Abstract
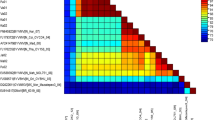
Yellow vein mosaic disease of okra is a whitefly transmitted begomovirus causing heavy economic loss in different parts of India. The okra isolate (OY131) of this virus from a bhendi plant [(Abelmoschus esculentus L.) Moench] showing yellow vein mosaic, vein twisting, reduced leaves, and a bushy appearance in the Palem region, New Delhi, India, was characterized in the present study. The complete DNA-A and DNA-B sequences have been determined and are comprised of 2,746 and 2,703 nucleotides, respectively. The betasatellite (DNA-β) component was absent in the sample. The genome organization was typically of biparite begomoviruses, which were characterized earlier. Comparison of DNA-A component with other known begomoviruses suggest that this virus, being only distantly related (<85.9% similarity with its nearest relative, BYVMV) to other known begomoviruses, is a new species. We have tentatively assigned the genome to a novel geminivirus species Bhendi yellow vein mosaic Delhi virus [BYVDV-IN (India: Delhi: okra)]. DNA-B showed highest sequence identity (87.8% identical) to that of a ToLCNDV (AY158080). The phylogenetic analysis of the present isolate is distinct from all other viruses; however clusters with ToLCNDV group infect different crops. The recombination analysis revealed that this isolate has sequences originated from ToLCNDV. This is the first known bhendi yellow vein mosaic disease associated bipartite begomovirus from India.



Similar content being viewed by others
References
S.G. Lazarowitz, Crit. Rev. Plant Sci. 11, 327–349 (1992)
R.W. Briddon, S. Mansoor, I.D. Bedford, M.S. Pinner, K. Saunders, J. Stanley, Y. Zafar, K. Malik, P.G. Markham, Virology 285, 234–243 (2001)
R.W. Briddon, S.E. Bull, I. Amin, A.M. Idris, S. Mansoor, I.D. Bedford, P. Dhawan, N. Rishi, S.S. Siwatch, A.M. Abdel-Salam, J.K. Brown, Y. Zafar, P.G. Markham, Virology 312, 106–121 (2003)
R.W. Briddon, C.M. Fauquet, J.K. Brown, E. Moriones, J. Stanley, M. Zerbini, X. Zhou, Arch. Virol. 153, 763–781 (2008)
K. Saunders, I.D. Bedford, R.W. Briddon, P.G. Markham, S.M. Wong, J. Stanley, Proc. Natl. Acad. Sci. U S A 97, 6890–6895 (2000)
R. Usha, in Characterization, diagnosis & management of plant viruses, ed. by G.P. Rao, P.L. Kumar, R.L. Holguín-Peña (Studium Press, Houston, 1980), pp. 387–392
S.P. Capoor, P.M. Varma, Indian J. Agric. Sci. 20, 217–230 (1950)
J. Jose, R. Usha, Virology 305, 310–317 (2003)
S.E. Bull, R.W. Briddon, W.S. Sserubombwe, K. Ngugi, P.G. Markham, J. Stanley, J. Gen. Virol. 87, 3053–3065 (2006)
R.V. Chowdareddy, J. Colvin, V. Muniyappa, S.E. Seal, Arch. Virol. 150, 845–867 (2005)
H. Delatte, D.P. Martin, F. Naze, R.W. Golbach, B. Reynaud, M. Peterschmitt, J.M. Lett, J. Gen. Virol. 86, 1533–1542 (2005)
P. Lefeuvre, D.P. Martin, M. Hoareau, F. Naze, H. Delatte, M. Thierry, A. Varsani, N. Becker, B. Reynaud, J.M. Lett, J. Gen. Virol. 88, 3458–3468 (2007)
S. Mansoor, R.W. Briddon, S.E. Bull, I.D. Bedford, A. Bashir, M. Hussain, M. Saeed, M.Y. Zafar, K.A. Malik, C. Fauquet, P.G. Markham, Arch. Virol. 148, 1969–1986 (2003)
F.J. Morales, Adv. Virus Res. 67, 127–162 (2006)
J. Ndunguru, J.P. Legg, T.A.S. Aveling, G. Thompson, C.M. Fauquet, Virol. J. 2, 21 (2005)
A.I. Sanz, A. Fraile, J.M. Gallego, J.M. Malpica, F. Garcia Arenal, J. Mol. Evol. 49, 672–681 (1999)
