Abstract
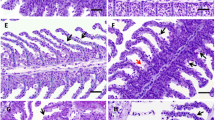
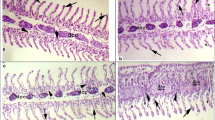
This study investigated changes in hematological and antioxidant parameters of carp exposed to two different doses of synthetic pyrethroid permethrin (control, vehicle, 10 ppm, and 20 ppm) for two different periods (4 days and 21 days). Hematological analyses were then performed on a veterinary Ms4 (Melet Schloesing, France) blood counter using commercially available kits (Cat. No. WD1153). Buege and Aust for MDA, Luck for CAT, McCord and Frivovich for SOD, Lawrence and Burk methods for GSH-Px were used to determine antioxidant parameters. Decreases in RBC count, Hb amount, Hct value, granulocyte ratios, and increases in total WBC and lymphocyte ratios were statistically significant in both dose groups treated with permethrin compared to the control group (p < 0.05). However, there was no statistically significant difference in monocyte ratios (p > 0.05). Overall, permethrin exposure caused an increase in MDA levels in the liver and gill tissues of carp in both dose and duration groups compared to the control group. Also, no statistically significant difference between the two dose groups in the liver tissue was observed in terms of duration (p > 0.05). Nonetheless, the increase in MDA levels in PERM10 and PERM20 dose groups in the gill tissues over 21 days was statistically significant (p < 0.05). Furthermore, permethrin exposure increased CAT, SOD, and GSH-Px enzyme activities in the gill tissue, while impacting in the opposite direction the liver tissue. Finally, regarding MDA, CAT, SOD, and GSH-Px levels, the control, and control acetone dose groups of all experimental groups were observed to be similar (p > 0.05). As a result, permethrin produced a toxic effect on Cyprinus carpio, triggering changes in blood parameters and inducing the antioxidant enzyme system.

Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author Nuh Korkmaz upon reasonable request at Osmaniye Korkut Ata University, Turkey.
References
Abdel-Daim MM, Abdelkhalek NKM, Hassan AM (2015) Antagonistic activity of dietary allicin against deltamethrin-induced oxidative damage in freshwater Nile tilapia; Oreochromis niloticus. Ecotoxicol Environ Saf 111:146–152. https://doi.org/10.1016/j.ecoenv.2014.10.019
Adhikari S, Sarkar B, Chatterjee A et al. (2004) Effects of cypermethrin and carbofuran on certain hematological parameters and prediction of their recovery in a freshwater teleost, Labeo rohita (Hamilton). Ecotoxicol Environ Saf 58:220–226. https://doi.org/10.1016/j.ecoenv.2003.12.003
Akinrotimi O, Gabriel U, Ariweriokuma S (2012) Haematotoxicity of cypermethrin to African catfish Clarias gariepinus under laboratory conditions. J Environ Eng Technol 1:13–19
Akinrotimi O, Amachree D (2016) Changes in haematological parameters of Tilapia guineensis exposed to different concentrations of detergent under laboratory conditions. J Aquat Sci 31:95. https://doi.org/10.4314/jas.v31i1.8
Alak G, Yeltekin AÇ, Özgeriş FB et al. (2019) Therapeutic effect of N- acetyl cysteine as an antioxidant on rainbow trout’s brain in cypermethrin toxicity. Chemosphere 221:30–36. https://doi.org/10.1016/j.chemosphere.2018.12.196
Amin KA, Hashem KS (2012) Deltamethrin-induced oxidative stress and biochemical changes in tissues and blood of catfish (Clarias gariepinus): antioxidant defense and role of alpha-tocopherol. BMC Vet Res 8:1–8. https://doi.org/10.1186/1746-6148-8-45
Atif F, Parvez S, Pandey S et al. (2005) Modulatory effect of cadmium exposure on deltamethrin-induced oxidative stress in Channa punctata Bloch. Arch Environ Contam Toxicol 49:371–377. https://doi.org/10.1007/s00244-003-9231-4
