Abstract
The human sex ratio at birth (SRB) undergoes temporary changes around a mean proportion of 0.51 male births. SRB has been well studied for historical, geographical, and secular trends, but until now not linked to health outcomes in the total population, e.g. for cardiovascular disease (CVD) or mortality during follow-up of birth cohorts. We used linkage analysis based on national registers in Sweden that cover all births from 1900 to 2016. SRB at birth was calculated by every 10-year birth cohort in all survivors living in 1997 for a follow-up analysis of risk of CVD and mortality with data from national registers. When the highest quartile of SRB was used as reference, a slightly increased risk of fatal CVD (HR 1.03 (95% confidence intervals, CI): 1.02–1.04), non-fatal CVD (HR 1.01; 95%CI: 1.01–1.02) and mortality (HR 1.02; 95%CI, 1.01–1.03) was found after full adjustments in men belonging to the lowest SRB quartile. A similar pattern was also found for fatal CHD in women. in the lowest SBR quartile compared to the highest, HR 1.03 (95%CI: 1.02–1.05). In conclusion, in birth cohorts with a relatively lower than expected number of males born, long-term adverse health effects were observed with slightly increased cardiovascular risk and total mortality at the population level. This could indicate that men belonging to so-called “culled cohorts” in a developed country during the 20th century are characterized by a slightly increased risk that could reflect negative early life influences and environmental exposures in pregnant women resulting in selective loss of male embryos or fetuses. In a public health perspective SRB could be of some importance to monitor as an aspect of birth statistics linked to relatively minor population health effects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiovascular disease (CVD) continues to be a major public health problem in western countries, even though age-adjusted trends for their incidence and mortality have been declining in many countries [1]. Despite corresponding age-adjusted declining trends for CVD risk factors, such as mean systolic blood pressure [2], total cholesterol [3], and smoking, the growth of ageing populations means that, in absolute terms, the number of CVD events will remain high.
Besides the well-known major conventional risk factors for CVD like older age, male sex, hypertension, hyperlipidaemia, obesity, diabetes and smoking, an increased interest has focused on the importance of reproductive history for evaluation of CVD risk, both in women [4,5,6] and in men [7]. For example, individual-level reproductive outcomes at birth such as prematurity, growth retardation, and low birth weight have been reported to predict CVD in adult life [8, 9]. Less well studied are population-level reproductive outcomes such as the so-called secondary sex ratio at birth, SRB (as opposed to the primary sex ratio at conception). SRB is a rather robust variable that is simple to measure over time [10]. Previous demographic studies have shown that, in general, the proportion of male births is about 0.51 of all births [11], which is slightly less than the proportion of male embryos that are initially conceived (primary sex ratio), due to unsuccessful implantations or spontaneous abortions [12].
The SRB and its secular trends over time have been a challenge in demographic research for many years [13,14,15] despite the relative simplicity of measuring them. This is because these trends may have been influenced by biological as well as social and cultural factors [16, 17]. As medical technologies evolve, pre-natal diagnosis based on ultrasound and selective abortions have been used to alter the natural SRB because in some countries having a male baby means securing the family name with implications for family economy [18, 19]. The SRB is currently declining in western countries [20, 21], including Sweden [22, 23], but increasing in other parts of the world [24]. However, underlying factors behind these secular trends and whether such trends are independently associated with a changing pattern of cardiovascular events and all-cause mortality are poorly understood.
Based on linkage of extensive national register data from Sweden our primary aim was to examine demographic change of SRB over a period of more than 100 years and its associations with risk of CVD overall, coronary heart disease (CHD), and total mortality, stratified by sex. Secondary aims included the description of SRB in relation to geographical regions and social as well as maternal background characteristics.
