Abstract
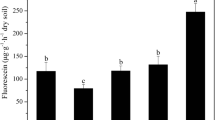
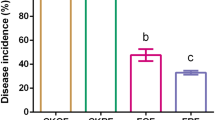
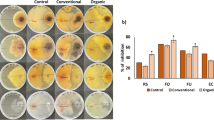
Soil fumigants constitute a major tool for the control of soil borne fungal plant pathogens in protected crops. Dimethyl disulfide (DMDS) is a novel soil fumigant used either alone or in combination with other fumigants for the control of soil borne pests and diseases. In a commercial greenhouse for tomato production, we evaluated the impact of DMDS, comparatively to the alternative fumigant metam sodium, on the population of the dominant fungal plant pathogens in the study soil via q-PCR. Prior to soil fumigation, estimation of the fungal diversity in the studied soil via clone libraries identified Fusarium oxysporum and Rhizoctonia solani as the most abundant soil borne plant pathogens, while Cladosporium spp., known as opportunistic airborne tomato pathogens, were the most dominant fungi and based on this their dynamics upon fumigation was also studied. DMDS, at two dose rates, induced a drastic reduction in the population of F. oxysporum and R. solani, which lasted for the whole cultivation season. On the contrary, metam sodium exhibited an inhibitory effect on F. oxysporum that was alleviated at 120 d post fumigation. Both DMDS and metam sodium induced only a temporal reduction in the soil population of Cladosporium sp. which recovered by 60 days post fumigation. Our data suggest that DMDS even at the low dose rate (56.4 g m−2) could drastically reduce the population of the major soil borne tomato pathogens F. oxysporum and R. solani. Establishment of population thresholds as determined by q-PCR could represent a valuable tool for the estimation of the risk for disease severity and crop yield losses.




Similar content being viewed by others
References
Arnault, I., Fleurance, C., Vey, F., Fretay, G. D., & Auger, J. (2013). Use of Alliaceae residues to control soil-borne pathogens. Industrial Crops and Products, 49, 265–272.
Bartz, F. E., Cubeta, M. A., Toda, T., Naito, S., & Ivors, K. L. (2010). An in planta method for assessing the role of basidiospores in Rhizoctonia foliar disease of tomato. Plant Disease, 94, 515–520.
Basallote-Ureba, M. J., Vela-Delgado, M. D., Maclas, F. J., Lope-Herrera, C. J., & Melero-Vara, J. M. (2010). Soil chemical treatments for the control of fusarium wilt of carnation in Spain. Acta Horticulturae, 883, 175–180.
Belova, A., Narayan, T., & Olkin, I. (2013). Methyl bromide alternatives for strawberry and tomato pre-plant uses: A meta-analysis. Crop Protection, 54, 1–14.
Bending, G. D., & Lincoln, S. D. (2002). Inhibition of soil nitrifying bacteria communities and their activities by glucosinolate hydrolysis products. Soil Biology and Biochemistry, 32, 1261–1269.
Bensch, K., Groenewald, J. Z., Dijksterhuis, J., Starink-Willemse, M., Andersen, B., Summerell, B. A., Shin, H. D., Dugan, F. M., Schroers, H. J., Braun, U., & Crous, P. W. (2010). Species and ecological diversity within the Cladosporium cladosporioides complex (Davidiellaceae, Capnodiales). Studies in Mycology, 67, 1–94.
Budge, G. E., Shaw, M. W., Colyer, A., Pietravalle, S., & Boonham, N. (2009). Molecular tools to investigate Rhizoctonia solani distribution in soil. Plant Pathology, 58, 1071–1080.
Cabrera, J. A., Wang, D., Gerik, J. S., & Gan, J. (2014). Spot drip application of dimethyl disulfide as a post-plant treatment for the control of plant parasitic nematodes and soilborne pathogens in grape production. Pest Management Science, 71, 1151–1157.
Cai, G., Gale, L. R., Schneider, R. W., Kistler, H. C., Davis, R. M., Elias, K. S., & Miyao, E. M. (2003). Origin of race 3 of Fusarium oxysporum f. sp. lycopersici at a single site in California. Phytopathology, 93, 1014–1022.
