. Boxes were kept at approximately 25 °C under natural light. Baits were checked under the microscope for the presence of sporangia.
Eleven months after sowing (October 2017), the plants were transplanted into 10 L squared pots (four plants per pot) filled with peat. Plants were maintained in a greenhouse under a mean temperature of 16.7 °C (minimum 7.7 °C and maximum 33.6 °C), and hand watered weekly according to their water needs.
Phytophthora cinnamomi inoculation
Phytophthora cinnamomi isolate PH1247 was collected in 2012 from a cork oak stand in Montemor-o-Novo, Portugal, and maintained in INIAV’s collection (Oeiras, Portugal). This isolate was selected based on preliminary aggressiveness tests with Trifolium subterraneum and Q. suber excised shoots (Bairrão et al., 2021).
Inoculum was prepared using vermiculite humidified with V8 medium (0.5 L medium per L of mixture). The mixture was autoclaved for 20 min at 120 °C/ 1 bar, in two successive days. Then, 12-day-old cultures of isolate PH1247 in potato dextrose agar (PDA) were cut in 0.5 cm plugs, and mixed with the vermiculite, using half of a Petri dish per litre. The final mixture was incubated at 24 °C ± 2 °C for three weeks.
Two months after transplantation, cork oak seedlings were inoculated by placing the inoculum (30 mL mixture per L of soil) in the center of each pot. Twenty-four hours of flooding per week were also provided after inoculation until the treatments application. Watering was suspended during the week of treatments application.
Treatments and experimental design
The experimental design included 5 modalities with 9 pots (36 plants) each: plants inoculated with P. cinnamomi and treated with BLAD (BL), potassium phosphonate (PP) or salicylic acid (SA) plus 2 non-treated controls, non-inoculated (NC) and inoculated (PC) plants. In treated modalities two applications were performed, with the first application 60 days after inoculation (DAI) with P. cinnamomi and the second 90 DAI (Table 1).
Eight mL of each final solution – BLAD (Fracture TM, 5 gL−1), potassium phosphonate (Alexin 75 LS, 2.42 gL−1) and salicylic acid (0.5 mM) – were sprayed on cork oak plants as foliar application (the 4 plants per pot were treated simultaneously). Control, inoculated and non-inoculated plants, were sprayed with water.
Observation of symptoms
Typical aerial symptoms of Phytophthora infection, such as yellowing and leaf fall or collar lesions, were checked during the experiment.
In order to quantify the severity of root lesions two observations were performed, 1 and 2 months after the end of treatments (Table 1). During the first observation, 4 pots from each modality were randomly selected. The remaining 5 pots per modality were processed in the second observation. The roots of the 4 plants per pot were carefully separated, washed in tap water and transferred to laboratory, where percentage of lesion was attributed. The roots were spared over a white support (only identified by numbers) and three independent raters attributed a root lesion percentage. The percentage takes in consideration the number of secondary roots with necrosis and the total number of secondary roots.
Re-isolations and histopathology
Re-isolations were performed from roots and stem samples of plants in order to check the presence of infection by P. cinnamomi. After the attribution of root lesion percentage, 3 plants per modality (with the highest percentage of lesions) were chosen and 5 mm long fragments of roots (10 per plant) and 1 cylinder at collar level (1 per plant) were cut with sterile scalpel. All the selected plant material was washed in sterilized water and plated on a selective medium for Phytophthora spp., the PARPNH medium (Jeffers & Martin, 1986). After 2–5 days of incubation at 24 °C in darkness, Petri dishes were observed and confirmation was achieved through visualization of typical mycelium of this species. Hyphae were subcultured onto PDA medium at 24 °C for further examinations and Phytophthora presence was confirmed through the visualization of its characteristic hyphal swellings and rosaceous colony shape (Eggers et al., 2012).
Root portions were fixed for ± 48 h, using a fixative (FAA) containing 5% formaldehyde, 5% glacial acetic acid, and 90% ethanol (70%) and dehydration was done through a progressive ethanol series. This material was clarified using Histo-Clear and then embedded in paraffin. The embedded samples were sectioned with a Leika RM2255 rotary microtome using a 10 μm thickness and transverse sections were stained with AstraBlue. Histological sections were observed under an Olympus BX51 microscope and hyphae and chlamydospores were sought out. Image acquisition was performed with Olympus DP-Soft software.