Materials and methods
Setting and participants
The dataset used in this study was constructed by linking several national Swedish registers. The Swedish government-owned Statistics Sweden provided the Nationwide Population Register and the Multi-Generation Register (MGR), which include persons born in Sweden in or after 1900 [25]. The MGR consists of data of more than nine million individuals, with information available on mothers in 97% and on fathers in 95% of index persons. Index persons are confined to those born from 1932 onwards and those alive on January 1, 1961 [25].
We calculated the SRB annually for all children born in Sweden from 1900 to 2016. Linkages were made to National Census data, available since 1960, to ascertain individual-level socioeconomic status. The final link was made by adding data from the Swedish Cause of Death Register (1961–2018) and the Swedish Hospital Discharge Register, with recorded dates of hospitalizations and hospital diagnoses since 1964, and almost complete national coverage since 1987, until the end of 2018. National Swedish registers have a high validity for medical research [26], including reproductive epidemiology [27, 28]. For analysing the risk of CVD, CHD, and total mortality in relation to SRB, we collected data from 4.07 million men and 4.14 million women who were born from 1900 to 1997 and still living in 1997, who were then followed up for the study outcomes through 2018.
Patient and public involvement
No participants or patients were involved in setting the research question or in developing plans for the study. The results will be disseminated to patients and the public through a website and/or press releases suitable for a non-specialized audience.
Follow-up of CVD/CHD events and total mortality
We used ICD-codes for non-fatal or fatal CVD (ICD-10, I00-I99) and CHD (ICD-10, I20-I25). Non-fatal events were identified from the National Hospital Discharge Register, and fatal events and total mortality from the Cause of Death Register, until 31st December 2018.
Definitions
Education was based on educational level, which was classified into three categories: ≤ 9 years, 10–11 years, and ≥ 12 years.
Geographic region was divided into large cities (cities with a population of more than 200,000 inhabitants), other/Southern Sweden, and other/Northern Sweden.
Co-morbidities were defined as the first hospitalization during the follow-up period of the following diseases: chronic obstructive pulmonary disease, COPD (both hospitalization and mortality were included) (ICD-9 490–496 and ICD-10 J40-J49), obesity (ICD-9 278 A and ICD-10 E65-E68), alcohol-related liver disease (both hospitalizations and mortality were included) (ICD-9 291, 303, 571 and ICD-10 F10 and K70), diabetes mellitus (both hospitalizations and mortality were included) (ICD-9 250 and ICD-10 E10-E14), and cancer diagnoses (ICD-7 140–209).
SRB was defined as the proportion of all births that were male, categorized in three groups as the lowest quartile, middle 50%, and highest quartile, based on the distribution within each 10-year birth cohort (Supplementary Table).
Statistical methods
Person-years at risk were calculated from the start of follow-up on 1st January 1997 until our outcomes, death from other causes, emigration, or the end of the follow-up on 31st December 2018. Age at baseline was included as a continuous variable in the adjustments. Thus, age-adjusted incidence rates for first hospitalization and mortality were calculated for the entire follow-up period. We used Cox proportional hazards models to calculate the hazard ratio (HR) with 95% confidence intervals (CI) for total (fatal and non-fatal) CVD and CHD event risk, and for total mortality, for both men and women, associated with the calculated SRB. Attained age was used as the underlying timescale to enable comparison between individuals of the same age at follow-up. This was done while adjusting for individual characteristics (family income, marital status, immigrant background and educational level, and region of residence), and co-morbidities (COPD, obesity, alcohol-related liver diseases, diabetes mellitus, and cancer). Individuals in the highest SRB category (i.e., highest quartile of the proportion of male to female births) were used as reference. The proportionality assumptions were checked by plotting the incidence rates over time and by calculating Schoenfeld (partial) residuals and these assumptions were fulfilled. We used SAS version 9.4 (SAS Institute Inc. Cary, NC, USA) for all statistical analyses.
In sensitivity analyses (based on a smaller study sample, N = 6 563 604, with available information), further adjustment was made for maternal age at delivery and total number of children (parity, family size), when the risk for the different outcomes was calculated. This was made because maternal age and parity are interlinked and that having more than 3 children in non-smoking women was associated with relatively less boys born than expected [29].