Coats, V. C., Pelletreau, K. N., & Rumpho, M. E. (2014). Amplicon pyrosequencing reveals the soil microbial diversity associated with invasive Japanese barberry (Berberisthun bergii DC.). Molecular Ecology, 23, 1318–1332.
Curto, G., Dongiovanni, C., Sasanelli, N., Santori, A., & Myrta, A. (2014). Efficacy of dimethyl disulfide (DMDS) in the control of the root-knot nematode Meloidogyne incognita and the cyst nematode Heterodera carotae on carrot in field condition in Italy. Proc. VIIIth IS on chemical and non-chemical soil and Substrate disinfestation. eds.: M.L. Gullino et al. Acta Horticulturae 1044. ISHS, 2014, 405–410.
Dangi, S. R., Tirado-Corbala, R., Cabrera, J. A., Wang, D., & Gerik, J. (2013). Soil biotic and abiotic responses to dimethyl disulfide spot drip fumigation in established grape vines. Soil Science Society of America Journal, 78, 520–530.
Debode, J., Van Poucke, K., França, S. C., Maes, M., Höfte, M., & Heungens, K. (2011). Detection of multiple Verticillium species in soil using density flotation and real-time polymerase chain reaction. Plant Disease, 95, 1571–1580.
Duniway, J. M. (2002). Status of chemical alternatives to methyl bromide for pre-plant fumigation in soil. Phytopathology, 92, 1337–1343.
Edel, V., Steinberg, C., Gautheron, N., Recorbet, G., & Allabouvette, C. (2001). Genetic diversity of Fusarium oxysporum populations isolated from different soils in France. FEMS Microbiology Ecology, 36, 61–71.
El-Deeb, H. M., & Arab, Y. A. (2013). Acremonium as an endophytic bioagent against date palm fusarium wilt. Archives of Phytopathology and Plant Protection, 46, 1214–1221.
Flannigan, B. (2001). Microorganisms in indoor air. In R. A. Samson & J. D. Miller (Eds.), Microorganisms in Home and indoor work Environments: diversity, Health Impacts, Investigation and control (Flannigan B (pp. 17–31). London: Taylor & Francis.
Fritsch, J. (2005). Dimethyl disulfide as a new chemical potential alternative to methyl bromide in soil disinfestation in France. Acta Horticulturae, 698, 71–76.
Fritsch, J., Fouillet, T., Charles, P., Fargier-Puech, P., Ramponi-Bur, C., Descamps, S., Du Fretay, G., & Myrta, A. (2014). French experiences with dimethyl disulfide (DMDS) as a nematicide in vegetable crops. Proc. VIIIth IS on chemical and non-chemical soil and Substrate disinfestation. eds.: M.L. Gullino et al. Acta Horticulturae 1044. ISHS, 2014, 427–434.
Gange, A. C., Eschen, R., Wearn, J. A., Thawer, A., & Sutton, B. C. (2012). Differential effects of foliar endophytic fungi on insect herbivores attacking a herbaceous plant. Oecologia, 168, 1023–1031.
Gerik, J. S. (2005). Evaluation of soil fumigants applied by drip irrigation for Liatris production. Plant Disease, 89, 883–887.
Giannakou, I. O., & Karpouzas, D. G. (2003). Evaluation of chemical and integrated strategies as alternatives to methyl bromide for the control of root-knot nematodes in Greece. Pest Management Science, 59, 883–892.
Good, I. J. (1953). The population frequencies of species and the estimation of population parameters. Biometrika, 43, 45–63.
Hirano, Y., & Arie, T. (2006). PCR-based differentiation of Fusarium oxysporum f. sp. lycopersici and radicis-lycopersici and races of F. oxysporum f. sp. lycopersici. Journal of General Plant Pathology, 72, 273–283.
Hollins, P. D., Kettlewell, P. S., Atkinson, M. D., Stephenson, D. B., Corden, J. M., Millington, W. M., & Mullins, J. (2004). Relationships between airborne fungal spore concentration of Cladosporium and the summer climate at two sites in Britain. International Journal of Biometeorology, 48, 137–141.
