Abstract
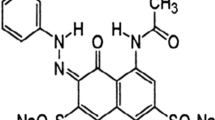
The present study is an attempt to investigate the potentiality of Rhizoclonium hieroglyphicum in the removal of reactive red 239 (RR239) from aqueous solution and to assess the toxicity of the treated dye solution. Optimisation of the process variables namely dye and biosorbent concentrations, pH, temperature and incubation time for RR239 removal was performed using Response Surface Methodology (RSM) assisted Box Behnken Design (BBD) model. The recycling and regeneration efficiency of the dye adsorbed alga was evaluated using different eluents under optimized conditions. Further to understand the adsorption mechanism, isotherms, kinetics and thermodynamic studies were performed. UV–vis and FT-IR spectroscopy was employed to confirm the interaction between the adsorbate and biosorbent. The nature of the treated dye solution was assessed using phyto, microbial and brine shrimp toxicity studies. On the basis of quadratic polynomial equation and response surfaces given by RSM, 90% decolorization of RR239 was recorded at room temperature under specified optimal conditions (300 mg/L of dye, 500 mg/L of biosorbent, pH 8 and 72 h of contact time). Desorption experiments demonstrated 88% of RR239 recovery using 0.1 N acetic acid as an eluent and 81% of dye removal in regeneration studies. The data closely aligned with Freundlich isotherm (R2 − 0.98) and pseudo-second-order kinetic model (R2 − 0.9671). Thermodynamic analysis revealed that the process of adsorption was endothermic, spontaneous, and favorable. UV–Vis and FT-IR analyses provided evidence for adsorbate–biosorbent interaction, substantiating the process of decolorization. In addition, the results of phyto, microbial and brine shrimp toxicity assays consistently confirmed the non-toxic nature of the treated dye. Thus, the study demonstrated that R. hieroglyphicum can act as a potent bioremediation agent in alleviating the environmental repercussions of textile dyeing processes.












Similar content being viewed by others
Avaliability of data and material
All data generated or analyzed during this study are included in this published article.
References
Abdallah, R., & Taha, S. (2012). Biosorption of methylene blue from aqueous solution by nonviable Aspergillus fumigatus. Chemical Engineering Journal, 195, 69–76. https://doi.org/10.1016/j.cej.2012.04.066
Abdelfattah, A., Ali, S. S., Ramadan, H., El-Aswar, E. I., Eltawab, R., Ho, S. H., Elsamahy, T., Li, S., El-Sheekh, M. M., Schagerl, M., Kornaros, M., & Sun, J. (2022). Microalgae-based wastewater treatment: Mechanisms, challenges, recent advances, and future prospects. Environmental Science & Ecotechnology. https://doi.org/10.1016/j.ese.2022.100205
Abdulhameed, A. S., Hum, N. N. M. F., Rangabhashiyam, S., Jawad, A. H., Wilson, L. D., Yaseen, Z. M., Al-Kahtani, A. A., & ALOthman, Z. A. (2021). Statistical modeling and mechanistic pathway for methylene blue dye removal by high surface area and mesoporous grass-based activated carbon using K2CO3 activator. Journal of Environmental Chemical Engineering, 9(4), 105530. https://doi.org/10.1016/j.jece.2021.105530
Abdulhameed, A. S., Mohammad, A. T., & Jawad, A. H. (2019). Modeling and mechanism of reactive orange 16 dye adsorption by chitosan-glyoxal/TiO2 nanocomposite: Application of response surface methodology. Desalination and Water Treatment, 164, 346–360. https://doi.org/10.5004/dwt.2019.24384
Abou Oualid, H., Abdellaoui, Y., Laabd, M., El Ouardi, M., Brahmi, Y., Iazza, M., & Abou Oualid, J. (2020). Eco-efficient green seaweed Codium decorticatum biosorbent for textile dyes: Characterization, mechanism, recyclability, and RSM optimization. ACS Omega, 5(35), 22192–22207. https://doi.org/10.1021/acsomega.0c02311
Akar, S. T., Akar, T. A. M. E. R., & Çabuk, A. (2009). Decolorization of a textile dye, reactive red 198 (RR198), by Aspergillus parasiticus fungal biosorbent. Brazilian Journal of Chemical Engineering, 26, 399–405. https://doi.org/10.1590/S0104-66322009000200018
Al-Ansari, M. M., Li, Z., Masood, A., & Rajaselvam, J. (2022). Decolourization of azo dye using a batch bioreactor by an indigenous bacterium Enterobacter aerogenes ES014 from the waste water dye effluent and toxicity analysis. Environmental Research, 205, 112189. https://doi.org/10.1016/j.envres.2021.112189
Al-Fawwaz, A. T., Al Shra’ah, A., & Elhaddad, E. (2023). Bioremoval of methylene blue from aqueous solutions by Green Algae (Bracteacoccus sp.) isolated from North Jordan: Optimization, Kinetic, and Isotherm studies. Sustainability, 15(1), 842. https://doi.org/10.3390/su15010842
Amit, & Ghosh, U. K. (2018). An approach for phycoremediation of different wastewaters and biodiesel production using microalgae. Environmental Science and Pollution Research, 25, 18673–18681. https://doi.org/10.1007/s11356-018-1967-5
Anderson, M. J., & Whitcomb, P. J. (2016). RSM simplified: Optimizing processes using response surface methods for design of experiments. Productivity press.
