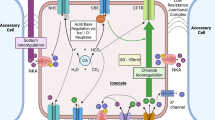
Abstract
Ammonia is one of the major aquatic environmental pollutants that can bring detrimental effects to the growth and survival of aquatic organisms. However, the molecular mechanisms of ammonia toxicity and ammonia excretion in marine invertebrates especially mollusks are still poorly understood. Cephalopods are exclusively ammonotelic with high protein metabolism and ammonia excretion rate, making this taxonomic group an ideal specimen to explore the ammonia detoxification mechanism. In this study, comparative transcriptomes were employed to investigate the transcriptional changes of O. minor in responses to acute ammonia exposure. A total of 63,237 unigenes with an average length of 811 bp were discovered and 25,708 unigenes were successfully annotated. The transcription of 1845 genes were significantly changed after ammonia stress, including 315 up-regulated genes and 1530 down-regulated genes. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis based on differentially expressed genes (DEGs) revealed that 44 GO terms and 55 KEGG pathways were over-represented. Notably, a large number of genes involved in immune defense, citric acid (TCA) cycle, oxidative phosphorylation and amino acid metabolisms were significantly down-regulated, indicating the decelerated energy production and amino acid rate in response to acute ammonia stress. These results provide new insights into the potential molecular mechanism of ammonia detoxification on transcriptomic level and will facilitate further mechanism studies on mollusks.



Similar content being viewed by others
References
Alexa A, Rahnenfuhrer J (2010) topGO: enrichment analysis for gene ontology. R package version 2.36.0
Andrews PLR, Darmaillacq AS, Dennison N et al. (2013) The identification and management of pain, suffering and distress in cephalopods, including anaesthesia, analgesia and humane killing. J Exp Mar Biol Ecol 447:46–64. https://doi.org/10.1016/j.jembe.2013.02.010
Arillo A, Margiocco C, Melodia F et al. (1981) Ammonia toxicity mechanism in fish: studies on rainbow trout (Salmo gairdneri Rich). Ecotoxicol Environ Saf 5:316–328. https://doi.org/10.1016/0147-6513(81)90006-3
Benli ACK, Koksal G, Ozkul A et al. (2008) Sublethal ammonia exposure of Nile tilapia (Oreochromis niloticus L.): effects on gill, liver and kidney histology. Chemosphere 72:1355–1358. https://doi.org/10.1016/j.chemosphere.2008.04.037
Boucher-Rodoni R, Mangold K (1989) Respiration and nitrogen excretion by the squid Loligo forbes. Mar Biol 103(3):333–338. https://doi.org/10.1007/BF00397267
Bower CE, Bidwell JP (1978) Ionization of ammonia in seawater: effects of temperature, pH, and salinity. J Fish Res Board Can 35:1012–1016. https://doi.org/10.1139/f78-165
Castellanos-Martinez S, Arteta D, Catarino S et al. (2014a) De novo transcriptome sequencing of the Octopus vulgaris hemocytes using Illumina RNA-Seq technology: response to the infection by the gastrointestinal parasite Aggregata octopiana. PLoS ONE 9:e107873. https://doi.org/10.1371/journal.pone.0107873
Castellanos-Martinez S, Prado-Alvarez M, Lobo-da-Cunha A et al. (2014b) Morphologic, cytometric and functional characterization of the common octopus (Octopus vulgaris) hemocytes. Dev Comp Immunol 44:50–58. https://doi.org/10.1016/j.dci.2013.11.013
Chen J (2000) Study on the free amino acid levels in the hemolymph, gill, hepatopancreas and muscle of Penaeus monodon exposed to elevated ambient ammonia. Aquatic Toxicol 50:27–37. https://doi.org/10.1016/s0166-445x(99)00095-8
Chen JC, Kou YZ (1992) Effects of ammonia on growth and molting of Penaeus japonicus juveniles. Aquaculture 104:249–260. https://doi.org/10.1016/0044-8486(92)90207-2