Inoue, I., Namiki, F., & Tsuge, T. (2002). Plant colonization by the vascular wilt fungus Fusarium oxysporum requires FOW1, a gene encoding a mitochondrial protein. Plant Cell, 14, 1869–1883.
Karpouzas, D. G., Karatasas, A., Spyridaki, E., Rousidou, C., Bekris, F., Ehaliotis, C., & Papadopoulou, K. K. (2011). Impact of a beneficial and of a pathogenic Fusarium strain on the fingerprinting-based structure of microbial communities in tomato (Lycopersicon esculentum Milll.) rhizosphere. European Journal of Soil Biology, 47, 400–408.
Kernaghan, G., Reeleder, R. D., & Hoke, S. M. T. (2008). Quantification of Pythium populations in ginseng soils by culture dependent and real-time PCR methods. Applied Soil Ecology, 40, 447–455.
Kirkegaard, J. A., & Sarwar, M. (1998). Biofumigation potential of brassicas I. Variation in glucosinolate profiles of diverse field-grown brassicas. Plant and Soil, 201, 71–89.
Lenc, L., Kwasna, H., Sadowski, C., & Grabowski, A. (2015). Microbiota in wheat roots, rhizosphere and soil in crops grown in organic and other production systems. Journal of Phytopathology, 163, 245–263.
Li, Y., Garibaldi, A., & Gullino, M. L. (2010). Molecular detection of Fusarium oxysporum f. sp. chrysanthemi on three host plants: Gerbera jamesonii, Osteospermum sp. and Argyranthemum frutescens. Journal of Plant Pathology, 92, 515–520.
Li, Y., Mao, L., Yan, D., Ma, T., Shen, J., Guo, M., Wang, Q., Ouyang, C., & Cao, A. (2014). Quantification of Fusarium oxysporum in fumigated soils by a newly developed real-time PCR assay to assess the efficacy of fumigants for fusarium wilt disease in strawberry plants. Pest Management Science, 70, 1669–1675.
Lievens, B., Brouwer, M., Vanachter, A. C. R. C., Levesque, C. A., Camue, B. P. A., & Thomma, B. P. H. J. (2005). Quantitative assessment of phytopathogenic fungi in various substrates using a DNA macroarray. Environmental Microbiology, 7, 1698–1710.
Ma, Y., Gentry, T., Hu, P., Pierson, E., Gu, M., & Yin, S. (2015). Impact of brassicaceous seed meals on the composition of the soil fungal community and the incidence of fusarium wilt on chili pepper. Applied Soil Ecology, 90, 41–48.
Mao, L., Yan, D., Wang, Q., Li, Y., Ouyang, C., Liu, P., Shen, J., Guo, M., & Cao, A. (2014). Evaluation of the combination of dimethyl disulfide and dazomet as an efficient methyl bromide alternative for cucumber production in China. Journal of Agricultural and Food Chemistry, 62, 4864–4869.
Mirtalebi, M., Banihashemi, Z., & Linde, C. C. (2013). Phylogenetic relationships of Fusarium oxysporum f. sp. melonis in Iran. European Journal of Plant Pathology, 136, 749–762.
Mullins, J. (2001). Microorganisms in outdoor air. In R. A. Samson & J. D. Miller (Eds.), Microorganisms in Home and indoor work Environments: diversity, Health Impacts, Investigation and control (Flannigan B (pp. 3–16). London: Taylor & Francis.
Omirou, M., Karpouzas, D. G., Papadopoulou, K. K., & Ehaliotis, C. (2013). The decomposition of pure and plant – derived glucosinolates in soil. European Journal of Soil Biology, 56, 49–55.
Rivas, S., & Thomas, C. M. (2005). Molecular interactions between tomato and the leaf mold pathogen Cladosporium fulvum. Annual Review of Phytopathology, 43, 395–436.