Asgher, M., & Bhatti, H. N. (2012). Evaluation of thermodynamics and effect of chemical treatments on sorption potential of Citrus waste biomass for removal of anionic dyes from aqueous solutions. Ecological Engineering, 38(1), 79–85. https://doi.org/10.1016/j.ecoleng.2011.10.004
Baek, M. H., Ijagbemi, C. O., Se-Jin, O., & Kim, D. S. (2010). Removal of Malachite Green from aqueous solution using degreased coffee bean. Journal of Hazardous Materials, 176(1–3), 820–828. https://doi.org/10.3390/su15010842
Balaram, V., Copia, L., Kumar, U. S., Miller, J., & Chidambaram, S. (2023). Pollution of water resources and application of ICP-MS techniques for monitoring and management—A comprehensive review. Geosystems and Geo Environment. https://doi.org/10.1016/j.geogeo.2023.100210
Banaei, A., Yaychi, M. F., Karimi, S., Vojoudi, H., Namazi, H., Badiei, A., & Pourbasheer, E. (2018). 2, 2’-(butane-1, 4-diylbis (oxy)) dibenzaldehyde cross-linked magnetic chitosan nanoparticles as a new adsorbent for the removal of reactive red 239 from aqueous solutions. Materials Chemistry and Physics, 212, 1–11. https://doi.org/10.1016/j.matchemphys.2018.02.036
Bapat, S., Jaspal, D., & Malviya, A. (2021). Efficacy of Parthenium hysterophorus waste biomass compared with activated charcoal for the removal of CI reactive red 239 textile dye from wastewater. Coloration Technology, 137(3), 234–250.
Barathi, S., Aruljothi, K. N., Karthik, C., & Padikasan, I. A. (2020). Optimization for enhanced ecofriendly decolorization and detoxification of Reactive Blue160 textile dye by Bacillus subtilis. Biotechnology Reports, 28, e00522. https://doi.org/10.1016/j.btre.2020.e00522
Barathi, S., Aruljothi, K. N., Karthik, C., Padikasan, I. A., & Ashokkumar, V. (2022). Biofilm mediated decolorization and degradation of reactive red 170 dye by the bacterial consortium isolated from the dyeing industry wastewater sediments. Chemosphere, 286, 131914. https://doi.org/10.1016/j.chemosphere.2021.131914
Bewley, J. D., & Black, M. (1994). Seeds physiology of development and germination (2nd ed.). Plenum Press.
Bhosale, A. H., & Khobragade, R. M. (2019). Biodecolourization and biodegradation of textile azo dye Golden Yellow HER by Pseudomonas sp. GYH-1. Research Journal of Science and Technology, 11(4), 246–258. https://doi.org/10.5958/2349-2988.2019.00035.4
Bilal, M., & Asgher, M. (2015). Dye decolorization and detoxification potential of Ca-alginate beads immobilized manganese peroxidase. BMC Biotechnology, 15, 1–14. https://doi.org/10.1186/s13065-015-0125-0
Bilal, M., Lam, S. S., & Iqbal, H. M. (2022). Biocatalytic remediation of pharmaceutically active micropollutants for environmental sustainability. Environmental Pollution, 293, 118582. https://doi.org/10.1016/j.envpol.2021.118582
Bliding, C. (1957). Studies on Rhizoclonium. I. Life history of two species. Botaniska Notiser, 110, 271–275.
Chapman, V. J. (1956). The marine algae of New Zealand. Part I. Myxophyceae and Chlorophyceae. Botanical Journal of the Linnean Society, 55, 333–501.
Chen, X. (2015). Modeling of experimental adsorption isotherm data. Information, 6(1), 14–22.