Awoyemi OM, Kumar N, Schmitt C et al. (2019) Behavioral, molecular and physiological responses of embryo-larval zebrafish exposed to types I and II pyrethroids. Chemosphere 219:526–537. https://doi.org/10.1016/j.chemosphere.2018.12.026
Birben E, Sahiner UM, Sackesen C et al. (2012) Oxidative stress and antioxidant defense. World Allergy Organ J 5:9–19
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310. https://doi.org/10.1016/S0076-6879(78)52032-6
Burgos-Aceves MA, Lionetti L, Faggio C (2019) Multidisciplinary haematology as prognostic device in environmental and xenobiotic stress-induced response in fish. Sci. Total Environ. 670:1170–1183
Carloni M, Nasuti C, Fedeli D et al. (2012) The impact of early life permethrin exposure on development of neurodegeneration in adulthood. Exp Gerontol 47:60–66. https://doi.org/10.1016/j.exger.2011.10.006
Chen X, Song M, Qi S, Wang C (2013) Safety Evaluation of Eleven Insecticides to Trichogramma nubilale (Hymenoptera: Trichogrammatidae). J Econ Entomol 106:136–141. https://doi.org/10.1093/jee/106.1.136
Christen V, Crettaz P, Fent K (2014) Additive and synergistic antiandrogenic activities of mixtures of azol fungicides and vinclozolin. Toxicol Appl Pharmacol 279:455–466. https://doi.org/10.1016/j.taap.2014.06.025
Dabas A, Nagpure NS, Mishra RM et al. (2014) Investigation of cadmium-induced genotoxicity and oxidative stress response in Indian major carp, Labeo rohita. Hum Ecol Risk Assess 20:510–526. https://doi.org/10.1080/10807039.2012.702591
David M, Sangeetha J, Shrinivas J et al. (2015) Effects of deltamethrin on haematological indices of indian major carp, Cirrhinus mrigala (Hamilton). Int J Pure Appl Zool 3:37–43
Davoodi R, Abdi G (2012) Comparative study on the acute toxicity of synthetic pesticides, permethrin 25% and monocrotophos 36%, and neem-based pesticide, neem gold EC 0.03%, to Juvenile Cyprinus carpio Linn. J Biol Environ Sci 6:105–108
de Moraes FD, Venturini FP, Rossi PA et al. (2018) Assessment of biomarkers in the neotropical fish Brycon amazonicus exposed to cypermethrin-based insecticide. Ecotoxicology 27:188–197. https://doi.org/10.1007/s10646-017-1884-2
Dhivya Vadhana MS, Nasuti C, Gabbianelli R (2010) Purine bases oxidation and repair following permethrin insecticide treatment in rat heart cells. Cardiovasc Toxicol 10:199–207. https://doi.org/10.1007/S12012-010-9079-6
Dhivya Vadhana MS, Siva Arumugam S, Carloni M et al. (2013) Early life permethrin treatment leads to long-term cardiotoxicity. Chemosphere 93:1029–1034. https://doi.org/10.1016/J.CHEMOSPHERE.2013.05.073
Durmaz H, Sevgiler Y, Üner N (2006) Tissue-specific antioxidative and neurotoxic responses to diazinon in Oreochromis niloticus. Pestic Biochem Physiol 84:215–226. https://doi.org/10.1016/j.pestbp.2005.07.004
Erkmen B (2015) Spermiotoxicity and embryotoxicity of permethrin in the sea urchin Paracentrotus lividus. Bull Environ Contam Toxicol 94:419–424. https://doi.org/10.1007/S00128-015-1482-Z
Falcioni ML, Nasuti C, Bergamini C et al. (2010) The primary role of glutathione against nuclear DNA damage of striatum induced by permethrin in rats. Neuroscience 168:2–10. https://doi.org/10.1016/j.neuroscience.2010.03.053
Fan B, Li J, Wang X et al. (2019) Study of aquatic life criteria and ecological risk assessment for triclocarban (TCC). Environ Pollut 254:112956. https://doi.org/10.1016/j.envpol.2019.112956
Fatima M, Ahmad I, Sayeed I et al. (2000) Pollutant-induced over-activation of phagocytes is concomitantly associated with peroxidative damage in fish tissues. Aquat Toxicol 49:243–250. https://doi.org/10.1016/S0166-445X(99)00086-7