Roots and aerial part dry weights
After the previously described steps, roots were separated from their respective aerial parts and all the material was stored at -18 ºC, avoiding weight loss due to respiration and, subsequently, were transferred into a 60 ºC oven to dry. After 4–5 days roots and aerial part dry weights were determined.
Statistical analysis
Experiment data regarding the percentage of root lesions severity and dry weights were subjected to a two-way ANOVA (treatment and sampling) and a post-hoc test (Tukey’s test) was done for multiple comparisons of means. Data were analysed in R (R Core Team, 2022) with RStudio software (RStudio Team, 2022) and the level of significance applied was always 95% (p < 0.05).
Results
Root lesions severity
No plant mortality was observed during the experiment and regarding possible symptoms in the aerial parts, no visually evident differences were detected among the five modalities at 150 DAI. However, some yellowing and leaf fall in all modalities could be attributed to successive Aphis spp. attacks.
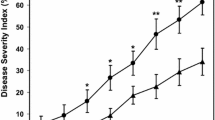
At the first observation (120 DAI) we could already check lesions in the roots. There were no differences among samplings (120 and 150 DAI) as the severity levels of root lesions remained relatively constant (Table 2), but there were differences among treatments (Fig. 1). Considering the mean of the two sampling dates, the non-inoculated control (NC) exhibited the lowest value of root lesions severity (10%), an expected result.
Visual estimation: average root lesions severity of cork oak seedlings infected with P. cinnamomi, according to NC (non-inoculated control), PC (P. cinnamomi inoculated control), BL (BLAD), SA (salicylic acid) and PP (potassium phosphonate) modalities and respective standard deviation (n = 9). The same letter means that the treatments are not significantly different using Tukey test (p < 0.05)
No external lesions were observed in stems throughout the experiment. Only two plants from inoculated control modality (PC) exhibited internal lesions along the stem at 150DAI and affected only 10% of the stem circumference at collar level.
Non-inoculated control (NC) differs significantly from positive control (PC).
Regarding the effect of the applied products, all treatments showed statistically lower values for the necrotic roots percentage than untreated plants (PC—29% severity), showing their effectiveness in controlling the disease. BLAD (BL) is the only treatment showing a percentage of root lesion that does not significantly differ from non-inoculated plants (NC) (Table 2). Also, BL differs from PP treatment, but not from SA and there are no differences between SA and PP treatments (Fig. 1).
Dry weights
Regarding the root dry weights, there were no significant differences between samplings and treatments. The NC had the highest dry weight value, with all inoculated modalities having lower values (Table 3).
Concerning the aerial part, there were no significant differences between samplings and treatments. On 1st sampling (120 DAI) PC showed the higher dry weight, but on 2nd sampling (150 DAI) NC was the modality with the highest dry weight (Table 3).
Reisolations and histopathology
Reisolations were possible in all the inoculated modalities and confirmed with the histological sections since the characteristic hyphae of P. cinnamomi had already been detected in the central cylinder of the roots. On the neighbouring of these structures, there was cell degradation (Fig. 2a) and P. cinnamomi even led to the death of colonized cells (Fig. 2b) (red-coloured cells). With Astrablue stain, living cells are coloured in a blue shade, while dead cells become red.
According to Fig. 3a the non-inoculated and symptomless root structure was practically intact, as there was apparently no degradation. On the other hand, in the roots with symptoms (Fig. 3b, c, d, e) cells were strongly degraded. In symptomless roots of non-inoculated plant, live (blue) cells are presented in both central cylinder and cortex (Fig. 3a). In the roots of plants treated with BLAD (BL) (modality with the best results), although the cells of cortex were dead (red), the cells of the central cylinder remained alive (blue) (Fig. 3b). In the structure of roots treated with salicylic acid (SA) and potassium phosphonate (PP), both the cortex and central cylinder cells were dead (red) (Fig. 3c, d) as well as in the plants inoculated with P. cinnamomi and non-treated (Fig. 3e).
General view of cross sections of cork oak roots: a—symptomless root from a non-inoculated plant (NC); b—root with symptoms from an inoculated plant treated with BLAD (BL); c—roots with symptoms from inoculated plants treated with salicylic acid (SA); d—roots with symptoms from inoculated plants treated with potassium phosphonate (PP); e—roots with symptoms from inoculated plants (PC)
Discussion
In this experiment a new product (BL) with antagonistic properties against Oomycetes or biostimulant properties, salicylic acid (SA) and potassium phosphonate (PP) were evaluated in order to control P. cinnamomi in Quercus suber. The efficacy of these substances was evaluated through the quantification of the severity of root lesions. Roots and foliage dry weights were also analysed.