Cossolin, A. S, Reis, H. C. O. D., Castro, K. C. D., Santos, B. A. P. D., Marques, M. Z., Parizotto, C. A., & Morais, E. B. D. (2019). Decolorization of textile azo dye reactive red 239 by the novel strain Shewanella xiamenensis G5-03 isolated from contaminated soil. Revista Ambiente e Agua, 14.
Dai, L., Zhu, W., He, L., Tan, F., Zhu, N., Zhou, Q., He, M., & Hu, G. (2018). Calcium-rich biochar from crab shell: An unexpected super adsorbent for dye removal. Bioresource Technology, 267, 510–516. https://doi.org/10.1016/j.biortech.2018.07.090
Daneshvar, E., Vazirzadeh, A., Niazi, A., Kousha, M., Naushad, M., & Bhatnagar, A. (2017). Desorption of methylene blue dye from brown macroalga: Effects of operating parameters, isotherm study and kinetic modeling. Journal of Cleaner Production, 152, 443–453. https://doi.org/10.1016/j.jclepro.2017.03.119
Daneshvar, N., Ayazloo, M., Khataee, A. R., & Pourhassan, M. (2007a). Biological decolorization of dye solution containing Malachite Green by microalgae Cosmarium sp. Bioresource Technology, 98(6), 1176–1182. https://doi.org/10.1016/j.biortech.2006.05.025
Daneshvar, N., Khataee, A. R., Rasoulifard, M. H., & Pourhassan, M. (2007b). Biodegradation of dye solution containing Malachite Green: Optimization of effective parameters using Taguchi method. Journal of Hazardous Materials, 143(1–2), 214–219.
Dias, N. C., Bassin, J. P., Sant’Anna, G. L., Jr., & Dezotti, M. (2019). Ozonation of the dye reactive red 239 and biodegradation of ozonation products in a moving-bed biofilm reactor: Revealing reaction products and degradation pathways. International Biodeterioration and Biodegradation, 144, 104742. https://doi.org/10.1016/j.ibiod.2019.104742
Doğar, Ç., Gürses, A., Açıkyıldız, M., & Özkan, E. (2010). Thermodynamics and kinetic studies of biosorption of a basic dye from aqueous solution using green algae Ulothrix sp. Colloids and Surfaces B: Biointerfaces, 76(1), 279–285. https://doi.org/10.1016/j.colsurfb.2009.11.004
Donmez, G., & Aksu, Z. (2002). Removal of chromium (VI) from saline wastewaters by Dunaliella species. Process Biochemistry, 38(5), 751–762.
El-Agawany, N. I., Abdel-Razik, S. A., Abdel-Kareem, M. S., & Kaamoush, M. I. (2023). Environmental performance of two marine algae Ulva fasciata and Pterocladia capillacea in the biological treatment of four reactive dyes from aqueous solutions based on fresh and dried biomass. Applied Water Science, 13(3), 82. https://doi.org/10.1007/s13201-023-01893-7
Eshghi, S., & Kashi, F. J. (2022). Bacterial synthesis of magnetic Fe3O4 nanoparticles: Decolorization acid red 88 using FeNPs/Ca-Alg beads. Arabian Journal of Chemistry, 15(9), 104032. https://doi.org/10.1016/j.arabjc.2022.104032
Freundlich, H. (1907). Absorption in solution. Physikalische. Chemie, 57, 384–470.
