Abstract
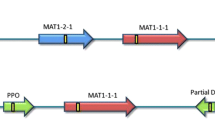
Sexual reproduction in fungi is controlled by mating type genes, which are located at the MAT locus. In this study, we investigated the structure of this locus in the phytopathogenic fungus Phyllosticta citricarpa, the causal agent of citrus black spot disease. Despite intensive study, its sexual state has never been observed in single-spore culture. Through analysis of the genome sequences of two individual P. citricarpa isolates, the sequence of the DNA lyase gene was identified and, as previously reported in the literature, the mating type genes were located in the 3′ flanking region of this gene. The results suggested that P. citricarpa is heterothallic, owing to the exclusive presence of the MAT1–1 or MAT1–2 gene in individual strains. In order to characterize the MAT locus, we designed primers to amplify this region. P. citricarpa was found to have complete and apparently functional copies of MAT genes, containing α-1 and HMG domains, present in different isolates. In addition to MAT1–2-1 and MAT1–1-1 genes, the MAT1–1-4 gene was located in the 5′ flanking region of the MAT1–1-1 gene and the MAT1–2-5 gene was located in 5′ flanking region of the MAT1–2-1 gene. A multiplex PCR protocol was also developed to differentiate P. citricarpa idiomorphs, which can be used in distribution and incidence studies of mating type strains, in order to determine the occurrence of sexual reproduction and to facility crossing studies. Furthermore, in Brazil, the two idiomorphs occur in a 1:1 ratio, which is expected in sexually reproducing populations.



Similar content being viewed by others
References
Azevedo, J. L., & Costa, S. O. P. (1973). Exercícios práticos de genética (p. 288). São Paulo: EDUSP.
Baayen, R. P., Bonants, P. J. M., Verkley, G., Carroll, G. C., Van der Aa, H. A., Weerdt, M., et al. (2002). Nonpathogenic isolates of the citrus black spot fungus, Guignardia citricarpa, identified as a cosmopolitan endophyte of woody plants, G. mangiferae (Phyllosticta capitalensis). Phytopathology, 92, 464–477.
Bihon, W., Wingfield, M. J., Slippers, B., Duong, T. A., & Wingfield, B. D. (2014). MAT gene idiomorphs suggest a heterothallic sexual cycle in a predominantly asexual and important pine pathogen. Fungal Genetics and Biology, 62, 55–61.
Blindauer, C. A., Harrison, M. D., Robinson, A. K., Parkinson, J. A., Bowness, P. W., Sadler, P. J., et al. (2002). Multiple bacteria encode metallothioneins and SmtA-like zinc fingers. Molecular Microbiology, 45, 1421–1432.
Bohm, J., Hoff, B., O’Gorman, C. M., Wolfers, S., Klix, V., Binger, D., et al. (2013). Sexual reproduction and mating-type-mediated strain development in the penicillin-producing fungus Penicillium chrysogenum. Proceedings of the National Academy of Sciences of the United States of America, 110, 1476–1481.
Brewer, M. T., Cadle-Davidson, L., Cortesi, P., Spanu, P. D., & Milgroom, M. G. (2011). Identification and structure of the mating-type locus and development of PCR-based markers for mating type in powdery mildew fungi. Fungal Genetics and Biology, 48, 704–713.
Burgess, T., Wubetu Bihon, W., Wingfield, M. J., & Wingfield, B. D. A. (2009). A singe and rapid method to determine vegetative compatibility groups in fungi. Mycology, 60, 6.
Coyle, P., Philcox, J. C., Carey, L. C., & Rofe, A. M. (2002). Metallothionein: The multipurpose protein. Cellular Molecular Life Science, 59, 627–647.
Debuchy, R., & Turgeon, B. G. (2006). Mating-type structure, function, and evolution in Euascomycetes. In U. Kües & R. Fischer (Eds.), The Mycota I Growth, Differentiation, and Sexuality (pp. 293–323). Berlin: Springer-Verlag.
Debuchy, R., Lecellir, V. B., & Sillar, P. (2010). Mating systems and sexual morphogenesis in Ascomycetes. In K. A. Borkovich & D. J. Ebbole (Eds.), Cellular and Molecular Biology of Filamentous Fungi (pp. 501–535). Washington, DC: AMS Press.
Dyer, P. S., Furneaux, P. A., Douhan, G., & Murray, T. D. (2001). A multiplex PCR test for determination of mating type applied to the plant pathogens Tapesia yallundae and Tapesia acuformis. Fungal Genetics and Biology, 33, 173–180.
