Abstract
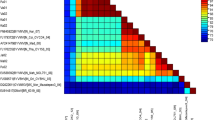
Yellow vein mosaic disease of okra is major constraint for the production of okra in India. It is caused by whitefly transmitted begomoviruses and results in severe economic losses of the crop. In the present study a new begomovirus (OYBHU isolate) infecting okra showing yellow vein symptoms from Bhubhaneswar, India was characterized. The complete genome sequence (homologous to DNA-A) was determined and it comprised 2757 nucleotides. Attempts to amplify DNA- B were not successful. However, the association of a betasatellite suggests that the isolate is monopartite and typical of the begomoviruses infecting malvaceous hosts in the Old World. The genome organization is characteristic of begomoviruses, encoding seven ORFs with two ORFs [AV1 (CP) and AV2] in virion sense strand and five ORFs (AC1-AC5) in complementary strand. Comparisons of the virus genome sequence with other known begomoviruses suggests that OYBHU virus isolate is distinct, with Croton yellow vein mosaic virus being its nearest relative, sharing the 81.4 to 86.1 % sequence identity. This is further supported by phylogenetic analyses with clustering of BYVBV on a well supported branch separated from all other begomoviruses. The name Bhendi yellow vein Bhubhaneswar virus (BYVBV) is proposed for virus isolate OYBHU with the additional descriptor [India: Bhubhaneswar:OYBHU:06]. Recombination analyses with BYVBV suggest that it is a recombinant and may have originated by the exchange of genomic segments between the begomoviruses Croton yellow vein mosaic virus, Bhendi yellow vein mosaic virus, Cotton leaf curl multan virus and Mesta yellow vein mosaic virus.




Similar content being viewed by others
References
Banks, G. K., Colvin, J., Chowda Reddy, R. V., Maruthi, M. N., Muniyappa, V., Venkatesh, H. M., et al. (2001). First report of the Bemisia tabaci B biotype in India and an associated tomato leaf curl virus disease epidemic. Plant Disease, 85, 231.
Berrie, L. C., Rybicki, E. P., & Rey, M. E. C. (2001). Complete nucleotide sequence and host range of South African cassava mosaic virus: further evidence for recombinations amongst begomoviruses. Jounalof General Virology, 82, 53–58.
Briddon, R. W., & Stanley, J. (2006). Subviral agents associated with plant single-stranded DNA viruses. Virology, 344, 198–210.
Briddon, R. W., Bull, S. E., Mansoor, S., Amin, I., & Markham, P. G. (2002). Universal primers for the PCR-mediated amplification of DNA-: a molecule associated with some monopartite begomoviruses. Molecular Biotechnology, 20, 315–318.
Briddon, R. W., Bull, S. E., Amin, I., Idris, A. M., Mansoor, S., Bedford, I. D., et al. (2003). Diversity of DNA beta: a satellite molecule associated with some monopartitebegomoviruses. Virology, 312, 106–121.
Briddon, R. W., Bull, S. E., Amin, I., Mansoor, S., Bedford, I. D., Rishi, N., et al. (2004). Diversity of DNA 1; a satellite-like molecule associated with monopartite begomovirus-DNA β complexes. Virology, 324(4), 62–474.
Chakraborty, S., Pandey, P. K., Banerjee, M. K., Kalloo, G., & Fauquet, C. M. (2003). Tomato leaf curl Gujarat virus, a new begomovirus species causing a severe leaf curl disease of tomato in Varanasi, India. Phytopathology, 93, 1485–1495.
Chatchawankanphanich, O., & Maxwell, D. P. (2002). Tomato leaf curl Karnataka virus from Bangalore, India, appears to be a recombinant begomovirus. Phytopathology, 92, 637–645.
Chatterjee, A., & Ghosh, S. K. (2007). A new monopartite begomovirus isolated from Hibiscus cannabinus L. in India. Archives of Virology, 152, 2113–2118.
Doyle, J. J., & Doyle, J. L. (1990). Isolation of plant DNA from fresh tissue. Focus, 12, 13–15.
Fauquet, C. M., Briddon, R. W., Brown, J. K., Moriones, E., Stanley, J., Zerbini, M., et al. (2008). Geminivirus strain demarcation and nomenclature. Archives of Virology, 153, 783–821.
Hall, T. A. (1999). BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symposium Series, 41, 95–98.