Cheng TC (1977) Biochemical and ultrastructural evidence for the double role of phagocytosis in molluscs: Defense and nutrition. In: Bulla LA, Cheng TC (eds) Comparative pathobiology, vol 3. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-7299-2_2
Chistiakov DA, Hellemans B, Volckaert FAM (2006) Microsatellites and their genomic distribution, evolution, function and applications: a review with special reference to fish genetics. Aquaculture 255:1–29. https://doi.org/10.1016/j.aquaculture.2005.11.031
Colt JE, Armstrong DA (2009) Nitrogen toxicity to crustaceans, fish, and molluscs. In: Allen LJ, Kinney EC (eds.) Proceedings of the bio-engineering symposium for fish culture. Northeast Society of Conservation Engineers, Bethesda, pp. 34–47
Dawson DA, Ball AD, Spurgin LG et al. (2013) High-utility conserved avian microsatellite markers enable parentage and population studies across a wide range of species. BMC Genom 14. https://doi.org/10.1186/1471-2164-14-176
Evans JJ, Pasnik DJ, Brill GC et al. (2006) Un-ionized ammonia exposure in Nile tilapia: toxicity, stress response, and susceptibility to Streptococcus agalactiae. N Am J Aquac 68:23–33. https://doi.org/10.1577/A05-032.1
Feyjoo P, Riera R, Felipe BC, Skalli A, Almansa E (2011) Tolerance response to ammonia and nitrite in hatchlings paralarvae of Octopus vulgaris and its toxic effects on prey consumption rate and chromatophores activity. Aquac Int 19:193–204
Ford LA (1992) Host defense mechanisms of cephalopods. Annu Rev Fish Dis 2:25–41. https://doi.org/10.1016/0959-8030(92)90054-2
Gao XL, Zheng XD, Bo QK et al. (2016) Population genetics of the common long-armed octopus Octopus minor (Sasaki, 1920) (Cephalopoda: Octopoda) in Chinese waters based on microsatellite analysis. Biochem Syst Ecol 66:129–136. https://doi.org/10.1016/j.bse.2016.03.014
Gao Y, Chen Y, Zhan S et al. (2017) Comprehensive proteome analysis of lysosomes reveals the diverse function of macrophages in immune responses. Oncotarget 8:7420–7440. https://doi.org/10.18632/oncotarget.14558
Gestal C, Castellanos-Martínez S et al. (2015) Understanding the cephalopod immune system based on functional and molecular evidence. Fish Shellfish Immunol 46:120–130. https://doi.org/10.1016/j.fsi.2015.05.005
Grabherr MG, Haas BJ, Yassour M et al. (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652. https://doi.org/10.1038/nbt.1883
Grimaldi AM, Belcari P, Pagano E et al. (2013) Immune responses of Octopus vulgaris (Mollusca: Cephalopoda) exposed to titanium dioxide nanoparticles. J Exp Mar Biol Ecol 447:123–127. https://doi.org/10.1016/j.jembe.2013.02.018
Hoeger U, Mommsen TP, O’Dor R et al. (1987) Oxygen uptake and nitrogen excretion in two cephalopods, octopus and squid. Comp Biochem Physiol Part A: Physiol 87(1):63–67. https://doi.org/10.1016/0300-9629(87)90426-9
Hu MY, Sung PH, Guh YJ et al. (2017) Perfused gills reveal fundamental principles of pH regulation and ammonia homeostasis in the Cephalopod Octopus vulgaris. Front Physiol 8:162. https://doi.org/10.3389/fphys.2017.00162
Hubert S, Higgins B, Borza T et al. (2010) Development of a SNP resource and a genetic linkage map for Atlantic cod (Gadus morhua). BMC Genom 11(1):191. https://doi.org/10.1186/1471-2164-11-191
Hurvitz A, Bercovier H, VanRijn J (1997) Effect of ammonia on the survival and the immune response of rainbow trout (Oncorhynchus mykiss, Walbaum) vaccinated against Streptococcus iniae. Fish Shellfish Immunol 7:45–53. https://doi.org/10.1006/fsim.1996.0062
Kim BM, Kang S, Ahn DH et al. (2018) The genome of common long-arm octopus Octopus minor. GigaScience 7(11):giy119. https://doi.org/10.1093/gigascience/giy119