EFSA- European Food Safety Authority (2014). Scientific opinion on the risk of Phyllosticta citricarpa (Guignardia citricarpa) for the EU territory with identification and evaluation of risk reduction options. EFSA Journal, 12(2), 243.
Fu, R., Yin, C., Deng, Q., Li, P., & Zheng, A. (2014). Mating type loci analysis indicates that Villosiclava virens, the causal agent of false smut disease of rice, is a homothallic fungus. Tropical Plant Pathology, 39, 203–209.
Glienke, C., Pereira, O. L., Stringari, D., Fabris, J., Kava-Cordeiro, V., Galli-Terasawa, L., et al. (2011). Endophytic and pathogenic Phyllosticta species, with reference to those associated with citrus black spot. Persoonia: Molecular Phylogeny and Evolution of Fungi, 26, 47–56.
Glienke-Blanco, C., Aguilar-Vildoso, C. I., Vieira, M. L. C., Barroso, P. A. V., & Azevedo, J. L. (2002). Genetic variability in the endophytic fungus Guignardia citricarpa isolated from citrus plants. Genetic and Molecular Biology, 25, 251–255.
Groenewald, M., Barnes, I., Bradshaw, R. E., Brown, A. V., Dlae, A., Groenewald, J. Z., et al. (2007). Characterization and distribution of mating type genes in the Dothistroma needle blight pathogens. Mycology, 97(7), 825–834.
Hall, T. A. (1999). BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/988/NT. Nuclei Acids Symposium Series, 41, 95–98.
Heitman, J. (2015). Evolution of sexual reproduction: a view from the fungal kingdom supports an evolutionary epoch with sex before sexes. Fungal Biology Reviews, 3, 108–117.
Jones, D. T., Taylor, W. R., & Thornton, J. M. (1992). The rapid generation of mutation data matrices from protein sequences. Computer Applications in the Biosciences, 8, 275–282.
Kanamori, M., Kato, H., Yasuda, N., Koizumi, S., Peever, T. L., et al. (2007). Novel mating type-dependent transcripts at the mating type locus in Magnaporthe oryzae. Gene, 403, 6–17.
Katoh, K., & Toh, H. (2008). Recent developments in the MAFFT multiple sequence alignment program. Briefings in Bioinformatics, 9, 286–298.
Kronstad, J. (2007). Self-fertility: the genetics of sex in lonely fungi. Current Biology, 17, 844.
Lee, S. C., Ni, M., Li, W., Shertz, C., & Heitman, J. (2010). The evolution of Sex: a Perspective from the Fungal Kingdom. Microbiology and Molecular Biology Reviews, 74, 298–340.
Mandel, M. A., Barker, B. M., Kroken, S., Rounsley, S. D., & Orbach, J. (2007). Genomic and population analyses of the mating type loci in Coccidioides species reveal evidence for sexual reproduction and gene acquisition. Eukaryotic Cell, 6, 1189–1199.
Martin, S. H., Wingfield, B. D., Wingfield, M. J., & Steenkamp, E. T. (2011). Structure and evolution of the Fusarium mating type locus, new insights from the Gibberella fujikuroi complex. Fungal Genetics Biology, 48, 731–740.
McDonald, B. A., & Linde, C. (2002). Pathogen population genetics, evolutionary potential, and durable resistance. Annual Review of Phytopathology, 40, 349–379.
O’Gorman, C. M., Fuller, H. T., & Dyer, P. S. (2009). Discovery of a sexual cycle in the opportunistic fungal pathogen Aspergillus fumigatus. Nature, 457, 471.
Pacheco, I. S. (2014). Estudo comparativo do genoma e do transcriptoma de Phyllosticta citricarpa (Guignardia citricarpa) agente da Mancha Preta dos Citros e Phyllosticta capitalensis. 92 f. Dissertação – Instituto de Biociências da Universidade Estadual Paulista “Júlio de Mesquita Filho”, Campus de Botucatu.
Palmer, J. M., Kubatova, A., Novakova, A., Minnis, A. M., Kolarik, M., & Lindner, D. L. (2014). Molecular characterization of a heterothallic mating system in Pseudogymnoascus destructans, the fungus causing white-nose syndrome of bats. G3 (Bethesda), 4, 1755–1763.
Perryman, S. A. M., Clark, S. J., & West, J. S. (2014). Splash dispersal of Phyllostica citricarpa conida from infected citrus fruit. Scientific Reports, 4, 1–8.
Pöggeler, S. (2001). Mating-type genes for classical strain improvements of ascomycetes. Applied Microbiology and Biotechnology, 56, 589–601.