Considering the evolution of symptoms in the aerial part, there were no important visual differences between non-inoculated control and the inoculated modalities. Similar results were found by Sánchez et al. (2002) verifying low foliar symptom levels and no differences between non-inoculated and inoculated modalities. Moreira et al. (2000) also reported that symptoms of decline in cork oak were only detected 9 months after inoculation, in 6 months old plants. This slow evolution may have also led to a weak effect in dry weights. Thus, although some of the tested products are known to stimulate plant growth, such as potassium phosphonate (Fernandez-Escobar et al., 1999; WIPO, 2007) and BLAD (Monteiro et al., 2015), this effect was not possible to confirm during our study. We can suggest, as a possible reason, the short time lapse between applications and sampling (2 and 3 months since the first application).
Concerning the visual estimation of lesions severity in roots, non-inoculated (NC) plants had values around 10% which may be related to the waterlogging cycles. These values are in line with the observations of Sánchez et al. (2005) reporting 1–33% of necrotic roots in control plants subjected to different flooding regimes.
Five months after inoculation (150 DAI), the plants inoculated with P. cinnamomi (PC) had an average of 29% of roots with lesions, which reflects the slow development of the disease in cork oak trees, as reported by Moreira et al. (2000). These authors emphasise the slow development of symptoms in this species, even under favourable conditions to infection, such as long term of soil saturation. Other similar studies, with observations at 3 and 6 months after inoculation, reported higher root lesion values comparing with our study (Sánchez et al., 2002, 2005).
All the treatments tested during this experiment, BLAD (BL), salicylic acid (SA) and potassium phosphonate (PP) were effective, as they presented lesions severity significantly lower than the inoculated control (PC).
Salicylic acid corresponds to one of the systemic acquired resistances (SAR) signals, leading to an increase of plants resistance to pathogens attack after an initial infection. High SA levels turn organism’s survival more difficult in plant tissues (An & Mou, 2011; Popova, 2013). Thereby, salicylic acid may have stimulated the cork oak defence response, leading to a level of root lesion severity lower than that of inoculated control (PC). Additionally, Amborabé et al. (2002), when studying SA effect in Eutypa lata, point out that it has fungicidal properties, having a direct effect in E. lata development by altering compartments of the fungal cell.
Potassium phosphonate results may be related to an indirect action, since it stimulates host defence responses and to a direct inhibition of the pathogen growth (Drenth & Guest, 2004; Eshraghi et al., 2011; FRAC, 2018; Silva et al., 2015). Several authors have emphasised that its action is mainly preventive (Gentile et al., 2009; Groves et al., 2015; Silva et al., 2015) and, recent works demonstrated the preventive and curative effect of fosetyl-Al used in trunk injections on cork and holm oaks (González et al., 2020; Romero et al., 2019) or potassium phosphite applied as a foliar or trunk spray in Q. rubra and Q. ilex (Solla et al., 2021).
Gentile et al. (2009) mentioned that potassium phosphonate application on chestnut trees infected with Phytophthora spp. induced plant defences that lead to pathogen confinement through localised deposition of occlusive and protective compounds while studies of Groves et al. (2015) observed the beneficial combination of phosphite and salicylic acid, stimulating the accumulation of salicylic acid in Lupinus angustifolius and controlling P. cinnamomi. On the other hand, Eshraghi et al. (2011) suggested that their close results of root lesion severity in treatments with salicylic acid and potassium phosphonate may be related to the fact that, with phosphonate foliar application, salicylic acid pathway may be activated, as they observed in Arabidopsis thaliana.
In this work BLAD (BL) was tested for the first time in an experimental trial with P. cinnamomi and proved to be the most effective treatment, once plants treated with BLAD had the closest root lesions severity to non-inoculated plants (NC), not differing significantly from this modality.
BLAD´s mode of action is still not clarified but it is known as a non-systemic product so it is unlikely that an interaction between the product and the oomycete, active on roots, has occurred. BLAD may have led to a growth rate increase (WIPO, 2007), which could have induced new roots growth, leading to a lower root lesion severity and to the absence of differences between modalities. It seems most likely that BLAD may enhance or even stimulate plants defence responses. This side effect enhancing plant’s capacity to defend itself against pathogens has already been registered in other contact fungicides, namely fludioxonil and pyrimethanil, by Saladin et al. (2003). The same authors mention studies reporting that fungicides may enhance plant defences through cell wall lignification, enzymes stimulation or synthesis of phytoalexin and phenolic compounds.