Kuznetsova I, Lugmayr A, Siira SJ et al. (2019) CirGO: an alternative circular way of visualising gene ontology terms. BMC Bioinform 20:84. https://doi.org/10.1186/s12859-019-2671-2
Lee PG (1994) Nutrition of cephalopods: fueling the system. Mar Freshw Behav Physiol 25:35–51. https://doi.org/10.1080/10236249409378906
Li Y, Zhou F, Huang J et al. (2018) Transcriptome reveals involvement of immune defense, oxidative imbalance, and apoptosis in ammonia-stress response of the black tiger shrimp (Penaeus monodon). Fish Shellfish Immunol 83:162–170. https://doi.org/10.1016/j.fsi.2018.09.026
Liu C (2013) Studies on culture of the life cycle of Octopus minor (Sasaki, 1920). Fisheries College, Ocean University of China, pp. 7–33. (Chinese in English abstract)
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome biology 15(12):550. https://doi.org/10.1186/s13059-014-0550-8
Lu X, Kong J, Luan S et al. (2016) Transcriptome analysis of the hepatopancreas in the Pacific white shrimp (Litopenaeus vannamei) under acute ammonia stress. PLoS ONE 11:e0164396. https://doi.org/10.1371/journal.pone.0164396
Luzio JP, Pryor PR, Bright NA (2007) Lysosomes: fusion and function. Nat Rev Mol Cell Biol 8:622–632. https://doi.org/10.1038/nrm2217
Mayrand E, St‐Jean SD, Courtenay SC (2005) Haemocyte responses of blue mussels (Mytilus edulis L.) transferred from a contaminated site to a reference site: can the immune system recuperate? Aquac Res 36:962–971. https://doi.org/10.1111/j.1365-2109.2005.01302.x
Messenger JB, Nixon M, Ryan KP (1985) Magnesium chloride as ananaesthetic for cephalopods. Comp Biochem Physiol C 82:203–205. https://doi.org/10.1016/0742-8413(85)90230-0
Ni D, Song L, Wu L et al. (2007) Molecular cloning and mRNA expression of peptidoglycan recognition protein (PGRP) gene in bay scallop (Argopecten irradians, Lamarck 1819). Dev Comp Immunol 31:548–558. https://doi.org/10.1016/j.dci.2006.09.001
Nie H, Jiang L, Chen P et al. (2017) High throughput sequencing of RNA transcriptomes in Ruditapes philippinarum identifies genes involved in osmotic stress response. Sci Rep 7:4953. https://doi.org/10.1038/s41598-017-05397-8
Niknahad H, Jamshidzadeh A, Heidari R et al. (2017) Ammonia-induced mitochondrial dysfunction and energy metabolism disturbances in isolated brain and liver mitochondria, and the effect of taurine administration: relevance to hepatic encephalopathy treatment. Clin Exp Hepatol 3:141–151. https://doi.org/10.5114/ceh.2017.68833
Ott P, Clemmesen O, Larsen FS (2005) Cerebral metabolic disturbances in the brain during acute liver failure: from hyperammonemia to energy failure and proteolysis. Neurochem Int 47:13–18. https://doi.org/10.1016/j.neuint.2005.04.002
Peng RB, Le KX, Wang PS et al. (2016) Detoxification pathways in response to environmental ammonia exposure of the cuttlefish, Sepia pharaonis: glutamine and urea formation. J World Aquac Soc 48:342–352. https://doi.org/10.1111/jwas.12341
Perez DG, Fontanetti CS (2011) Hemocitical responses to environmental stress in invertebrates: a review. Environ Monit Assess 177:437–447. https://doi.org/10.1007/s10661-010-1645-7
Peyghan R, Takamy GA (2002) Histopathological, serum enzyme, cholesterol and urea changes in experimental acute toxicity of ammonia in common carp Cyprinus carpio and use of natural zeolite for prevention. Aquac Int 10:317–325. https://doi.org/10.1023/A:1022408529458
Pierce G, Valavanis V, Pereira J et al. (2006) Fishing for cephalopods. ICES CIEM Newsl43:24–28
Qian Y, Zheng X, Liu C et al. (2013) Studies on the reproductive habit and embryonic development of Octopus minor under the artificial conditions. Oceanol Limnol Sin 44:165–170. https://doi.org/10.11693/hyhz201301024024. Chinese in English abstract