To assess the potential influence of survivor bias, two different time periods of births (1900 to 1934 and 1935 to 1980) were compared in a second sub-analysis. A p-value < 0.05 was considered statistically significant.
Results
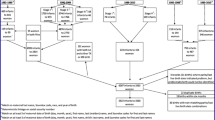
There was a total of 10,081,447 registered births (51.1% men) between 1900 and 1997 (Fig. 1). Among 8,202,688 people still living in 1997, 49.6% were men. From 1997 to 2018, there was a total of 2,716,574 incident CVD events, of which 873,149 were fatal events. Correspondingly there were 463,369 incident CHD events and 340,796 deaths from CHD. Total mortality included 1,802,452 deaths (Table 1).
Almost half of the subjects lived in large cities (49.7%) and had a lower education level, 9 years or less (49.8%) (Supplementary Tables S1 and S2).
A shifting trend appeared for the cumulative rates of non-fatal CVD in men and women by sex ratio (males/all births) at birth 1900–1997 and during the follow-up 1997–2018 (Fig. 2).
Risk of CVD and CHD in men in relation to sex ratio at birth
The risk of non-fatal CVD in men born in those years with the lowest SRB compared to the highest (reference) was slightly but significantly higher, HR 1.01 (95% CI: 1.01–1.02) when adjusted for age, region of residence and educational level, and remained significant HR 1.01 (1.01–1.02) when also adjusted for comorbidities, i.e. hospitalization for COPD, alcoholism, diabetes, obesity, and cancer (Table 2, supplementary Table S5). A similar risk was also noted for men in the middle SBR category after full adjustment, HR 1.01 (1.00-1.01).
A similar pattern was observed for non-fatal CHD in men in the lowest category of SRB with a HR of 1.03 (1.02–1.05) when adjusted for age, region of residence and educational level, and remained significant with a HR of 1.03 (1.02–1.04) after full adjustment for co-morbidities (Table 2, supplementary Table S6). For men in the middle SBR category, the HR was 1.02 (1.01–1.03) after full adjustment.
The risk of fatal CVD in men born in years with the lowest SRB compared to the highest was weakly but significantly higher with a HR of 1.03 (95% CI: 1.02–1.04) when adjusted for age, region of residence, and educational level, and remained significant, HR 1.03 (1.02–1.04), when also adjusted for comorbidities (Table 3, supplementary Table S3). A similar risk was noted for men in the middle SBR category after full adjustment with a HR of 1.03 (1.02–1.03).
The risks of fatal CHD in men born in years with the lowest or middle SBR categories after full adjustment were also modestly higher with HRs of 1.04 (1.03–1.05) and 1.03 (1.02–1.05), respectively (Table 3, supplementary Table S4).
Risk of CVD and CHD in women in relation to sex ratio at birth
The corresponding risk of non-fatal CVD in women with the lowest SRB compared to the highest (reference) was not increased with a HR of 1.00 (95% CI: 1.00–1.01) when adjusted for age, region of residence and educational level, and remained with a HR of 1.00 (1.00–1.00) when also adjusted for comorbidities (Table 2, supplementary Table S9). A similar risk was also noted for women in the middle SBR category after full adjustment, HR 1.00 (0.99-1.00).
For non-fatal CHD in women in the lowest SBR categories, the HRs were 1.03 (1.02–1.05) and 1.02 (1.01–1.04), respectively (Table 2, supplementary Table S10). The risk in women in the middle SBR category was similar after full adjustment with a HR of 1.03 (1.01–1.04).
The risk of fatal CVD in women born in years with the lowest SRB category compared to the highest was weakly but significantly higher with a HR of 1.01 (95% CI: 1.00-1.02) when adjusted for age, region of residence and educational level, and remained significant, HR 1.01 (1.00-1.02) when also adjusted for comorbidities (Table 3, supplementary Table S7). For women in the middle SBR category the risk was similar after full adjustment with a HR of 1.02 (1.01–1.03).