Fazio F (2019) Fish hematology analysis as an important tool of aquaculture: a review. Aquaculture 500:237–242
Fazio F, Saoca C, Ferrantelli V et al. (2019a) Relationship between arsenic accumulation in tissues and hematological parameters in mullet caught in Faro Lake: a preliminary study. Environ Sci Pollut Res 26:8821–8827. https://doi.org/10.1007/s11356-019-04343-7
Fazio F, Saoca C, Sanfilippo M et al. (2019b) Response of vanadium bioaccumulation in tissues of Mugil cephalus (Linnaeus 1758). Sci Total Environ 689:774–780. https://doi.org/10.1016/j.scitotenv.2019.06.476
Ferreira D, Motta AC, da, Kreutz LC et al. (2010) Assessment of oxidative stress in Rhamdia quelen exposed to agrichemicals. Chemosphere 79:914–921. https://doi.org/10.1016/j.chemosphere.2010.03.024
Firat Ö, Aytekin T (2018) Neonikotinoid insektisit thiamethoxamın Oreochromis niloticus’ta oksidatif stres parametreleri üzerine etkisi. Balıkesir Üniversitesi Fen Bilim Enstitüsü Derg 20:1–11. https://doi.org/10.25092/baunfbed.427757
Gabbianelli R, Falcioni ML, Cantalamessa F, Nasuti C (2009a) Permethrin induces lymphocyte DNA lesions at both Endo III and Fpg sites and changes in monocyte respiratory burst in rats. Wiley Online Libr 29:317–322. https://doi.org/10.1002/jat.1412
Gabbianelli R, Falcioni ML, Nasuti C et al. (2009b) Effect of permethrin insecticide on rat polymorphonuclear neutrophils. Chem Biol Interact 182:245–252. https://doi.org/10.1016/J.CBI.2009.09.006
Gabbianelli R, Nasuti C, Falcioni G, Cantalamessa F (2004) Lymphocyte DNA damage in rats exposed to pyrethroids: effect of supplementation with Vitamins E and C. Toxicology 203:17–26. https://doi.org/10.1016/J.TOX.2004.05.012
Gabbianelli R, Palan M, Flis DJ et al. (2013) Imbalance in redox system of rat liver following permethrin treatment in adolescence and neonatal age. Xenobiotica 43:1103–1110. https://doi.org/10.3109/00498254.2013.796427
Gautam RK, Kumar S (2008) Alteration in haematology of Channa punctatus (Bloch). J Exp Zool India 11:309–310
George ADI, Akinrotimi OA, Nwokoma UK (2017) Haematological changes in African catfish (Clarias Gariepinus) exposed to mixture of atrazine and metolachlor in the laboratory. J Fish 11:48–54. https://doi.org/10.21767/1307-234x.1000129
Hedayati A, Hassan Nataj Niazie E (2015) Hematological changes of silver carp (Hypophthalmichthys molitrix) in response to Diazinon pesticide. J Environ Heal Sci Eng 13:52. https://doi.org/10.1186/s40201-015-0208-9
Hu F, Li L, Wang C et al. (2010) Enantioselective induction of oxidative stress by permethrin in rat adrenal pheochromocytoma (PC12) cells. Environ Toxicol Chem 29:683–690. https://doi.org/10.1002/etc.73
Imamura L, Hasegawa H, Kurashina K et al. (2000) Repression of activity-dependent c-fos and brain-derived neurotrophic factor mRNA expression by pyrethroid insecticides accompanying a decrease in Ca2+ influx into neurons. J Pharmacol Exp Ther 295:1175–1182
Issam C, Zohra H, Monia Z, Hassen BC (2011) Effects of dermal sub-chronic exposure of pubescent male rats to permethrin (PRMT) on the histological structures of genital tract, testosterone and lipoperoxidation. Exp Toxicol Pathol 63:393–400. https://doi.org/10.1016/J.ETP.2010.02.016
Jayaprakash C, Shettu N (2013) Changes in the hematology of the freshwater fish, Channa punctatus (Bloch) exposed to the toxicity of deltamethrin. J Chem Pharm Res 5:178–183
Jee J-H, Masroor F, Kang J-C (2005) Responses of cypermethrin-induced stress in haematological parameters of Korean rockfish, Sebastes schlegeli (Hilgendorf). Aquac Res 36:898–905. https://doi.org/10.1111/j.1365-2109.2005.01299.x