Garcia, V. S., Rosa, J. M., & Borrely, S. I. (2020). Toxicity and color reduction of a textile effluent containing reactive red 239 dye by electron beam irradiation. Radiation Physics and Chemistry, 172, 108765. https://doi.org/10.1016/j.radphyschem.2020.108765
Ghanadzadeh, A., Zeini, A., Kashef, A., & Moghadam, M. (2008). Concentration effect on the absorption spectra of oxazine1 and methylene blue in aqueous and alcoholic solutions. Journal of Molecular Liquids, 138(1–3), 100–106. https://doi.org/10.1016/j.molliq.2007.09.005
Ghasemi, J., & Asadpour, S. (2007). Thermodynamics’ study of the adsorption process of methylene blue on activated carbon at different ionic strengths. The Journal of Chemical Thermodynamics, 39(6), 967–971. https://doi.org/10.1016/j.jct.2006.10.018
Goswami, R. K., Mehariya, S., Verma, P., Lavecchia, R., & Zuorro, A. (2021). Microalgae-based biorefineries for sustainable resource recovery from wastewater. Journal of Water Process Engineering, 40, 101747. https://doi.org/10.1016/j.jwpe.2020.101747
Gupta, V. K. (2009). Application of low-cost adsorbents for dye removal—a review. Journal of Environmental Management, 90(8), 2313–2342. https://doi.org/10.1016/j.jenvman.2008.11.017
Gurav, R., Bhatia, S. K., Choi, T. R., Choi, Y. K., Kim, H. J., Song, H. S., Lee, S. M., Park, S. L., Lee, H. S., Koh, J., Jeon, J. M., Yoon, J. J., & Yang, Y. H. (2021). Application of macroalgal biomass derived biochar and bioelectrochemical system with Shewanella for the adsorptive removal and biodegradation of toxic azo dye. Chemosphere, 264, 128539. https://doi.org/10.1016/j.chemosphere.2020.128539
Hambisa, A. A., Regasa, M. B., Ejigu, H. G., & Senbeto, C. B. (2023). Adsorption studies of methyl orange dye removal from aqueous solution using Anchote peel-based agricultural waste adsorbent. Applied Water Science, 13(1), 24. https://doi.org/10.1007/s13201-022-01832-y
Hamouda, R. A., El-Naggar, N. E., & Abou-El-Souod, G. W. (2022). Simultaneous bioremediation of Disperse orange-2RL Azo dye and fatty acids production by Scenedesmus obliquus cultured under mixotrophic and heterotrophic conditions. Scientific Reports, 12, 20768. https://doi.org/10.1038/s41598-022-22825-6
Hasanpour, M., Motahari, S., Jing, D., & Hatami, M. (2021). Statistical analysis and optimization of photodegradation efficiency of methyl orange from aqueous solution using cellulose/zinc oxide hybrid aerogel by response surface methodology (RSM). Arabian Journal of Chemistry, 14(11), 103401. https://doi.org/10.1016/j.arabjc.2021.103401
Hernández-Zamora, M., Cristiani-Urbina, E., Martínez-Jerónimo, F., Perales-Vela, H. V., Ponce-Noyola, T., Montes-Horcasitas, M. D. C., & Cañizares-Villanueva, R. O. (2015). Bioremoval of the azo dye Congo Red by the microalga Chlorella vulgaris. Environmental Science and Pollution Research, 22, 10811–10823. https://doi.org/10.1007/s11356-015-4277-1
Ho, Y. S., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34(5), 451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Huiping, L., Guoqun, Z., Shanting, N., & Yiguo, L. (2007). Technologic parameter optimization of gas quenching process using response surface method. Computational Materials Science, 38(4), 561–570. https://doi.org/10.1016/j.commatsci.2006.03.014
Indah, S., Helard, D., & Binuwara, A. (2018). Studies on desorption and regeneration of natural pumice for iron removal from aqueous solution. Water Science and Technology, 2017(2), 509–515. https://doi.org/10.2166/wst.2018.177
Jawad, A. H., Bardhan, M., Islam, M. A., Islam, M. A., Syed-Hassan, S. S. A., Surip, S. N., Alothman, Z. A., & Khan, M. R. (2020). Insights into the modeling, characterization and adsorption performance of mesoporous activated carbon from corn cob residue via microwave-assisted H3PO4 activation. Surfaces and Interfaces, 21, 100688. https://doi.org/10.1016/j.surfin.2020.100688
Jawad, A. H., Rashid, R. A., Mahmuod, R. M., Ishak, M. A. M., Kasim, N. N., & Ismail, K. (2016). Adsorption of methylene blue onto coconut (Cocos nucifera) leaf: Optimization, isotherm and kinetic studies. Desalination and Water Treatment, 57(19), 8839–8853. https://doi.org/10.1080/19443994.2015.1026282
John, A., Luqman, M., Muhammad, S., Hanif, U., Sardar, A. A., Ali, S., Hasnian, A., Tufail, M., Khan, Z., Chaudhry, M. S., Yang, H.-H., & Awan, M. U. F. (2023). Genotoxicity of synthetic food colors on nitrogen-fixing bacteria in agricultural lands irrigated with wastewater of corresponding industries. Sustainability, 15(4), 2897. https://doi.org/10.3390/su15042897
John, D. M., Whitton, B. A., & Brook, A. J. (2002). The freshwater algal flora of the British Isles: An identification guide to freshwater and terrestrial algae. Cambridge University Press.