Weakly increased risks were also observed for fatal CHD in women in the lowest SBR category with HRs of 1.03 (1.02–1.05) and 1.03 (1.02–1.05) after full adjustment, respectively (Table 3, supplementary Table S8). For women in the middle SBR category the risk was similar after full adjustment with HR 1.04 (1.03–1.06).
Risk of all-cause mortality in men and women in relation to sex ratio at birth
Men born in years with the lowest SRB category had slightly increased total mortality compared to the highest SRB category (HR, 1.02; 95% CI, 1.01–1.03) after full adjustment, as had men born in the middle SRB category, HR 1.02 (1.01–1.02). Women born in years with the lowest or middle SRB category had almost no increased total mortality risk compared to the highest SRB category (HR, 1.00; 95% CI, 1.00-1.01; and 1.01; 1.01–1.02, respectively) (Table 3, supplementary Tables S11 and S12).
Sensitivity analyses after adjustment for maternal age at birth and family size
In a subsample based on 6,563,604 births, a further adjustment was made for family size (number of children) and maternal age at birth. The risk of non-fatal CVD in both men and women in the lowest SRB category compared to the highest was unchanged (HR, 1.01; 95% CI: 1.00-1.01). The corresponding risk for non-fatal CHD was, however, significantly lower in men (HR, 0.95; 95% CI, 0.94–0.97) and in women (0.93; 0.91–0.95) (supplementary Table S13).
For fatal CVD in men born in years with the lowest SRB compared to the highest the HR was significantly lower at 0.93 (95% CI: 0.92–0.95) and for women the HR was 0.96 (0.94–0.99). The corresponding risk for fatal CHD was lower in both men with a HR of 0.92 (0.90–0.94) and in women with a HR of 0.91 (0.87–0.95).
Finally, the corresponding risk for total mortality in men in the lowest SBR category was significantly lower with a HR of 0.98 (0.97–0.99), but non-significant in women with a HR of 1.01 (0.99–1.02) (supplementary Table S14).
Sensitivity analyses in relation to period and gender
In a further sensitivity analysis (Table S15), it was shown that cardiovascular outcomes were similar between two separate time periods comparing children born 1900–1934 with children born 1935–1980, except for total mortality, with a higher risk observed in men during the first period RR 1.12 (95% CI: 1.11–1.13) compared to the second period, RR 1.04 (1.02–1.05), (Table S16). The risk ratio for men versus women was higher for all fatal outcomes (Table S17).
Discussion
The most important finding of this register-based study that spanned over a period of almost 100 years in Sweden was the association between a lower SRB (i.e. fewer boys born than expected) and slightly increased risks of cardiovascular and coronary events (non-fatal and fatal) in both men and women during the follow-up and after adjustment for several background factors including comorbidities. However, for total mortality, the risk was higher in men but almost unchanged in women belonging to the same lowest SBR category. These findings are observational and admittedly quite weak. Their underlying explanation cannot be conclusively determined; thus, they are primarily hypothesis-generating. In sensitivity analyses, some of these findings became reduced or even reversed (i.e. showing lower, not higher risk associated with lower SRB) after adjustment for maternal age at delivery and parity status. This could imply that these maternal factors are associated with SRB in a way that increases the risk associated with low SRB. For example, during periods of adverse environmental influences, such as financial recession, unemployment, or shortage of material resources, women may both postpone childbearing until later during their reproductive life (older age), and have fewer children, thereby affecting parity status [30, 31].
Furthermore, two time periods were compared as well as the relative risk for men and women. The findings indicated a similar risk for cardiovascular events during the two periods, but a higher mortality risk in men from the lowest sex ratio at birth category during the first period (1900–1934) than during the second period (1935–1980). In general, the relative risk for fatal events in men was higher than that for women during both periods.