Considering the promising results obtained with the treatment with BLAD, the use of this non-conventional product, may alter P. cinnamomi control prospects on cork oak. To continue this work new tests will be carried out in nursery, keeping the plants for longer periods, and BLAD will be tested in a field experiment.
References
Amborabé, B. E., Fleurat-Lessard, P., Chollet, J. F., & Roblin, G. (2002). Antifungal effects of salicylic acid and other benzoic acid derivatives towards Eutypa lata: Structure–activity relationship. Plant Physiology and Biochemistry, 40, 1051–1060. https://doi.org/10.1016/S0981-9428(02)01470-5
An, C., & Mou, Z. (2011). Salicylic acid and its function in plant immunity. Journal of Integrative Plant Biology, 53(6), 412–428. https://doi.org/10.1111/j.1744-7909.2011.01043.x
APCOR. (2020). APCOR’s Cork Yearbook 2020. https://www.apcor.pt/wp-content/uploads/2021/08/Cork_BoletimEstatistico_APCOR_2020.pdf. Accessed Dec 2022.
Aronson, J., Pereira, J., & Pausas, J. (2010). Cork Oak Woodlands on the Edge - Ecology, Adaptative Management, and Restoration. Island Press.
Bairrão, M., Trindade, M., Valdiviesso, T., Silva, M., Trindade, C. S., Ferreira, R. B., Cordovil, C., & Machado, H. (2021). Comparing intraspecific aggressiveness in Phytophthora cinnamomi isolates. Silva Lusitana, 29(2), 115–131. https://doi.org/10.1051/silu/20212902115
Brandano, A., Serra, S., Hardy, G. E. S. J., & Scanu, B. (2023). Potassium phosphonate induces resistance in sweet chestnut against ink disease caused by Phytophthora species. Pathogens, 12, 365. https://doi.org/10.3390/pathogens12030365
Brasier, C. M. (1996). Phytophthora cinnamomi and oak decline in southern Europe. Environmental constraints including climate change. Annales Des Sciences Forestières, 53, 347–358. https://doi.org/10.1051/forest:19960217
Brasier, C. M., Robredo, F., & Ferraz, J. F. P. (1993). Evidence for Phytophthora cinnamomi involvement in Iberian oak decline. Plant Pathology, 42(1), 140–145. https://doi.org/10.1111/j.1365-3059.1993.tb01482.x
Camilo-Alves, C., Clara, M., & Ribeiro, N. (2013). Decline of Mediterranean oak trees and its association with Phytophthora cinnamomi: A review. European Journal of Forest Research, 132, 411–432. https://doi.org/10.1007/s10342-013-0688-z
Drenth, E. A., & Guest, D. I. (2004). Principles of Phytophthora disease management. In A Drenth, EA, & Guest, DI. Diversity and Management of Phytophthora in Southeast Asia. ACIAR Monograph, 114, 154–159.
Duque-Lazo, J., Navarro-Cerrillo, R. M., van Gils, H., & Groen, T. A. (2018). Forecasting oak decline caused by Phytophthora cinnamomi in Andalusia: Identification of priority areas for intervention. Forest Ecology and Management, 417, 122–136. https://doi.org/10.1016/j.foreco.2018.02.045
Eggers, J. E., Balci, Y., & MacDonald, W. L. (2012). Variation among Phytophthora cinnamomi isolates from oak forest soils in the Eastern United States. Plant Disease, 96(11), 1608–1614. https://doi.org/10.1094/PDIS-02-12-0140-RE
Erwin, D. C., & Ribeiro, O. K. (1996). Phytophthora diseases worldwide. St. Paul: American Phytopathological Society (APS Press).
Eshraghi, L., Anderson, J., Aryamanesh, N., Shearer, B., Mccomb, J., Hardy, G. E., & O’Brien, P. A. (2011). Phosphite primed defence responses and enhanced expression of defence genes in Arabidopsis thaliana infected with Phytophthora cinnamomi. Plant Pathology, 60, 1086–1095. https://doi.org/10.1111/j.1365-3059.2011.02471.x
Fernandez-Escobar, R., Gallego, F. J., Benlloch, M., Membrillo, J., Infante, J., & Pérez de Algaba, A. (1999). Treatment of oak decline using pressurized injection capsules of antifungal materials. European Journal of Forest Pathology, 29, 29–38. https://doi.org/10.1046/j.1439-0329.1999.00127.x
FRAC. (2018). FRAC Code List 2018: Fungicides sorted by mode of action. http://www.phibase.org/images/fracCodeList.pdf. Accessed Nov 2022.