Jin Y, Chen R, Liu W, Fu Z (2010) Effect of endocrine disrupting chemicals on the transcription of genes related to the innate immune system in the early developmental stage of zebrafish (Danio rerio). Fish Shellfish Immunol 28:854–861. https://doi.org/10.1016/J.FSI.2010.02.009
Keramati V, Jamili S, Ramin M (2010) Effect of diazinon on catalase antioxidant enzyme activity in liver tissue of Rutilus rutilus. J Fish Aquat Sci 5:368–376. https://doi.org/10.3923/jfas.2010.368.376
Kong Y, Li M, Shan X et al. (2021) Effects of deltamethrin subacute exposure in snakehead fish, Channa argus: Biochemicals, antioxidants and immune responses. Ecotoxicol Environ Saf 209:111821. https://doi.org/10.1016/j.ecoenv.2020.111821
Kumar Maurya P, Malik DS, Kumar Yadav K et al. (2019) Haematological and histological changes in fish Heteropneustes fossilis exposed to pesticides from industrial waste water. Hum Ecol Risk Assess 25:1251–1278. https://doi.org/10.1080/10807039.2018.1482736
Lagadic L, Caquet T, Amiard JC (1997) Biomarqueurs en ecotoxicologie: aspect fondamentaux. In: Collection “Recherche en biologie.” p 479
Lawrence RA, Burk RF (1976) Glutathione peroxidase activity in selenium-deficient rat liver. Biochem Biophys Res Commun 71:952–958. https://doi.org/10.1016/0006-291X(76)90747-6
Li ZH, Zlabek V, Grabic R et al. (2010) Effects of exposure to sublethal propiconazole on the antioxidant defense system and Na+-K+-ATPase activity in brain of rainbow trout, Oncorhynchus mykiss. Aquat Toxicol 98:297–303. https://doi.org/10.1016/j.aquatox.2010.02.017
Luck H (1963) Catalase//Methods in enzymatic analysis/H.–U. Bergmeyer ed. Verlag Chemie, GmbH, Acad Press New York 885–894
Lushchak VI (2011) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101:13–30. https://doi.org/10.1016/j.aquatox.2010.10.006
Mahmoud N, Dellali M, Aissa P, Mahmoudi E (2012) Acute toxicities of cadmium and permethrin on the pre-spawning and post-spawning phases of Hexaplex trunculus from Bizerta Lagoon, Tunisia. Environ Monit Assess 184:5851–5861. https://doi.org/10.1007/s10661-011-2385-z
Malik DS, Maurya PK, Kumar H (2015) Alteration in haematological indices of Heteropneustis fossilis under stress heavy metals pollution in the Kali river, Uttar Pradesh, India. Int J Curr Res 7:15567–15573
McCord JM, Fridovich I (1969) Superoxide Dismutase. J Biol Chem 244:6049–6055. https://doi.org/10.1016/S0021-9258(18)63504-5
Modesto KA, Martinez CBR (2010a) Effects of roundup transorb on fish: hematology, antioxidant defenses and acetylcholinesterase activity. Chemosphere 81:781–787. https://doi.org/10.1016/j.chemosphere.2010.07.005
Modesto KA, Martinez CBR (2010b) Roundup® causes oxidative stress in liver and inhibits acetylcholinesterase in muscle and brain of the fish Prochilodus lineatus. Chemosphere 78:294–299. https://doi.org/10.1016/j.chemosphere.2009.10.047
Nasuti C, Cantalamessa F, Falcioni G, Gabbianelli R (2003) Different effects of type I and type II pyrethroids on erythrocyte plasma membrane properties and enzymatic activity in rats. Toxicology 191:233–244. https://doi.org/10.1016/S0300-483X(03)00207-5
Nasuti C, Carloni M, Fedeli D et al. (2014) Effect of 17β-estradiol on striatal dopaminergic transmission induced by permethrin in early childhood rats. Chemosphere 112:496–502. https://doi.org/10.1016/j.chemosphere.2014.05.035
Nasuti C, Falcioni ML, Nwankwo IE et al. (2008) Effect of permethrin plus antioxidants on locomotor activity and striatum in adolescent rats. Toxicology 251:45–50. https://doi.org/10.1016/J.TOX.2008.07.049
Nasuti C, Gabbianelli R, Falcioni ML et al. (2007) Dopaminergic system modulation, behavioral changes, and oxidative stress after neonatal administration of pyrethroids. Toxicology 229:194–205. https://doi.org/10.1016/j.tox.2006.10.015