Y.C. Zhou, M. Nonssourou, Kon, M.R. Rajas, H. Jiang, L.F. Chen, K. Gamby, R. Foster, R.L. Gilbertson, Arch. Virol. 153, 693–706 (2008)
L.C. Berrie, E.P. Rybicki, M.E.C. Rey, J. Gen. Virol. 82, 53–58 (2001)
H. Jeske, M. Lutgemeier, W. Preiss, EMBO J. 20, 6158–6167 (2001)
M. Padidam, S.Y. Sawyer, C.M. Fauquet, Virology 265, 218–225 (1999)
X.P. Zhou, Y.L. Liu, L. Calvert, C. Munoz, G.W. Otim-Nape, D.J. Robinson, B.D. Harrison, J. Gen. Virol. 78, 2101–2111 (1997)
W.H. Schnippenkoetter, D.P. Martin, J.A. Willment, E.P. Rybicki, J. Gen. Virol. 82, 3081–3090 (2001)
J.J. Doyle, J.L. Doyle, Focus 12, 13–15 (1990)
R.W. Briddon, S.E. Bull, S. Mansoor, I. Amin, P.G. Markham, Mol. Biotechnol. 20, 315–318 (2002)
R.E. Boulton, G.J. Jellis, D.C. Balcoumbe, A.M. Squire, in proceedings of the british crop protection conference, British crop protection council, Thorton Heath, surrey, 1984, pp. 177–180
T.A. Hall, Nucl. Acids Symp. Ser. 41, 95–98 (1999)
K. Tamura, D. Peterson, N. Peterson, G. Stecher, M. Nei, S. Kumar, Mol. Biol. Evol. (2011). doi:10.1093/molbev/msr121
D.H. Huson, D. Bryant, Mol. Biol. Evol. 23, 254–267 (2006)
D.P. Martin, P. Lemey, M. Lott, M. Vincent, D. Posada, P. Lefeuvre, Bioinformatics 26(19), 2462–2463 (2010)
C.M. Fauquet, R.W. Briddon, J.K. Brown, E. Moriones, J. Stanley, M. Zerbini, X. Zhou, Arch. Virol. 153, 783–821 (2008)
J.E. Polston, P.L. Anderson, Plant Dis. 81, 1358–1369 (1997)
A. Varma, V.G. Malathi, Ann Appl Biol. 142, 145–164 (2003)
S. Paull, R. Ghosh, S. Chaudhuri, S.K. Ghosh, A. Roy, J. Plant Pathol. 91(3), 637–647 (2009)
V. Muniyappa, in Vectors of Plant Pathogens, ed. by K.F. Harris, K. Maramorosch (Academic Press, New York, 1980), pp. 39–85
S.J. Singh, Indian J. Mycol. Plant Pathol. 10, 35–39 (1980)
R.W. Briddon, J. Stanley, Virology 344, 198–210 (2006)
V. Venkataravanappa, C.N.L. Reddy, P. Swaranalatha, S. Jalali, R.W. Briddon, M.K. Reddy, Virol. J. 8, 855 (2011)
A.P. Graham, D.P. Martin, M.E. Roye, Virus Genes 40, 256–266 (2010)
C.H. Zepeda, M. Ali, Idris, G. Carnevali, J.K. Brown, O.A.M. Valenzuela, Virus Genes 35, 369–377 (2007)
M. Shafiq, S. Asad, Y. Zafar, R.W. Briddon, S. Mansoor, Virol. J. 7, 367 (2010)
P. Kumari, A.K. Singh, B. Chattopadhyay, S. Chakraborty, Virus Res. 152, 19–29 (2010)
Acknowledgments
The research was supported by ICAR NETWORK project on development of diagnostics to emerging plant viruses, Indian Council of Agricultural Research, Government of India, New Delhi. We thank DP Martin, Computational Biology Group, Institute of Infectious Disease and Molecular Medicine, University of Cape Town, South Africa for going through the manuscript and suggesting the corrections.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.

11262_2012_732_MOESM1_ESM.jpg
S1 Detection of BYVDV in the total DNA isolated from naturally infected bhendi samples by dot blot hybridization using nonradioactive digoxginin labelled Probe of betasatellite of Bhendi yellow vein Dehli virus (the dot circle depicts the +ve betasatellite clone). (JPEG 4 kb)
Rights and permissions
About this article
Cite this article
Venkataravanappa, V., Lakshminarayana Reddy, C.N., Jalali, S. et al. Molecular characterization of distinct bipartite begomovirus infecting bhendi (Abelmoschus esculentus L.) in India. Virus Genes 44, 522–535 (2012). https://doi.org/10.1007/s11262-012-0732-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11262-012-0732-y