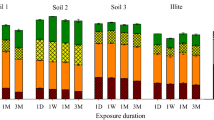
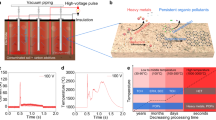
Abstract
This review summarizes the mechanisms for desorbing and extracting cesium (Cs+) from clay minerals and soil. Most techniques use ion exchange with acids, cations, polymers, and surfactants. Some improve desorption of Cs+ from clay minerals, while surfactants and polymers expand the interlayer. Mixtures of acids/polymers, acids/surfactants, cations/polymers, and cations/surfactants are therefore more effective agents for desorption of Cs+ from clay minerals. Hydrothermal treatment plays a role similar to that of polymers and surfactants in expanding the interlayer of clay minerals. The primary desorption mechanism expands the interlayer and desorbs Cs+, but multiple sequential extractions based on these techniques can more effectively desorb Cs+ from clay minerals and field-contaminated soils. Desorption techniques for Cs+ based on multiple sequential extractions can reportedly achieve an efficiency greater than 90%, and such approaches are likely to be important technologies for remediation of Cs+-contaminated soils and industrial accident sites, as well as the dismantling of nuclear power plants.

Similar content being viewed by others
References
Bond, A., Palerm, J., & Haigh, P. (2004). Public participation in EIA of nuclear power plant decommissioning projects: A case study analysis. Environmental Impact Assessment Review, 24, 617–641.
Bouzidi, A., Souahi, F., & Hanini, S. (2010). Sorption behavior of cesium on Ain Oussera soil under different physicochemical conditions. Journal of Hazardous Materials, 184, 640–646.
Cornell, R. M. (1993). Adsorption of cesium on minerals: A review. Journal of Radioanalytical and Nuclear Chemistry-Articles, 171, 483–500.
Dermont, G., Bergeron, M., Mercier, G., & Richer-Laflèche, M. (2008). Soil washing for metal removal: A review of physical/chemical technologies and field applications. Journal of Hazardous Materials, 152, 1–31.
Djedidi, S., Kojima, K., Yamaya, H., Ohkama-Ohtsu, N., Bellingrath-Kimura, S. D., Watanabe, I., et al. (2014). Stable cesium uptake and accumulation capacities of five plant species as influenced by bacterial inoculation and cesium distribution in the soil. Journal of Plant Research, 127, 585–597.
Endo, S., Kimura, S., Takatsuji, T., Nanasawa, K., Imanaka, T., & Shizuma, K. (2012). Measurement of soil contamination by radionuclides due to the Fukushima Dai-ichi Nuclear Power Plant accident and associated estimated cumulative external dose estimation. Journal of Environmental Radioactivity, 111, 18–27.
Fujita, T., Wang, L. P., Yabui, K., Dodbiba, G., Okaya, K., Matsuo, S., et al. (2013). Adsorption of cesium ion on various clay minerals and remediation of cesium contaminated soil in Japan. Resources Processing, 60, 13–17.
Fukushi, K., Sakai, H., Itono, T., Tamura, A., & Arai, S. (2014). Desorption of intrinsic cesium from smectite: Inhibitive effects of clay particle organization on cesium desorption. Environmental Science and Technology, 48, 10743–10749.
Fuller, A. J., Shaw, S., Ward, M. B., Haigh, S. J., Mosselmans, J. F. W., Peacock, C. L., et al. (2015). Caesium incorporation and retention in illite interlayers. Applied Clay Science, 108, 128–134.
Hirose, M., Kikawada, Y., Tsukamoto, A., Oi, T., Honda, T., Hirose, K., et al. (2015). Chemical forms of radioactive Cs in soils originated from Fukushima Dai-ichi nuclear power plant accident studied by extraction experiments. Journal of Radioanalytical and Nuclear Chemistry, 303, 1357–1359.
Kim, G.-N., Jung, Y.-H., Lee, J.-J., Moon, J.-K., & Jung, C.-H. (2008). An analysis of a flushing effect on the electrokinetic-flushing removal of cobalt and cesium from a soil around decommissioning site. Separation and Purification Technology, 63, 116–121.
Kim, G.-N., Kim, S.-S., Park, U.-R., & Moon, J.-K. (2015). Decontamination of soil contaminated with cesium using electrokinetic-electrodialytic method. Electrochimica Acta, 181, 233–237.