Naz H, Abdullah S, Abbas K et al. (2019) Toxic effect of insecticides mixtures on antioxidant enzymes in different organs of fish, Labeo rohita. Pak J Zool 51:1355–1361. https://doi.org/10.17582/journal.pjz/2019.51.4.1363.1369
Olgun S, Misra HP (2006) Pesticides induced oxidative stress in thymocytes. Mol Cell Biochem 290:137–144. https://doi.org/10.1007/s11010-006-9178-7
Oruç EÖ, Usta D (2007) Evaluation of oxidative stress responses and neurotoxicity potential of diazinon in different tissues of Cyprinus carpio. Environ Toxicol Pharmacol 23:48–55. https://doi.org/10.1016/j.etap.2006.06.005
Orun I, Dogru MIi, Erdogan K et al. (2013) Effects of acute and chronic exposure to glyphosate on common carp (Cyprinus carpio L.) hematological parameters: the beneficial effect of propolis. Fresenius Environ Bull 22:2504–2509
Ozcan Oruc E, Sevgiler Y, Uner N (2004) Tissue-specific oxidative stress responses in fish exposed to 2,4-D and azinphosmethyl. Comp Biochem Physiol - C Toxicol Pharmacol 137:43–51. https://doi.org/10.1016/j.cca.2003.11.006
Özok N (2020) Effects of cypermethrin on antioxidant enzymes and lipid peroxidation of Lake Van fish (Alburnus tarichi). Drug Chem Toxicol 43:51–56. https://doi.org/10.1080/01480545.2019.1660363
Palaniappan PR, Vijayasundaram V (2008) FTIR study of arsenic induced biochemical changes on the liver tissues of fresh water fingerlings Labeo rohita. Rom J Biophys 18:135–144
Pandey S, Ahmad I, Parvez S et al. (2001) Effect of endosulfan on antioxidants of freshwater fish Channa punctatus bloch: 1. Protection against lipid peroxidation in liver by copper preexposure. Arch Environ Contam Toxicol 41:345–352. https://doi.org/10.1007/s002440010258
Parent LM, DeLorenzo ME, Fulton MH (2011) Effects of the synthetic pyrethroid insecticide, permethrin, on two estuarine fish species. J Environ Sci Heal - Part B Pestic Food Contam Agric Wastes 46:615–622. https://doi.org/10.1080/03601234.2011.589316
Parlak V (2018) Evaluation of apoptosis, oxidative stress responses, AChE activity and body malformations in zebrafish (Danio rerio) embryos exposed to deltamethrin. Chemosphere 207:397–403. https://doi.org/10.1016/j.chemosphere.2018.05.112
Parlak V, Atamanalp M, Alak G, Uçar A (2018) Alfa Sipermetrinin Gökkuşağı Alabalığı Solungaç Dokusunda Antioksidan Enzim Aktivitesi Üzerine Etkisi. In SETSCI Conference Indexing System 3:322–323
Patil VK, David M (2013) Oxidative stress in freshwater fish, Labeo rohita as a biomarker of malathion exposure. Environ Monit Assess 185:10191–10199. https://doi.org/10.1007/s10661-013-3323-z
Qayoom I, Balkhi MH, Shah FA, Bhat BA (2018) Toxicological evaluation and effects of oganophosphate compounds on hematological profile of juvenile common carps (Cyprinus carpio var. Communis). Indian J Anim Res 52:1469–1475. https://doi.org/10.18805/ijar.B-3526
Sah R, Baroth A, Hussain SA (2020) First account of spatio-temporal analysis, historical trends, source apportionment and ecological risk assessment of banned organochlorine pesticides along the Ganga River. Environ Pollut 263:114229. https://doi.org/10.1016/j.envpol.2020.114229
Salvo LM, Bainy ACD, Ventura EC et al. (2012) Assessment of the sublethal toxicity of organochlorine pesticide endosulfan in juvenile common carp (Cyprinus carpio). J Environ Sci Heal - Part A Toxic/Hazardous Subst Environ Eng 47:1652–1658. https://doi.org/10.1080/10934529.2012.687236
Saxena KK, Seth N (2002) Toxic effects of cypermethrin on certain hematological aspects of fresh water fish Channa punctatus. Bull Environ Contam Toxicol 69:364–369. https://doi.org/10.1007/s00128-002-0071-0