Pontecorvo, G., Roper, J. A., Hemmons, L. M., Mac Donald, K. D., & Bufton, A. W. J. (1953). The genetics of Aspergillus nidulans. Advances in Genetics, 5, 141–238.
Rambaut, A. (2012). FigTree (version 1.4.0). http://tree.bio.ed.ac.uk/software/figtree
Ronquist, F., & Huelsenbeck, J. P. (2003). Mrbayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics, 19, 1572–1574.
Rubini, A., Belfiori, B., Riccioni, C., Tisserant, E., Arcioni, S., Martin, F., et al. (2010). Isolation and characterization of MAT genes in the symbiotic ascomycete Tuber melanosporum. New Phytologist, 189, 710–722.
Sanger, F., Nicklen, S., & Coulson, A. R. (1977). DNA sequencing with chain-terminating inhibitors. Proceedings of the National Academy of Sciences of the United States of America, 74, 5463–5467.
Santos, J. M., Correia, V. G., & Phillips, A. J. L. (2010). Primers for mating-type diagnosis in Diaporthe and Phomopsis: their use in teleomorph induction in vitro and biological species definition. Fungal Biology, 114, 255–270.
Schoch, C. L., Crous, P. W., Groenewald, J. Z., Boehm, E. W. A., Burgess, T. I., Gruyter, J., et al. (2009). A class-wide phylogenetic assessment of Dothideomycetes. Studies in Mycology, 64, 1–15.
Seidl, V., Seibel C., Kubicek C.P.,, & Schmoll, M. (2009) Sexual development in the industrial workhorse Trichoderma reesei. Proceeding of the National Academy of Sciences, 106, 13909–13914.
Singh, G., & Ashby, A. M. (1998). Cloning of the mating type loci from Pyrenopeziza brassicae reveals the presence of a novel mating type gene within a discomycete MAT 1–2 locus encoding a putative metallothionein-like protein. Molecular Microbiology, 30, 799–806.
Sposito, M. B., Amorin, L., Bassanezi, R. B., Filho, A. B., & Hau, B. (2008). Spatial pattern of black spot incidence within citrus trees related to disease severity and pathogen dispersal. Plant Pathology, 57, 103–108.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., & Kumar, S. (2013). MEGA6: Molecular evolutionary genetics analysis version 6.0. Molecular. Biology and Evolutionary, 16, 1–5.
Thompson, J. D., Gibson, T. J., Plewniak, F., Jeanmougin, F., & Higgins, D. G. (1997). The CLUSTAL_X Windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Research, 25, 4876–4882.
Truter, M., Kotzé, J. M., Janse van Rensberg, T. N., & Korsten, L. (2004). A sampler to determine available Guignardia citricarpa inoculum on citrus leaf litter. Biosystems Engineering, 89, 515–519.
Tsui, C. K. M., DiGuistini, S., Wang, Y., Feau, N., Dhillon, B., Bohlmann, J., et al. (2013). Unequal recombination and evolution of the mating-type (MAT) loci in the pathogenic fungus Grosmannia clavigera and relatives. G3 (Bethesda), 3, 465–480.
Tucker, S. L., Thornton, C. R., Tasker, K., Jacob, C., Giles, G., Egan, M., et al. (2004). A fungal metallothionein is required for pathogenicity of Magnaporthe grisea. The Plant Cell, 16, 1575–1588.
Turgeon, B. G., & Yoder, O. C. (2000). Proposed nomenclature for mating type genes of filamentous Ascomycetes. Fungal Genetics and Biology, 31, 1–5.
Wikee, S., Lombard, L., Nakashima, C., Motohashi, K., Cheewangkoon, R., Mckenzie, E. H. C., et al. (2013). A phylogenetic re-evaluation of Phyllosticta (Botryosphaeriales). Studies in Mycology, 76, 1–29.
Ye, J., Coulouris, G., Zaretskaya, I., Cutcutache, I., Rozen, S., & Madden, T. (2012). Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics, 13, 134.
Acknowledgments
We are grateful to the Instituto Agronômico de Campinas for the partnership. This work is financially supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), and Fundação Araucária.
Author information
Authors and Affiliations
Corresponding author
Additional information
Renata Amorim and Daiani Cristina Savi have contributed equally to this work
Rights and permissions
About this article
Cite this article
Amorim, R., Savi, D.C., Ferreira-Maba, L. et al. MAT gene idiomorphs suggest a heterothallic sexual cycle in the citrus pathogen Phyllosticta citricarpa . Eur J Plant Pathol 147, 325–337 (2017). https://doi.org/10.1007/s10658-016-1005-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-1005-8