During this long period Sweden underwent a transition from a country dominated by agriculture and a larger part of the population living in rural areas to a highly developed industrialised country with most people living in urban areas. Important social changes took place with women entering the labour market in the 1960s and family size gradually decreasing with a lower number of children in each family. Other time trends include the demographic changes with increasing lifespan and more elderly people surviving into age ranges when cardiovascular and coronary disease become more prevalent.
No previous study, to the best of our knowledge, has addressed the same research question on trends for associations between SRB and cardiovascular outcomes as well as total mortality in both sexes following adjustment for several background factors. However, the association between SRB oscillations and longevity in men has previously been studied in some historical studies from the Nordic area [32,33,34]. These studies have indicated that adverse exogenous influences during gestation such as temperature changes or stress exposure could cause an increased abortion rate of the weaker male foetuses (“culled birth cohorts”) resulting in a lower SRB [32,33,34]. On the other hand, the male babies belonging to these “culled birth cohorts” during mostly pre-industrial historical periods (before 1913) who had less cardiovascular morbidity and mortality were selected for survival and, in addition, also had a longer life expectancy in unadjusted analyses, probably due to selection bias [33]. Catalano et al. suggested that oscillation of SRB appears to be due to social processes rather than heritable mechanisms [34]. This agrees with a modern study from Sweden reporting that environmental factors are more important determinants of SRB than genetic factors [17]. Another indication of environmental influences of SRB are studies in immigrant populations showing variation of SRB in relation to parity, country of origin, and time trends [35]. Finally, parental occupation has also been linked to various patterns of SRB [36, 37]. Taken together, these studies emphasise the importance of environmental factors for trends in SRB (Trivers-Willard Hypothesis) [38] acting through mechanisms during gestation including lack of proper implantation of male embryos or spontaneous abortions of male foetuses during adverse conditions related to external exposures during pregnancy. This might be of importance for cardiovascular risk in the offspring as maternal risk factors such as pre-pregnancy hypertension could influence offspring risk of hypertension, which also has been associated with a variation in SRB [39].
Strengths of the study include complete nationwide coverage from 1997 for outcomes in a country with high standards of diagnosis, and with diagnoses often being made by clinical specialists. Another important strength of our study is that it was based on nationwide registers and thus free of recall bias. The Swedish MGR and the Swedish Hospital Discharge Register are validated data sources that have been proven to be reliable in studies of many diseases [25, 26]. Data in our dataset are almost 100% complete [25] and were used for a previous study of ours evaluating sibling rank in relation to cardiovascular risk and total mortality [28].
A potential limitation is the possibility of survivor bias in people who lived until the beginning of follow-up in 1997. However, a sensitivity analysis yielded similar risk estimates in people born before or after 1935, suggesting that survivor bias did not strongly influence the main findings. Another limitation is that we had no access to data on individual lifestyle-related factors such as body mass index, smoking, or diet, because it is impossible to gather such data for an entire national population. However, we did adjust for socioeconomic status, obesity, diabetes, COPD, and cancer as well as alcoholism and related liver disorders, which are associated with lifestyle factors. No data reflecting psychology or personality traits were available to us.
Future research should be directed to find and characterize biological or environmental/social mechanisms linking low SRB to a higher individual risk of CVD, CHD, and mortality [40], as suggested by our observational findings. SRB has been shown to be lowered by several environmental factors and stressors, such as terrorist attacks during pregnancy [41], different catastrophes [42], and the Covid-19 pandemic [43]. This lends further evidence for the importance of the developmental origin of adult health (DoHAD) hypothesis with its many applications in preventive cardiology [44]. These associations could be further studied in other high-quality registers that can be linked by personal ID number in the Nordic countries where secular time trends of SRB during the 20th century have been well described [45].