Kim, Y., Kirkpatrick, R. J., & Cygan, R. T. (1996). Cs-133 NMR study of cesium on the surfaces of kaolinite and illite. Geochimica et Cosmochimica Acta, 60, 4059–4074.
Korobova, E., Ermakov, A., & Linnik, V. (1998). 137Cs and 90Sr mobility in soils and transfer in soil–plant systems in the Novozybkov district affected by the Chernobyl accident. Applied Geochemistry, 13, 803–814.
Lee, M. E., Jeon, E.-K., Tsang, D. C., & Baek, K. (2018). Simultaneous application of oxalic acid and dithionite for enhanced extraction of arsenic bound to amorphous and crystalline iron oxides. Journal of Hazardous Materials, 354, 91–98.
Lee, J., Park, S. M., Jeon, E. K., & Baek, K. (2017). Selective and irreversible adsorption mechanism of cesium on illite. Applied Geochemistry, 85, 188–193.
Liu, C. X., Zachara, J. M., Smith, S. C., McKinley, J. P., & Ainsworth, C. C. (2003). Desorption kinetics of radiocesium from subsurface sediments at Hanford Site, USA. Geochimica et Cosmochimica Acta, 67, 2893–2912.
Lu, N., Mason, C. F., & Turney, W. R. (1996). Characterization and immobilization of cesium-137 in soil at Los Alamos National Laboratory. Los Alamos: Los Alamos National Lab.
Mallampati, S. R., Mitoma, Y., Okuda, T., Sakita, S., & Kakeda, M. (2012). High immobilization of soil cesium using ball milling with nano-metallic Ca/CaO/NaH2PO4: Implications for the remediation of radioactive soils. Environmental Chemistry Letters, 10, 201–207.
McKinley, J. P., Zachara, J. M., Heald, S. M., Dohnalkova, A., Newville, M. G., & Sutton, S. R. (2004). Microscale distribution of cesium sorbed to biotite and muscovite. Environmental Science and Technology, 38, 1017–1023.
Miranda-Trevino, J. C., & Coles, C. A. (2003). Kaolinite properties, structure and influence of metal retention on pH. Applied Clay Science, 23, 133–139.
Mukai, H., Hirose, A., Motai, S., Kikuchi, R., Tanoi, K., Nakanishi, T. M., et al. (2016). Cesium adsorption/desorption behavior of clay minerals considering actual contamination conditions in Fukushima. Scientific Reports, 6, 21543.
Murota, K., Saito, T., & Tanaka, S. (2016). Desorption kinetics of cesium from Fukushima soils. Journal of Environmental Radioactivity, 153, 134–140.
Nakao, A., Thiry, Y., Funakawa, S., & Kosaki, T. (2008). Characterization of the frayed edge site of micaceous minerals in soil clays influenced by different pedogenetic conditions in Japan and northern Thailand. Soil Science and Plant Nutrition, 54, 479–489.
Ndlovu, B., Farrokhpay, S., & Bradshaw, D. (2013). The effect of phyllosilicate minerals on mineral processing industry. International Journal of Mineral Processing, 125, 149–156.
Parajuli, D., Takahashi, A., Tanaka, H., Sato, M., Fukuda, S., Kamimura, R., et al. (2015). Variation in available cesium concentration with parameters during temperature induced extraction of cesium from soil. Journal of Environmental Radioactivity, 140, 78–83.
Park, S.-M., Alessi, D. S., & Baek, K. (2019a). Selective adsorption and irreversible fixation behavior of cesium onto 2: 1 layered clay mineral: A mini review. Journal of Hazardous Materials, 369, 569–576.
Park, C. W., Kim, B. H., Yang, H. M., Seo, B. K., & Lee, K. W. (2017a). Enhanced desorption of Cs from clays by a polymeric cation-exchange agent. Journal of Hazardous Materials, 327, 127–134.
Park, C. W., Kim, B. H., Yang, H. M., Seo, B. K., Moon, J. K., & Lee, K. W. (2017b). Removal of cesium ions from clays by cationic surfactant intercalation. Chemosphere, 168, 1068–1074.
Park, S.-M., Lee, J., Jeon, E.-K., Kang, S., Alam, M. S., Tsang, D. C., et al. (2019b). Adsorption characteristics of cesium on the clay minerals: Structural change under wetting and drying condition. Geoderma, 340, 49–54.