Sayeed I, Parvez S, Pandey S et al. (2003) Oxidative stress biomarkers of exposure to deltamethrin in freshwater fish, Channa punctatus Bloch. Ecotoxicol Environ Saf 56:295–301. https://doi.org/10.1016/S0147-6513(03)00009-5
Sellami B, Khazri A, Mezni A et al. (2015) Effect of permethrin, anthracene and mixture exposure on shell components, enzymatic activities and proteins status in the Mediterranean clam Venerupis decussata. Aquat Toxicol 158:22–32. https://doi.org/10.1016/j.aquatox.2014.10.020
Sellami B, Louati H, Dellali M et al. (2014) Effects of permethrin exposure on antioxidant enzymes and protein status in Mediterranean clams Ruditapes decussatus. Environ Sci Pollut Res 21:4461–4472. https://doi.org/10.1007/s11356-013-2404-4
Sepici-Dinçel A, Çağlan Karasu Benli A, Selvi M et al. (2009) Sublethal cyfluthrin toxicity to carp (Cyprinus carpio L.) fingerlings: Biochemical, hematological, histopathological alterations. Ecotoxicol Environ Saf 72:1433–1439. https://doi.org/10.1016/j.ecoenv.2009.01.008
Sharom MS, Solomon KR (1981) Adsorption-desorption, degradation, and distribution of permethrin in aqueous systems. J Agric Food Chem 29:1122–1125. https://doi.org/10.1021/jf00108a005
Shen HM, Liu ZG (2006) JNK signaling pathway is a key modulator in cell death mediated by reactive oxygen and nitrogen species. Free Radic Biol Med 40:928–939
Siegler K, Phillips BM, Anderson BS et al. (2015) Temporal and spatial trends in sediment contaminants associated with toxicity in California watersheds. Environ Pollut 206:1–6. https://doi.org/10.1016/j.envpol.2015.06.028
Singh NN, Srivastava AK (2010) Haematological parameters as bioindicators of insecticide exposure in teleosts. Ecotoxicology 19:838–854
Sinhorin VDG, Sinhorin AP, Teixeira JM, dos S et al. (2014) Effects of the acute exposition to glyphosate-based herbicide on oxidative stress parameters and antioxidant responses in a hybrid Amazon fish surubim (Pseudoplatystoma sp.). Ecotoxicol Environ Saf 106:181–187. https://doi.org/10.1016/j.ecoenv.2014.04.040
Slaninová A, Smutna M, Modra H, Svobodova Z (2009) A review: oxidative stress in fish induced by pesticides. Neuroendocrinol. Lett 30:2–12
Touaylia S, Ali M, Abdellhafidh K, Bejaoui M (2019) Permethrin induced oxidative stress and neurotoxicity on the freshwater beetle Laccophilus minutus. Chem Ecol 35:459–471. https://doi.org/10.1080/02757540.2019.1567718
Turkez H, Aydin E (2012) The effects of taurine on permethrin-induced cytogenetic and oxidative damage in cultured human lymphocytes. Arh Hig Rada Toksikol 63:27–34. https://doi.org/10.2478/10004-1254-63-2012-2114
Turkez H, Aydin E (2013) The genoprotective activity of resveratrol on permethrin- induced genotoxic damage in cultured human lymphocytes. Brazilian Arch Biol Technol 56:405–411. https://doi.org/10.1590/S1516-89132013000300008
Türkez H, Toǧar B (2011) Olive (Olea europaea L.) leaf extract counteracts genotoxicity and oxidative stress of permethrin in human lymphocytes. J Toxicol Sci 36:531–537. https://doi.org/10.2131/jts.36.531
Turkez H, Togar BŞ, Polat E (2012) Olive leaf extract modulates permethrin induced genetic and oxidative damage in rats. Cytotechnology 64:459–464. https://doi.org/10.1007/S10616-011-9424-Z
Ullah R, Zuberi A, Naeem M, Ullah S (2015) Toxicity to hematology and morphology of liver, brain and gills during acute exposure of mahseer (Tor putitora) to cypermethrin. Int J Agric. Biol 17:199–204
Ullah S, Hasan Z, Dhama K (2016) Toxic effects of endosulfanon on behaviour, protein contents and antioxidant enzyme system in gills, brain, liver and muscle tissues of Rohu, Labeo Rohita. Int J Pharmacol 12:1–10. https://doi.org/10.3923/ijp.2016.1.10