Conclusion
Based on national registers covering more than a century in Sweden we have shown that a lower SRB, i.e., fewer males than expected being born during some periods (“culled cohorts”), is associated with a slightly increased cardiovascular and coronary risk during follow-up, especially for men. For total mortality, the risk was higher in men, but unchanged in women, for those born during years with a lower SRB. This points to the influence of still undefined environmental factors of importance during gestation that regulate male survival, i.e. lack of implantation of male embryos or spontaneous abortion of male foetuses, with its further implications for reduced longevity in men and increased risk of cardiovascular disease. In addition, maternal characteristics such as age at pregnancy and parity seem to be of importance but could also be influenced by adverse environmental factors during periods of financial recession and lack of resources. Although the observed effects were very weak in general, they could still be of interest at the population level. In a public health perspective, SRB could be of some importance to monitor as an aspect of birth statistics linked to relatively minor population health effects.
Data availability
No additional data are available. The manuscript guarantors affirm that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted.
Abbreviations
- CHD:
-
Coronary Heart Disease
- COPD:
-
Chronic Obstructive Pulmonary Disease
- CVD:
-
Cardiovascular disease
- DOHaD:
-
Developmental Origins of Health and Disease
- SRB:
-
Sex Ratio at Birth
References
Timmis A, Townsend N, Gale CP, Torbica A, Lettino M, Petersen SE, Mossialos EA, Maggioni AP, Kazakiewicz D, May HT, De Smedt D, Flather M, Zuhlke L, Beltrame JF, Huculeci R, Tavazzi L, Hindricks G, Bax J, Casadei B, Achenbach S, Wright L, Vardas P. European Society of Cardiology. European Society of Cardiology: Cardiovascular Disease Statistics 2019. Eur Heart J. 2020; 41(1):12–85. Erratum in: Eur Heart J. 2020;41(47):4507.
Danaei G, Finucane MM, Lin JK, Singh GM, Paciorek CJ, Cowan MJ, Farzadfar F, Stevens GA, Lim SS, Riley LM, Ezzati M, Global Burden of Metabolic Risk Factors of Chronic Diseases Collaborating Group (Blood Pressure). National, regional, and global trends in systolic blood pressure since 1980: systematic analysis of health examination surveys and epidemiological studies with 786 country-years and 5·4 million participants. Lancet. 2011;377(9765):568–77.
Farzadfar F, Finucane MM, Danaei G, Pelizzari PM, Cowan MJ, Paciorek CJ, Singh GM, Lin JK, Stevens GA, Riley LM, Ezzati M, Global Burden of Metabolic Risk Factors of Chronic Diseases Collaborating Group (Cholesterol). National, regional, and global trends in serum total cholesterol since 1980: systematic analysis of health examination surveys and epidemiological studies with 321 country-years and 3·0 million participants. Lancet. 2011;377(9765):578–86.
Okoth K, Chandan JS, Marshall T, Thangaratinam S, Thomas GN, Nirantharakumar K, Adderley NJ. Association between the reproductive health of young women and cardiovascular disease in later life: umbrella review. BMJ 2020;371:m3502. Erratum in: BMJ 2020;371:m3963.
Carlqvist E, Johnson L, Nilsson PM. Shorter reproductive life span is associated with increased cardiovascular risk and total mortality in Swedish women from an observational, population-based study. Maturitas. 2022;164:69–75.
Crump C, Sundquist J, McLaughlin MA, Dolan SM, Govindarajulu U, Sieh W, Sundquist K. Adverse pregnancy outcomes and long-term risk of ischemic heart disease in mothers: national cohort and co-sibling study. BMJ. 2023;380:e072112.
Elenkov A, Melander O, Nilsson PM, Zhang H, Giwercman A. Impact of genetic risk score on the association between male childlessness and cardiovascular disease and mortality. Sci Rep. 2021;11(1):18526.