Gentile, S., Valentino, D., & Tamietti, G. (2009). Control of ink disease by trunk injection of potassium phosphite. Journal of Plant Pathology, 91(3), 565–571.
González, M., Caetano, P., & Sánchez, M. E. (2017). Testing systemic fungicides for control of Phytophthora oak root disease. Forest Pathology, 47, e12343. https://doi.org/10.1111/efp.12343
González, M., Romero, M. Á., Serrano, M. S., & Sánchez, M. E. (2020). Fosetyl-aluminium injection controls root rot disease affecting Quercus suber in southern Spain. European Journal Plant Pathology, 156, 101–109. https://doi.org/10.1007/s10658-019-01865-1
Groves, E., Howard, K., Hardy, G., & Burgess, T. (2015). Role of salicylic acid in phosphite-induced protection against Oomycetes; a Phytophthora cinnamomi - Lupinus augustifolius model system. European Journal of Plant Pathology, 141(3), 559–569. https://doi.org/10.1007/s10658-014-0562-y
Hardham, A. R. (2005). Phytophthora cinnamomi. Molecular Plant Pathology, 6, 589–604. https://doi.org/10.1111/j.1364-3703.2005.00308.x
Hardham, A. R., & Blackman, L. M. (2018). Phytophthora cinnamomi. Molecular Plant Pathology, 19, 260–285. https://doi.org/10.1111/mpp.12568
Jeffers, S. N., & Martin, S. B. (1986). Comparison of two media selective for Phytophthora and Pythium species. Plant Disease, 70, 1038–1043.
Jung, T., Blaschke, H., & Neumann, P. (1996). Isolation, identification and pathogenicity of Phytophthora species from declining oak stands. European Journal Forest Pathology, 26, 253–272. https://doi.org/10.1111/j.1439-0329.1996.tb00846.x
Kuberan, T., Balamurugan, A., Vidhyapallavi, R., Nepolean, P., Jayanthi, R., Beulah, T., & Premkumar, R. (2012). In vitro evaluation certain plant extracts against Glomerella cingulata causing brown blight disease of tea. World Journal of Agricultural Sciences, 8(5), 464–467.
Monteiro, S., Freitas, R., Rajasekhar, B. T., Teixeira, A. R., & Ferreira, R. B. (2010). The unique biosynthetic route from Lupinus β-conglutin gene to blad. PLoS ONE, 5(1), e8542. https://doi.org/10.1371/journal.pone.0008542
Monteiro, S., Carreira, A., Freitas, R., & Pinheiro, A. M. (2015). A nontoxic polypeptide oligomer with a fungicide potency under agricultural conditions which is equal or greater than that of their chemical counterparts. PLOS ONE, 10(4). https://doi.org/10.1371/journal.pone.0122095
Moreira, A. C., Ferraz, J., & Clegg, J. (2000). Phytophthora diseases of forest trees. IUFRO Working Party 7.02.09 Proceedings from the First International Meeting on Phytophthoras in Forest and Wildland Ecosystems.
Nashwa, S. M. A., & Abo-Elyousr, K. A. M. (2012). Evaluation of various plant extracts against the early blight disease of tomato plants under greenhouse and field conditions. Plant Protection Science, 48(2), 74–79. https://doi.org/10.17221/14/2011-PPS
Popova, L. (2013). Recent advances and future prospects on practical use of salicylic acid. In S. Hayat, A. Ahmad, & M. Alyemeni (Eds.), Salicylic Acid - Plant growth and development. Springer.
R Core Team. (2022). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed Dec 2022.