Hanley-Bowdoin, L., Settlage, S. B., Orozco, B. M., Nagar, S., & Robertson, D. (2000). Geminiviruses: models for plant DNA replication, transcription, and cell cycle regulation. Critical Review in Biochemistry and Molecular Biology, 35(2), 105–140.
Huson, D. H., & Bryant, D. (2006). Application of phylogenetic networks in evolutionary studies. Molecular Biology and Evolution, 23, 254–267.
Jeanmougin, F., Thompson, J. D., Gouy, M., Higgins, D. G., & Gibson, T. J. (1998). Multiple sequence alignment with Clustal X. Trends in Biochemical Science, 23, 403–405.
Jeske, H., Lutgemeier, M., & Preiss, W. (2001). DNA forms indicate rolling circle and recombination-dependent replication of Abutilon mosaic virus. European Molecular Biology Journal, 20, 6158–6167.
Kirthi, N., Maiya, S. P., Murthy, M. R. N., & Savitri, H. S. (2002). Evidence of recombination among the tomato leaf curl virus strains/species from Bangalore, India. Archives of Virology, 147, 255–272.
Krishnareddy, M. (2010). Climate change and virus diseases of Horticultural crops. In H. P. Singh, J. P. Singh, & S. S. Lal (Eds.), Challenges of climate change – Indian Horticulture (pp. 153–165). New Delhi, India: Westville Publishing house. 209P.
Kumari, P., Singh, A. K., Chattopadhyay, B., & Chakraborty, S. (2010). Molecular characterization of a new species of Begomovirus and betasatellite causing leaf curl disease of tomato in India. Virus Research, 152, 19–29.
Lazarowitz, S. G. (1992). Geminiviruses: genome structure and gene function. Critical Review in Plant Science, 11, 327–349.
Lefeuvre, P., Martin, D. P., Hoareau, M., Naze, F., Delatte, H., Thierry, M., et al. (2007). Begomovirus ‘melting pot’ in the southwest Indian Ocean islands: molecular diversity and evolution through recombination. Journal of General Virology, 88, 3458–3468.
Mansoor, S., Khan, S. H., Bashir, A., Saeed, M., Zafar, Y., Malik, K. A., et al. (1999). Identification of a novel circular single-stranded DNA associated with cotton leaf curl disease in Pakistan. Virology, 259, 190–199.
Martin, D. P., Lemey, P., Lott, M., Vincent, M., Posada, D., & Lefeuvre, P. (2010). RDP3: a flexible and fast computer program for analyzing recombination. Bioinformatics, 26(19), 2462–2463.
Nawaz-ul-Rehman, M. S., Nahid, N., Mansoor, S., Briddon, R. W., & Fauquet, C. M. (2010). Post-transcriptional gene silencing suppressor activity of the alpha-Rep of non-pathogenic alphasatellites associated with begomoviruses. Virology, 405, 300–308.
Padidam, M., Sawyer, S., & Fauquet, C. M. (1999). Possible emergence of new geminiviruses by frequent recombination. Virology, 265, 218–225.
Pandey, P., Mukhopadhya, S., Naqvi, A. R., Mukherjee, S. K., Shekhawat, G. S., & Choudhury, N. R. (2010). Molecular characterization of two distinct monopartite begomoviruses infecting tomato in India. Virology Journal, 7, 337.
Polston, J. E., & Anderson, P. L. (1997). The emergence of whitefly transmitted geminiviruses in tomato in the Western Hemisphere. Plant Disease, 81, 1358–1369.
Prasanna, H. C., & Rai, M. (2007). Detection and frequency of recombination in tomato infecting begomoviruses of South and Southeast Asia. Virology Journal, 4, 111. doi:10.1186/1743-422X-4-111.
Rekha, A. R., Maruthi, M. N., Muniyappa, V., & Colvin, J. (2005). Occurrence of three genotypic clusters of Bemisia tabaci and the rapid spread of the B biotype in south India. Entomologia Experimentaliset Applicata, 117, 221–233.
Rojas, M. R., Gilbertson, R. L., Russel, D. R., & Maxwell, D. P. (1993). Use of degenerate primers in the polymerase chain reaction to detect whitefly-transmitted geminiviruses. Plant Disease, 77, 340–347.
Rojas, M. R., Hagen, C., Lucas, W. J., & Gilbertson, R. L. (2005). Exploiting chinks in the plant’s armor: evolution and emergence of geminiviruses. Annuals Review of Phytopathology, 43, 361–394.