Mohseni R, Mohammed SH, Safabakhsh M, Mohseni F, Monfared ZS, Seyyedi J, Mejareh ZN, Alizadeh S. Birth Weight and Risk of Cardiovascular Disease incidence in Adulthood: a dose-response Meta-analysis. Curr Atheroscler Rep. 2020;22(3):12.
Crump C. Preterm birth and mortality in adulthood: a systematic review. J Perinatol. 2020;40(6):833–43.
van Zoest M, Bijker EM, Kortmann BBM, Kempers M, van Herwaarden AE, van der Velden J. Claahsen-Van Der Grinten HL. Sex Assignment and Diagnostics in infants with ambiguous genitalia - A single-Center Retrospective Study. Sex Devel. 2019;13(3):109–17.
Ein-Mor E, Mankuta D, Hochner-Celnikier D, Hurwitz A, Haimov-Kochman R. Sex ratio is remarkably constant. Fertil Steril. 2010;93:1961–5.
James WH, Grech V. The human sex ratio at conception. Early Hum Dev. 2020;140:104862.
Gellatly C. The sex ratio: a Biological and Statistical Conundrum. Curr Biol. 2020;30(20):R1261–3.
Long Y, Chen Q, Larsson H, Rzhetsky A. Observable variations in human sex ratio at birth. PLoS Comput Biol. 2021;17(12):e1009586.
D’Alfonso A, Ludovisi M, Palermo P, Serva A, Polsinelli V, Bruno M, Tiberi A, Tabacco S, DI Florio C, Guido M. Sex ratio at birth: causes of variation and narrative review of literature. Minerva Obstet Gynecol 2022 Mar 2. https://doi.org/10.23736/S2724-606X.22.05054-0. Epub ahead of print.
Kolk M, Schnettler S. Socioeconomic status and sex ratios at birth in Sweden: no evidence for a Trivers-Willard effect for a wide range of status indicators. Am J Hum Biol. 2016;28(1):67–73.
Zietsch BP, Walum H, Lichtenstein P, Verweij KJH, Kuja-Halkola R. No genetic contribution to variation in human offspring sex ratio: a total population study of 4.7 million births. Proc Biol Sci. 2020;287(1921):20192849.
Hesketh T, Xing ZW. Abnormal sex ratios in human populations: causes and consequences. Proc Natl Acad Sci U S A. 2006;103:13271–5.
Das Gupta M. Family systems, political systems, and Asia’s missing girls Policy Research Working Paper 5148. The World Bank: Development Research Group: Human Development and Public Services Team. 2009.
Grech V, Vassallo-Agius P, Savona-Ventura C. Secular trends in sex ratios at birth in North America and Europe over the second half of the 20th century. J Epidemiol Community Health. 2003;57:612–5.
Davis DL, Gottlieb MB, Stampnitzky JR. Reduced ratio of male to female births in several industrial countries: a sentinel health indicator? JAMA. 1998;279:1018–23.
Grech V. Sex ratios at birth in Scandinavia over the past sixty years. Scand J Public Health. 2012;40:761–4.
Wu H. Maternal stress and sex ratio at birth in Sweden over two and a half centuries: a retest of the trivers-Willard hypothesis. Hum Reprod. 2021;36(10):2782–92.
Parazzini F, La Vecchia C, Levi F, Franceschi S. Trends in male: female ratio among newborn infants in 29 countries from five continents. Hum Reprod. 1998;13:1394–6.
Ekbom A. The Swedish Multi-generation Register. Methods Mol Biol. 2011;675:215–20.
Ludvigsson JF, Andersson E, Ekbom A, et al. External review and validation of the Swedish national inpatient register. BMC Public Health. 2011;11:450.
Nilsson PM, Li X, Sundquist J, Sundquist K. Maternal cardiovascular disease risk in relation to the number of offspring born small for gestational age: national, multi-generational study of 2.7 million births. Acta Paediatr. 2009;98:985–9.