Kalsoom, U., Bhatti, H. N., Aftab, K., Amin, F., Jesionowski, T., & Bilal, M. (2023). Biocatalytic potential of Brassica oleracea L. var. botrytis leaves peroxidase for efficient degradation of textile dyes in aqueous medium. Bioprocess and Biosystems Engineering, 46(3), 453–465. https://doi.org/10.1007/s13201-023-01893-7
Kamal, I. M., Abdeltawab, N. F., Ragab, Y. M., Farag, M. A., & Ramadan, M. A. (2022). Biodegradation, decolorization, and detoxification of di-azo dye direct Red 81 by halotolerant, alkali-thermo-tolerant bacterial mixed cultures. Microorganisms, 10(5), 994. https://doi.org/10.3390/microorganisms10050994
Karmaker, S., Sintaha, F., & Saha, T. K. (2019). Kinetics, isotherm and thermodynamic studies of the adsorption of reactive red 239 dye from aqueous solution by chitosan 8B. Biological Chemistry, 9(01), 1. https://doi.org/10.4236/abc.2019.91001
Kartal, O. E. (2019). Decolorization of reactive red 239 using UV-C activated Peroxydisulfate. Polish Journal of Environmental Studies. https://doi.org/10.15244/pjoes/94049
Kasraee, M., Dehghani, M. H., Hamidi, F., Mubarak, N. M., Karri, R. R., Rajamohan, N., & Solangi, N. H. (2023). Adsorptive removal of acid red 18 dye from aqueous solution using hexadecyl-trimethyl ammonium chloride modified nano-pumice. Scientific Reports, 13(1), 13833. https://doi.org/10.1038/s41598-023-41100-w
Kaya, A., Yigit, E., & Akbulut, G. B. (2012). Effects of reactive red 239 textile dye on total soluble protein content, peroxidase activity and lipid peroxidation of Zea mays L. cv. “Martha F1.” African Journal of Agricultural Research, 7(24), 3588–3593. https://doi.org/10.5897/AJAR12.310
Khalaf, H. A., El-Sheekh, M. M., & Makhlof, M. E. M. (2023). Lychaete pellucida as a novel biosorbent for the biodegradation of hazardous azo dyes. Environmental Monitoring and Assessment, 195, 929. https://doi.org/10.1007/s10661-023-11518-w
Khan, S. A., Mehmood, S., Shabbir, S. B., Ali, S., Alrefaei, A. F., Albeshr, M. F., & Hamayun, M. (2023). Efficacy of fungi in the decolorization and detoxification of Remazol Brilliant Blue dye in aquatic environments. Microorganisms, 11(3), 703. https://doi.org/10.3390/microorganisms11030703
Khataee, A. R., & Dehghan, G. (2011). Optimization of biological treatment of a dye solution by macroalgae Cladophora sp. using response surface methodology. Journal of the Taiwan Institute of Chemical Engineers, 42(1), 26–33. https://doi.org/10.1016/j.jtice.2010.03.007
Khataee, A. R., Vafaei, F., & Jannatkhah, M. (2013). Biosorption of three textile dyes from contaminated water by filamentous green algal Spirogyra sp.: Kinetic, isotherm and thermodynamic studies. International Biodeterioration and Biodegradation, 83, 33–40. https://doi.org/10.1016/j.ibiod.2013.04.004
Koster, J. T. (1955). The genus Rhizoclonium Kuetz in the Netherlands. Pubbl Staz Zool Napoli, 27, 335–357.
Kumar, N., Sinha, S., Mehrotra, T., Singh, R., Tandon, S., & Thakur, I. S. (2019). Biodecolorization of azo dye Acid Black 24 by Bacillus pseudomycoides: Process optimization using Box Behnken design model and toxicity assessment. Bioresour Technol Rep, 8, 100311. https://doi.org/10.1016/j.biteb.2019.100311
Kumar, M., Sujatha, S., Gokulan, R., Vijayakumar, A., Praveen, S., & Elayaraja, S. (2021). Prediction of RSM and ANN in the decolorization of Reactive Orange 16 using biochar derived from Ulva lactuca. Desalination and Water Treatment, 211, 304–318. https://doi.org/10.5004/dwt.2021.26615
Kyzas, G. Z., Lazaridis, N. K., & Mitropoulos, A. C. (2012). Removal of dyes from aqueous solutions with untreated coffee residues as potential low-cost adsorbents: Equilibrium, reuse and thermodynamic approach. Journal of Chemical Engineering, 189, 148–159. https://doi.org/10.1016/j.cej.2012.02.045
Langmuir, I. (1918). The adsorption of gases on plane surfaces of glass, mica and platinum. Journal of the American Chemical Society, 40(9), 1361–1403.
Lata, H., Garg, V. K., & Gupta, R. K. (2007). Removal of a basic dye from aqueous solution by adsorption using Parthenium hysterophorus: An agricultural waste. Dyes and Pigments, 74(3), 653–658. https://doi.org/10.1016/j.dyepig.2006.04.007
Leliaert, F., & Boedeker, C. (2007). Cladophorales. In J. Brodie, C. A. Maggs, & D. M. John (Eds.), Green seaweeds of Britain and Ireland (pp. 131–183). Natural History Museum Publications.