Ullah S, Li Z, Ul Arifeen MZ et al. (2019) Multiple biomarkers based appraisal of deltamethrin induced toxicity in silver carp (Hypophthalmichthys molitrix). Chemosphere 214:519–533. https://doi.org/10.1016/j.chemosphere.2018.09.145
Vadhana D, Carloni M, Fedeli D et al. (2011a) Perturbation of rat heart plasma membrane fluidity due to metabolites of permethrin insecticide. Cardiovasc Toxicol 11:226–234. https://doi.org/10.1007/S12012-011-9116-0
Vadhana MSD, Carloni M, Nasuti C et al. (2011b) Early life permethrin insecticide treatment leads to heart damage in adult rats. Exp Gerontol 46:731–738. https://doi.org/10.1016/J.EXGER.2011.05.005
Vaiyanan V, Sridharan G, Raveendran S, Chairman K (2015) Impact of pesticide on haematological parameters of Cyprinus carpio. World J Pharm Pharm Sci 4:1424–1430
Valavanidis A, Vlahogianni T, Dassenakis M, Scoullos M (2006) Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicol Environ Saf 64:178–189. https://doi.org/10.1016/j.ecoenv.2005.03.013
Van der Oost R, Beyer J, Vermeulen NPE (2003) Fish bioaccumulation and biomarkers in environmental risk assessment: a review. Environ Toxicol Pharmacol. 13:57–149
Veedu SK, Ayyasamy G, Tamilselvan H, Ramesh M (2022) Single and joint toxicity assessment of acetamiprid and thiamethoxam neonicotinoids pesticides on biochemical indices and antioxidant enzyme activities of a freshwater fish Catla catla. Comp Biochem Physiol Part - C Toxicol Pharmacol 257. https://doi.org/10.1016/j.cbpc.2022.109336
Wu P, Liu Y, Jiang WD, et al. (2017) A comparative study on antioxidant system in fish hepatopancreas and intestine affected by choline deficiency: different change patterns of varied antioxidant enzyme genes and nrf2 signaling factors. PLoS One 12. https://doi.org/10.1371/journal.pone.0169888
Xu LC, Liu L, Ren XM et al. (2008) Evaluation of androgen receptor transcriptional activities of some pesticides in vitro. Toxicology 243:59–65. https://doi.org/10.1016/j.tox.2007.09.028
Yekeen TA, Fawole OO (2011) Toxic effects of endosulfan on haematological and biochemical indices of Clarias gariepinus. African J Biotechnol 10:14090–14096. https://doi.org/10.5897/AJB10.2468
Yonar ME (2013) Protective effect of lycopene on oxidative stress and antioxidant status in Cyprinus carpio during cypermethrin exposure. Environ Toxicol 28:609–616. https://doi.org/10.1002/tox.20757
Acknowledgements
We would like to acknowledge Aksaray University, Science and Technology Application and Research Center, (Aksaray, Turkey) for the experimental process of this study.
Funding
This study was supported by the Amasya University Scientific Research Project Unit (FMB-BAP-17-0285).
Author information
Authors and Affiliations
Contributions
NK and İÖ designed the project, analyzed the data, and wrote the manuscript. NK, İÖ and OU performed the experiments and collected the data.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Ethics Approval
The experiment was carried out according to the international animal welfare and to the national Ethical Committee of Animal Sciences (Ahi Evran University Animal Experiments Local Ethics Committee-approval letter dated and the number is: 16.12.2016/4).
Consent to Participate
All authors consent the participation.
Consent for Publication
All authors consent the publication.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Korkmaz, N., Uğurer, O. & Örün, İ. Toxic effects of the synthetic pyrethroid permethrin on the hematological parameters and antioxidant enzyme systems of the freshwater fish Cyprinus carpio L. Ecotoxicology 32, 646–655 (2023). https://doi.org/10.1007/s10646-023-02675-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-023-02675-2