Romero, M. A., González, M., Serrano, M. S., & Sánchez, M. E. (2019). Trunk injection of fosetyl-aluminium controls the root disease caused by Phytophthora cinnamomi on Quercus ilex Woodlands. Annals of Applied Biology, 174(3), 313–318. https://doi.org/10.1111/aab.12503
Rosário, J. N., Coelho, V., Rodrigues, M. Â., Raimundo, S., Afonso, S., Arrobas, M., & Gouveia, E. (2021). Metalaxyl-M, phosphorous acid and potassium silicate applied as soil drenches show different chestnut seedling performance and protection against Phytophthora root rot. European Journal of Plant Pathology, 161, 147–159. https://doi.org/10.1007/s10658-021-02309-5
RStudio Team. (2022). RStudio: Integrated Development for R. RStudio, Inc. http://www.rstudio.com/. Accessed Dec 2022.
Saladin, G., Magné, C., & Clément, C. (2003). Effects of fludioxonil and pyrimethanil, two fungicides used against Botrytis cinerea, on carbohydrate physiology in Vitis vinifera L. Pest Management Science, 59, 1083–1092. https://doi.org/10.1002/ps.733
Sánchez, M. E., Caetano, P., Ferraz, J., & Trapero, A. (2002). Phytophtora disease of Quercus ilex in southwestern Spain. Forest Pathology, 32, 5–18. https://doi.org/10.1046/j.1439-0329.2002.00261.x
Sánchez, M., Andicoberry, S., & Trapero, A. (2005). Pathogenicity of three Phytophthora spp. causing late seedling rot of Quercus ilex ssp. ballota. Forest Pathology, 35, 115–125. https://doi.org/10.1111/j.1439-0329.2004.00392.x
Seddaiu, S., Brandano, A., Ruiu, P. A., Sechi, C., & Scanu, B. (2020). An Overview of Phytophthora Species Inhabiting Declining Quercus suber Stands in Sardinia (Italy). Forests, 11, 971. https://doi.org/10.3390/f11090971
Silva, P. V., Vélez, M. L., Otaño, D. H., Nuñez, C., & Greslebin, A. G. (2015). Action of fosetyl-al and metalaxyl against Phytophthora austrocedri. Forest Pathology, 46(1), 54–66. https://doi.org/10.1111/efp.12216
Sivanandhan, S., Khusro, A., Paulraj, M. G., Ignacimuthu, S., & Al-Dhabi, N. A. (2017). Biocontrol properties of Basidiomycetes: An overview. Journal of Fungi, 3(1), 2. https://doi.org/10.3390/jof3010002
Solla, A., Moreno, G., Malewski, T., Jung, T., Klisz, M., Tkaczyk, M., Siebyla, M., Pérez, A., Cubera, E., Hrynyk, H., Szulc, W., Rutkowska, B., Martín, J. A., Belbahri, L., & Oszako, T. (2021). Phosphite spray for the control of oak decline induced by Phytophthora in Europe. Forest Ecology and Management, 485, 118938. https://doi.org/10.1016/j.foreco.2021.118938
WIPO. (2007). Protein extracted from plants of the genus Lupinus or produced in recombinant form, nucleotide sequence encoding it and its use in animal nutrition, as a plant growth promoter and in the fight against pathogenic fungi - WO 2007/010459 A3. https://patentimages.storage.googleapis.com/d5/60/d7/2f39a59947e377/WO2007010459A3.pdf. Accessed Dec 2022.
Acknowledgments
We thank Santo Isidro nursery (Pegões, Portugal) for providing the plant material used in this study.
Funding
Open access funding provided by FCT|FCCN (b-on). This work was supported by national funds from Fundação para a Ciência e Tecnologia through the research unit UID/AGR/04129/2013 (LEAF) and the Project FCT/PTDC/ASP-SIL/29776/2017: Unconventional approaches in our struggle against Phytophthora in the cork oak forest. Zooming-in on the microbiome.
Author information
Authors and Affiliations
Contributions
Conceptualization: Margarida Bairrão, Teresa Valdiviesso, Helena Machado, Ricardo Boavida Ferreira; Writing: original draft preparation: Margarida Bairrão — Writing, review and editing: Manuel Trindade; Histology: Margarida Bairrão; Candida Sofia Trindade; Teresa Valdiviesso; Supervision: Teresa Valdiviesso, Helena Machado, Cláudia Marques-dos-Santos, Ricardo Boavida Ferreira; Funding acquisition: Ricardo Boavida Ferreira.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
All authors have read and agreed to the published version of the manuscript.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bairrão, M., Trindade, M., Trindade, C.S. et al. Non-conventional treatments to control cork oak infection by Phytophthora cinnamomi. Eur J Plant Pathol 168, 625–633 (2024). https://doi.org/10.1007/s10658-023-02789-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-023-02789-7