Lima, E. C., Gomes, A. A., & Tran, H. N. (2020). Comparison of the nonlinear and linear forms of the van’t Hoff equation for calculation of adsorption thermodynamic parameters (∆S° and ∆H°). Journal of Molecular Liquids, 311, 113315. https://doi.org/10.1016/j.molliq.2020.113315
Majumder, S., Sharma, P., Singh, S. P., Nadda, A. K., Sahoo, P. K., Xia, C., Sharma, S., Ganguly, R., Lam, S. S., & Kim, K. H. (2023). Engineered biochar for the effective sorption and remediation of emerging pollutants in the environment. Journal of Environmental Chemical Engineering, 11(2), 109590. https://doi.org/10.1016/j.jece.2023.109590
Mehra, S., Singh, M., & Chadha, P. (2021). Adverse impact of textile dyes on the aquatic environment as well as on human beings. Toxicology International, 28(2), 165. https://doi.org/10.18311/ti/2021/v28i2/26798
Mekki, A., Dhouib, A., & Sayadi, S. (2007). Polyphenols dynamics and phytotoxicity in a soil amended by olive mill wastewaters. Journal of Environmental Management, 84(2), 134–140. https://doi.org/10.1016/j.jenvman.2006.05.015
Meng, X., Liu, G., Zhou, J., & Fu, Q. S. (2014). Effects of redox mediators on azo dye decolorization by Shewanella algae under saline conditions. Bioresource Technology, 151, 63–68. https://doi.org/10.1016/j.biortech.2013.09.131
Mishra, S., & Maiti, A. (2019). Optimization of process parameters to enhance the bio-decolorization of reactive red 21 by Pseudomonas aeruginosa 23N1. International Journal of Environmental Science and Technology, 16, 6685–6698. https://doi.org/10.1007/s13762-018-2023-1
Mokhtar, N., Aziz, E. A., Aris, A., Ishak, W. F. W., & Ali, N. S. M. (2017). Biosorption of azo-dye using marine macro-alga of Euchema Spinosum. Journal of Environmental Chemical Engineering, 5(6), 5721–5731. https://doi.org/10.1016/j.jece.2017.10.043
Nandi, B. K., Goswami, A., & Purkait, M. K. (2009). Adsorption characteristics of brilliant green dye on kaolin. Journal of Hazardous Materials, 161(1), 387–395. https://doi.org/10.1016/j.jhazmat.2008.03.110
Nejad, Z. G., Borghei, S. M., & Yaghmaei, S. (2019). Biodegradation of synthetic dye using partially purified and characterized laccase and its proposed mechanism. International Journal of Environmental Sciences, 16, 7805–7816. https://doi.org/10.1007/s13762-019-02226-5
Nienhuis, P. H. (1975). Biosystematics and ecology of Rhizoclonium riparium (Roth) Harv.(Chlorophyceae: Cladophorales) in the estuarine area of the rivers Rhine, Meuse and Scheldt. Bronder Offset BV.
Ozer, A., Akkaya, G., & Turabik, M. (2006). The removal of Acid Red 274 from wastewater: Combined biosorption and biocoagulation with Spirogyra rhizopus. Dyes and Pigments, 71(2), 83–89.
Padmesh, T. V. N., Vijayaraghavan, K., Sekaran, G., & Velan, M. (2005). Batch and column studies on biosorption of acid dyes on fresh water macro alga Azolla filiculoides. Journal of Hazardous Materials, 125(1–3), 121–129. https://doi.org/10.1016/j.jhazmat.2005.05.014
Prasad, A. A., & Rao, K. B. (2013). Aerobic biodegradation of Azo dye by Bacillus cohnii MTCC 3616; an obligately alkaliphilic bacterium and toxicity evaluation of metabolites by different bioassay systems. Applied Microbiology and Biotechnology, 97, 7469–7481. https://doi.org/10.1007/s00253-012-4492-3
Pujari, M., Srikanth, K., Sunil, K., & Arya, A. K. (2023). Equilibrium, kinetic, and thermodynamic study on biosorption of indigo carmine using Hypnea musciformis algae. Environmental Progress & Sustainable Energy, 42(2), e13990.
Redlich, O. J. D. L., & Peterson, D. L. (1959). A useful adsorption isotherm. Journal of Physical Chemistry, 63(6), 1024–1024. https://doi.org/10.1021/j150576a611
Robson, T. O., Fowler, M. C., & Barrett, P. R. (1976). Effect of some herbicides on freshwater algae. Pesticide Science, 7(4), 391–402.