Sanz, A. I., Fraile, A., GarciaArenal, F., Zhou, X. P., Robinson, D. J., Khalid, S., et al. (2000). Multiple infection, recombination and genome relationships among begomovirus isolates found in cotton and other plants in Pakistan. Journal of General Virology, 81, 1839–1849.
Sastry, K. S. M., & Singh, S. J. (1974). Effect of yellow vein mosaic virus infection on growth and yield of okra crop. Indian Phytopathology, 27, 294–297.
Saunders, K., & Stanley, J. (1999). A nanovirus-like component associated with yellow vein disease of Ageratum conyzoides: evidence for interfamilial recombination between plant DNA viruses. Virology, 264, 142–152.
Saunders, K., Bedford, I. D., Briddon, R. W., Markham, P. G., Wong, S. M., & Stanley, J. (2000). A novel virus complex causes Ageratum yellow vein disease. Proceedings of the National Academy of Sciences of the United States of America, 97, 6890–6895.
Schnippenkoetter, W. H., Martin, D. P., Willment, J. A., & Rybicki, E. P. (2001). Forced recombination between distinct strains of Maize streak virus. Journal of General Virology, 82, 3081–3090.
Seal, S. E., Vandenbosch, F., & Jeger, M. J. (2006). Factors influencing begomovirus evolution and their increasing global significance: implications for sustainable control. Critical Reviews in Plant Sciences, 25, 23–46.
Shankarappa, K. S., Rangaswamy, K. T., Aswathanarayana, D. S., Rekha, A. R., Raghavendra, N., Reddy, C. N. L., et al. (2007). Development and optimization of silverleaf assay, protein and nucleic acid based diagnostic techniques for quick and reliable detection and monitoring biotype B of Bemisia tabaci in India. Bulletin of Entomological Research, 97, 503–513.
Sinha, S. N., & Chakrabarthi, A. K. (1978). Effect of yellow vein mosaic virus infection on okra seed production. Seed Research, 6, 67–70.
Stanley, J., Bisaro, D. M., Briddon, R. W., Brown, J. K., Fauquet, C. M., & Harrison, B. D. (Eds.). (2005). Virus taxonomy, VIIIth report of the ICTV (pp. 301–326). London: Elsevier/Academic Press.
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M., & Kumar, S. (2011). MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molecular Biology and Evolution. doi:10.1093/molbev/msr 121.
Varma, A., & Malathi, V. G. (2003). Emerging geminivirus problems: a serious threat to crop production. Annuals of Applied Biology, 142, 145–164.
Venkataravanappa, V., Reddy, C. N. L., Swaranalatha, P., Jalali, S., Briddon, R. W., & Krishna Reddy, M. (2011). Diversity and phylogeography of begomovirus-associated beta satellites of okra in India. Virology Journal, 8, 555. doi:10.1186/1743-422X-8-555.
Venkataravanappa, V., Reddy, C. N. L., Jalali, S., & Krishna Reddy, M. (2012a). Molecular characterization of distinct bipartite begomovirus infecting bhendi (Abelmoschus esculentus L.) in India. Virus Genes, 44, 522–535. doi:10.1007/s11262-012-0732-y.
Venkataravanappa, V., Lakshminarayana Reddy, C. N., Jalali, S., Swarnalatha, C. N., Devaraju, P., et al. (2012b). Molecular evidence for association of Cotton leaf curl Alabad virus with yellow vein mosaic disease of okra in North India. Archives of Phytopathology and Plant Protection, 45, 2095–2113. doi:10.1080/03235408.2012.721682. Corrigendum, 2013. 46, 127.
Zhou, X. P., Liu, Y. L., Calvert, L., Munoz, C., Otim-Nape, G. W., Robinson, D. J., et al. (1997). Evidence that DNA-A of a geminivirus associated with severe cassava mosaic disease in Uganda has arisen by interspecific recombination. Journal of General Virology, 78, 2101–2111.
Acknowledgments
The research was supported by ICAR NETWORK project on development of diagnostics to emerging plant viruses, Indian Council of Agricultural Research, Government of India, New Delhi, India.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Venkataravanappa, V., Reddy, C.N.L., Jalali, S. et al. Molecular characterization of a new species of begomovirus associated with yellow vein mosaic of bhendi (Okra) in Bhubhaneswar, India. Eur J Plant Pathol 136, 811–822 (2013). https://doi.org/10.1007/s10658-013-0209-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-013-0209-4