Rousidou C, Papadopoulou E, Kortsinidou M, Giannakou IO, Singh BK, Menkissoglou-Spiroudi U, Karpouzas DG (2013) Bio-pesticides: harmful or harmless to ammonia oxidizing microorganisms? The case of a Paecilomyces lilacinus-based nematicide. Soil Biology & Biochemistry, 67 98–105.
Sarwar, M., Kirkegaard, J. A., Wong, P. T. W., & Desmarchelier, J. M. (1998). Biofumigation potential of brassicas III in vitro toxicity of isothiocyanates to soil-borne fungal pathogens. Plant and Soil, 201, 103–112.
Sasanelli, N., Dongiovanni, C., Santori, A., & Myrta, A. (2014). Control of the root-knot nematode Meloidogyne incognita by dimethyl disulfide (DMDS) applied in drip irrigation on melon and tomato in Apulia and Basilicata (Italy). Proc. VIIIth IS on chemical and non-chemical soil and Substrate disinfestation. eds.: M.L. Gullino et al. Acta Horticulturae 1044. ISHS, 2014, 401–404.
Schoch, C. L., Seifert, K. A., Huhndorf, S., Robert, V., Spouge, J. L., Levesque, C. A., & Chen, W. (2012). Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for fungi. Proceedings of the National Academy of Sciences, 109, 6241–6246.
Siles, J. A., Pascual, J., González-Menéndez, V., Sampedro, I., García-Romera, I., et al. (2014). Short-term dynamics of culturable bacteria in a soil amended with biotransformed dry olive residue. Systems of Applied Microbiology, 37, 113–120.
Singleton, L. L., Mihail, J. D., & Rush, C. M. (1992). Methods for research on soil borne phytopathogenic fungi. St. Paul, MN, USA: APS Press.
Soytong, K., Kanokmedhakul, S., Kukongviriyapa, V., & Isobe, M. (2001). Application of Chaetomium species (Ketomium) as a new broad spectrum biological fungicide for plant disease control: A review article. Fungal Diversity, 7, 1–15.
Termorshuizen, A. J., Davis, J. R., Gort, G., Harris, D. C., Huisman, O. C., Lazarovits, G., et al. (1998). Inter-laboratory comparison of methods to quantify microsclerotia of Verticillium dahliae in soil. Applied and Environmental Microbiology, 64, 3846–3853.
Thorn, R. G., Reddy, C. A., Harris, D., & Paul, E. A. (1996). Isolation of saprophytic Basidiomycetes from soil. Applied and Environmental Microbiology, 62, 4288–4292.
Vakalounakis, D. J., & Williams, P. H. (1989). A cotyledon screen for resistance to scab (Cladosporium cucumerinum) in cucumber (Cucumis sativus) seedlings. Annals of Applied Biology, 115, 443–450.
Yan, L., Zhang, C., Ding, L., & Ma, Z. (2008). Development of a real-time PCR assay for the detection of Cladosporium fulvum in tomato leaves. Journal of Applied Microbiology, 104, 1417–1424.
Yan, D., Wang, Q., Mao, L., Li, W., Xie, H., Guo, M., & Cao, A. (2013). Quantification of the effects of various soil fumigation treatments on nitrogen mineralization and nitrification in laboratory incubation and field studies. Chemosphere, 90, 1210–1215.
Zheng, Q.-Y., Westermark, S.-O., Rasmunson-Lestander, A., & Wang, X.-R. (2005). Detection and quantification of Cladosporium in aerosols by real-time PCR. Journal of Environmental Monitoring, 8, 153–160.
Acknowledgments
This work was financial supported by the agrochemical company K + N EFTHYMIADIS (Project No. 4703) and by the Postgraduate Program “Biotechnology- Quality assessment in Nutrition and the Environment”, Department of Biochemistry and Biotechnology, University of Thessaly.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papazlatani, C., Rousidou, C., Katsoula, A. et al. Assessment of the impact of the fumigant dimethyl disulfide on the dynamics of major fungal plant pathogens in greenhouse soils. Eur J Plant Pathol 146, 391–400 (2016). https://doi.org/10.1007/s10658-016-0926-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-0926-6