Rudzinski, W., & Plazinski, W. (2007). Studies of the kinetics of solute adsorption at solid/solution interfaces: On the possibility of distinguishing between the diffusional and the surface reaction kinetic models by studying the pseudo-first-order kinetics. Journal of Physical Chemistry C, 111(41), 15100–15110. https://doi.org/10.1021/jp073249c
Rusan, M. J., Albalasmeh, A. A., Zuraiqi, S., & Bashabsheh, M. (2015). Evaluation of phytotoxicity effect of olive mill wastewater treated by different technologies on seed germination of barley (Hordeum vulgare L.). Environmental Science and Pollution Research, 22(2015), 9127–9135. https://doi.org/10.1007/s11356-014-4004-3
Salman, J. M., Grmasha, R. A., & Al-sareji, O. J., (2022). Potential efficiency of macroalgae Cladophora sp. to remove toxic industrial dye (safranin O) from aqueous solution. In AIP Conference Proceedings (Vol. 2386, No. 1). AIP Publishing. https://doi.org/10.1063/5.0066794
Santhi, T., Manonmani, S., & Smitha, T. (2010). Kinetics and isotherm studies on cationic dyes adsorption onto Annona squmosa seed activated carbon. International Journal of Engineering Science and Technology, 2(3), 287–295.
Saravanan, A., Kumar, P. S., Jeevanantham, S., Karishma, S., Tajsabreen, B., Yaashikaa, P. R., & Reshma, B. (2021). Effective water/wastewater treatment methodologies for toxic pollutants removal: Processes and applications towards sustainable development. Chemosphere, 280, 130595. https://doi.org/10.1016/j.chemosphere.2021.130595
Sarojini, G., Babu, S. V., Rajamohan, N., & Rajasimman, M. (2022a). Performance evaluation of polymer-marine biomass based bionanocomposite for the adsorptive removal of malachite green from synthetic wastewater. Environmental Research, 204, 112132. https://doi.org/10.1016/j.envres.2021.112132
Sarojini, G., Babu, S. V., Rajamohan, N., Rajasimman, M., & Pugazhendhi, A. (2022b). Application of a polymer-magnetic-algae based nano-composite for the removal of methylene blue—characterization, parametric and kinetic studies. Environmental Pollution, 292, 118376. https://doi.org/10.1016/j.envpol.2021.118376
Sarojini, G., Kannan, P., Rajamohan, N., Rajasimman, M., & Vo, D. V. N. (2023). Dyes removal from water using polymeric nanocomposites: A review. Environmental Chemistry Letters, 21(2), 1029–1058. https://doi.org/10.1007/s10311-022-01547-1
Sarwa, P., & Verma, S. K. (2013). Decolourization of Orange G dye by microalgae Acutodesmus obliquues strain PSV2 isolated from textile industrial site. International Journal of Applied Sciences and Biotechnology, 1(4), 247–252. https://doi.org/10.3126/.ijasbt.v1i4.9141
Setchell, W. A., & Gardner, N. L. (1920). The marine algae of the Pacific coast of North America (Vol. 8, pp. 139–374). University of California Press.
Seth, B. M., Uniyal, V., Kumar, D., & Singh, A. (2022). Sorption of cationic and anionic dyes by dead biomass of filamentous green alga Cladophora sp. (Chlorophyceae). International Journal of Environmental Science and Technology, 19, 12079–12090. https://doi.org/10.1007/s13762-021-03802-4
Sharma, P., & Dubey, R. S. (2005). Lead toxicity in plants. Brazilian Journal of Plant Physiology, 17, 35–52.
Sharma, P., & Kaur, H. (2011). Sugarcane bagasse for the removal of erythrosin B and methylene blue from aqueous waste. Applied Water Science, 1, 135–145. https://doi.org/10.1007/s13201-011-0018-x
Subramaniam, R., & Ponnusamy, S. K. (2015). Novel adsorbent from agricultural waste (cashew NUT shell) for methylene blue dye removal: Optimization by response surface methodology. Water Resources and Industry, 11, 64–70. https://doi.org/10.1016/j.wri.2015.07.002
Sudha, M., Bakiyaraj, G., Saranya, A., Sivakumar, N., & Selvakumar, G. (2018). Prospective assessment of the Enterobacter aerogenes PP002 in decolorization and degradation of azo dyes DB 71 and DG 28. Journal of Environmental Chemical Engineering, 6(1), 95–109. https://doi.org/10.1016/j.jece.2017.11.050
Temkin, M. J., & Pyzhev, V. (1940). Recent modifications to Langmuir isotherms. Acta Physicochimica U.R.S.S., 12, 217–225
Thiruchelvi, R., Venkataraghavan, R., & Sharmila, D. (2021). Optimization of environmental parameters by Plackett-Burman design and response surface methodology for the adsorption of Malachite green onto Gracilaria edulis. Materials Today: Proceedings, 37, 1859–1864. https://doi.org/10.1016/j.matpr.2020.07.448
Tien, C. J. (2002). Biosorption of metal ions by freshwater algae with different surface characteristics. Process Biochemistry, 38(4), 605–613.