Park, S. M., Yang, J. S., Tsang, D. C. W., Alessi, D. S., & Baek, K. (2019c). Enhanced irreversible fixation of cesium by wetting and drying cycles in soil. Environmental Geochemistry and Health, 41, 149–157.
Pöllänen, R., Valkama, I., & Toivonen, H. (1997). Transport of radioactive particles from the Chernobyl accident. Atmospheric Environment, 31, 3575–3590.
Rigol, A., Vidal, M., & Rauret, G. (1999). Effect of the ionic status and drying on radiocesium adsorption and desorption in organic soils. Environmental Science and Technology, 33, 3788–3794.
Sato, K., Fujimoto, K., Dai, W., & Hunger, M. (2013). Molecular mechanism of heavily adhesive Cs: Why radioactive Cs is not decontaminated from soil. Journal of Physical Chemistry C, 117, 14075–14080.
Sawhney, B. L. (1972). Selective sorption and fixation of cations by Clay-minerals: Review. Clays and Clay Minerals, 20, 93–100.
Sharma, S., Singh, B., & Manchanda, V. (2015). Phytoremediation: Role of terrestrial plants and aquatic macrophytes in the remediation of radionuclides and heavy metal contaminated soil and water. Environmental Science and Pollution Research, 22, 946–962.
Steinhauser, G., Brandl, A., & Johnson, T. E. (2014). Comparison of the Chernobyl and Fukushima nuclear accidents: A review of the environmental impacts. Science of the Total Environment, 470, 800–817.
Tamura, K., Sato, H., & Yamagishi, A. (2015). Desorption of Cs+ ions from a vermiculite by exchanging with Mg2+ ions: effects of Cs+-capturing ligand. Journal of Radioanalytical and Nuclear Chemistry, 303, 2205–2210.
Tateda, Y., Tsumune, D., & Tsubono, T. (2013). Simulation of radioactive cesium transfer in the southern Fukushima coastal biota using a dynamic food chain transfer model. Journal of Environmental Radioactivity, 124, 1–12.
Wang, T. H., Li, M. H., Wei, Y. Y., & Teng, S. P. (2010). Desorption of cesium from granite under various aqueous conditions. Applied Radiation and Isotopes, 68, 2140–2146.
Willms, C., Li, Z. H., Allen, L., & Evans, C. V. (2004). Desorption of cesium from kaolinite and illite using alkylammonium salts. Applied Clay Science, 25, 125–133.
Yin, X. B., Horiuchi, N., Utsunomiya, S., Ochiai, A., Takahashi, H., Inaba, Y., et al. (2018). Effective and efficient desorption of Cs from hydrothermal-treated clay minerals for the decontamination of Fukushima radioactive soil. Chemical Engineering Journal, 333, 392–401.
Yin, X., Wang, X. P., Wu, H., Ohnuki, T., & Takeshita, K. (2017). Enhanced desorption of cesium from collapsed interlayer regions in vermiculite by hydrothermal treatment with divalent cations. Journal of Hazardous Materials, 326, 47–53.
Yoo, J.-C., Beiyuan, J., Wang, L., Tsang, D. C., Baek, K., Bolan, N. S., et al. (2018). A combination of ferric nitrate/EDDS-enhanced washing and sludge-derived biochar stabilization of metal-contaminated soils. Science of the Total Environment, 616, 572–582.
Yoo, J.-C., Lee, C.-D., Yang, J.-S., & Baek, K. (2013). Extraction characteristics of heavy metals from marine sediments. Chemical Engineering Journal, 228, 688–699.
Yoshida, Y., Sekiya, K., Nomura, N., Mishima, F., Akiyama, Y., & Nishijima, S. (2015). Study on volume reduction of contaminated soil by radioactive cesium using magnetic separation. IEEE Transactions on Applied Superconductivity, 25, 1–5.
Young, D. A., & Smith, D. E. (2000). Simulations of clay mineral swelling and hydration: Dependence upon interlayer ion size and charge. Journal of Physical Chemistry B, 104, 9163–9170.
Acknowledgements
This subject is supported by the Korea Ministry of Environment as “The SEM (Subsurface Environment Management) Projects; G232019011571” and partially supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (Grant No: 2019R1A4A1027795).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Park, SM., Kim, JG., Kim, HB. et al. Desorption technologies for remediation of cesium-contaminated soils: a short review. Environ Geochem Health 43, 3263–3272 (2021). https://doi.org/10.1007/s10653-020-00667-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10653-020-00667-3