Nilsson PM, Sundquist J, Sundquist K, Li X. Sibling rank and sibling number in relation to cardiovascular disease and mortality risk: a nationwide cohort study. BMJ Open. 2021;11(6):e042881.
Beratis NG, Asimacopoulou A, Varvarigou A. Association of secondary sex ratio with smoking and parity. Fertil Steril. 2008;89(3):662–7.
Neels K, Theunynck Z, Wood J. Economic recession and first births in Europe: recession-induced postponement and recuperation of fertility in 14 European countries between 1970 and 2005. Int J Public Health. 2013;58(1):43–55.
Matysiak A, Sobotka T, Vignoli D. The great recession and fertility in Europe: a sub-national analysis. Eur J Popul. 2020;37(1):29–64.
Catalano R, Bruckner T. Male lifespan and the secondary sex ratio. Am J Hum Biol. 2006;18(6):783–90.
Bruckner T, Catalano R. The sex ratio and age-specific male mortality: evidence for culling in utero. Am J Hum Biol. 2007;19(6):763–73.
Catalano R, Casey JA, Bruckner TA. A test of oscillation in the human secondary sex ratio. Evol Med Public Health. 2020; 2020(1):225–233.
Mussino E, Miranda V, Ma L. Changes in sex ratio at birth among immigrant groups in Sweden. Genus. 2018;74(1):13.
Magnuson A, Bodin L, Montgomery SM. Father’s occupation and sex ratio of offspring. Scand J Public Health. 2007;35(5):454–9.
Ruckstuhl KE, Colijn GP, Amiot V, Vinish E. Mother’s occupation and sex ratio at birth. BMC Public Health. 2010;10:269.
Veller C, Haig D, Nowak MA. The trivers-Willard hypothesis: sex ratio or investment? Proc Biol Sci. 2016;283:20160126.
Grech V. Pregravid hypertension may have different secondary sex ratio effects in different races in the United States. Int J Cardiol. 2017;248:333–5.
Grech V. Correlation of sex ratio at birth with health and socioeconomic indicators. Early Hum Dev. 2018;118:22–4.
Grech V, Zammit D. A review of terrorism and its reduction of the gender ratio at birth after seasonal adjustment. Early Hum Dev. 2017;115:2–8.
D’Alfonso A, Ludovisi M, Palermo P, Serva A, Polsinelli V, Bruno M, Tiberi A, Tabacco S, DI Florio C, Guido M. Sex ratio at birth: causes of variation and narrative review of literature. Minerva Obstet Gynecol. 2023;75(2):189–200.
Masukume G, Ryan M, Masukume R, Zammit D, Grech V, Mapanga W, Inoue Y. COVID-19 induced birth sex ratio changes in England and Wales. PeerJ. 2023;11:e14618.
Arima Y, Fukuoka H. Developmental origins of health and disease theory in cardiology. J Cardiol. 2020;76(1):14–7.
Fellman J. Temporal and regional trends in the secondary sex ratio: the nordic experience. Early Hum Dev. 2015;91(12):801–6.
Funding
This work was supported by grants to Kristina Sundquist from The Swedish Research Council; the Swedish Heart Lung Foundation, ALF funding from Region Skåne and the National Heart, Lung, and Blood Institute of the National Institutes of Health [R01 HL139536 to C.C. and K.S.]
Open access funding provided by Lund University.
Author information
Authors and Affiliations
Contributions
PMN and XL conceptualized the study design and interpreted the results. XL analysed the data and PMN drafted the manuscript. KS and CC provided the methodological suggestions. XL managed the register data. PMN, XL, KS and CC revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Regional Ethics Review Board at the Lund University, Sweden (ID number 2012/795; approved 2013-02-06).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nilsson, P.M., Sundquist, K., Sundquist, J. et al. Sex ratio at birth across 100 years in Sweden and risk of cardiovascular disease and all-cause mortality – a national register study. Eur J Epidemiol (2024). https://doi.org/10.1007/s10654-024-01137-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10654-024-01137-1