Vaithiyanathan, T., & Sundaramoorthy, P. (2017). Analysis of sugar mill effluent and its influence on germination and growth of African marigold (Tagetes erecta L.). Applied Water Science, 7, 4715–4723. https://doi.org/10.1007/s13201-017-0634-1
Venkataraghavan, R., Thiruchelvi, R., & Sharmila, D. (2020). Statistical optimization of textile dye effluent adsorption by Gracilaria edulis using Plackett-Burman design and response surface methodology. Heliyon, 6(10), e05219. https://doi.org/10.1016/j.heliyon.2020.e05219
Vinayak, A., & Singh, G. B. (2022). Synthetic azo dye bio-decolorization by Priestia sp. RA1: Process optimization and phytotoxicity assessment. Archives of Microbiology, 204(6), 318. https://doi.org/10.1007/s00203-022-02931-9
Wang, H., Su, J. Q., Zheng, X. W., Tian, Y., Xiong, X. J., & Zheng, T. L. (2009). Bacterial decolorization and degradation of the reactive dye reactive red 180 by Citrobacter sp. CK3. International Biodeterioration and Biodegradation, 63(4), 395–399. https://doi.org/10.1016/j.ibiod.2008.11.006
Waqas, M. R., Nadeem, S. M., Khan, M. Y., Ahmad, Z., Ali, L., Asghar, H. N., & Khalid, A. (2022). Phycoremediation of textile effluents with enhanced efficacy of biodiesel production by algae and potential use of remediated effluent for improving growth of wheat. Environmental Science and Pollution Research, 29, 46118–46126. https://doi.org/10.1007/s11356-022-19140-y
Witek-Krowiak, A., Chojnacka, K., Podstawczyk, D., Dawiec, A., & Bubała, K. (2014). Application of response surface methodology and artificial neural network methods in modelling and optimization of biosorption process. Bioresource Technology, 160, 150–160. https://doi.org/10.1016/j.biortech.2014.01.021
Yang, C., Luo, H., Cheng, W., Jiang, K., Lu, L., & Ling, L. (2022). Decolorization characteristics and mechanism of methyl orange dye by using Stenotrophomonas acidaminiphila EFS1. International Journal of Environmental Science and Technology, 19(10), 10353–10362. https://doi.org/10.1007/s13762-021-03846-6
Yeddou, N., & Bensmaili, A. (2005). Kinetic models for the sorption of dye from aqueous solution by clay-wood sawdust mixture. Desalination, 185(1–3), 499–508. https://doi.org/10.1016/j.desal.2005.04.053
Zhang, Y., Xia, K., Liu, X., Chen, Z., Du, H., & Zhang, X. (2019). Synthesis of cationic-modified silica gel and its adsorption properties for anionic dyes. Journal of the Taiwan Institute of Chemical Engineers, 102, 1–8. https://doi.org/10.1016/j.jtice.2019.05.005
Acknowledgements
The authors wish to place their thanks to the authorities and management of Avinashilingam Institute for Home Science and Higher Education for Women, Coimbatore for the successful conduct of the study. The authors express their sincere appreciation to the Researchers Supporting Project Number (RSP2024R228) King Saud University, Riyadh, Saudi Arabia.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
A V Swathilakshmi—Investigation, Methodology, Writing—Original draft, G V Geethamala and R Mythili—Software, Formal analysis and Data curation, M Poonkothai and Kadarkarai Govindan—Conceptualization, Supervision and Validation, Mysoon M. Al-Ansari and Nora Dahmash Al-Dahmash—Data curation and Validation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Swathilakshmi, A.V., Geethamala, G.V., Poonkothai, M. et al. A response surface model to examine the reactive red 239 sorption behaviors on Rhizoclonium hieroglyphicum: isotherms, kinetics, thermodynamics and toxicity analyses. Environ Geochem Health 46, 37 (2024). https://doi.org/10.1007/s10653-023-01805-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10653-023-01805-3