Qu F, Xiang Z, Zhou Y et al. (2017) A molluscan TNF receptor-associated factor 2 (TRAF2) was involved in host defense against immune challenges. Fish Shellfish Immunol 71:105–115. https://doi.org/10.1016/j.fsi.2017.09.076
Schmittgen TD, Livak KJ et al. (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3:1101–1108. https://doi.org/10.1038/nprot.2008.73
Smart GR (1978) Investigations of the toxic mechanisms of ammonia to fish-gas exchange in rainbow trout (Salmo gairdneri) exposed to acutely lethal concentrations. J Fish Biol 12:93–104. https://doi.org/10.1111/j.1095-8649.1978.tb04155.x
Thiel T, Michalek W, Varshney RK et al. (2003) Exploiting EST databases for the development and characterization of gene-derived SSR-markers in barley (Hordeum vulgare L.). Theor Appl Genet 106:411–422. https://doi.org/10.1007/s00122-002-1031-0
Tomasso JR (1994) Toxicity of nitrogenous wastes to aquaculture animals. Rev Fish Sci 2:291–314. https://doi.org/10.1080/10641269409388560
Tomanek L (2015) Proteomic responses to environmentally induced oxidative stress. J Exp Biol 218:1867–1879. https://doi.org/10.1242/jeb.116475
Van der Auwera GA, Carneiro MO, Hartl C et al. (2013) From FastQ data to high confidence variant calls: the Genome Analysis Toolkit best practices pipeline. Curr Protoc Bioinform 43:11 10 11–33. https://doi.org/10.1002/0471250953.bi1110s43
Vaz-Pires P, Seixas P, Barbosa A (2004) Aquaculture potential of the common octopus (Octopus vulgaris Cuvier, 1797): a review. Aquaculture 238:221–238. https://doi.org/10.1016/j.aquaculture.2004.05.018
Vedel NE, Korsgaard B, Jensen FB (1998) Isolated and combined exposure to ammonia and nitrite in rainbow trout (Oncorhynchus mykiss): effects on electrolyte status, blood respiratory properties and brain glutamine/glutamate concentrations. Aquat Toxicol 41:325–342. https://doi.org/10.1016/S0166-445X(97)00071-4
Wang K, Li M, Hakonarson H (2010) ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38:e164. https://doi.org/10.1093/nar/gkq603
Wang MQ, Wang BJ, Liu M et al. (2019) The first identification of a malectin gene (CfMal) in scallop Chlamys farreri: sequence features and expression profiles. Invertebr Surviv J 16:25–33. https://doi.org/10.25431/1824-307X/isj.v0i0.25-33
Wang W, Yang S, Wang C et al. (2017) Gill transcriptomes reveal involvement of cytoskeleton remodeling and immune defense in ammonia stress response in the banana shrimp Fenneropenaeus merguiensis. Fish Shellfish Immunol 71:319–328. https://doi.org/10.1016/j.fsi.2017.10.028
Weihrauch D, Allen GJP (2018) Ammonia excretion in aquatic invertebrates: new insights and questions. J Exp Biol 221(2):jeb169219. https://doi.org/10.1242/jeb.169219
Wells MJ (1978) Octopus: physiology and behaviour of an advanced invertebrate. University Printing House, Cambridge
Xie C, Mao X, Huang J et al. (2011) KOBAS 2.0: a web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res 39:W316–322. https://doi.org/10.1093/nar/gkr483
Xiao J, Li QY, Tu JP et al. (2019) Stress response and tolerance mechanisms of ammonia exposure based on transcriptomics and metabolomics in Litopenaeus vannamei. Ecotoxicol Environ Saf 180:491–500. https://doi.org/10.1016/j.ecoenv.2019.05.029
Xu R, Zheng X (2018) Selection of reference genes for quantitative real-time PCR in Octopus minor (Cephalopoda: Octopoda) under acute ammonia stress. Environ Toxicol Pharmacol 60:76–81. https://doi.org/10.1016/j.etap.2018.04.010
Zannella C, Mosca F, Mariani F et al. (2017) Microbial diseases of bivalve mollusks: infections, immunology and antimicrobial defense. Mar Drugs 15(6):182. https://doi.org/10.3390/md15060182
Zheng, X, Qian, Y, Liu, C et al. (2014) Octopus minor. In: Iglesias J, Fuentes L, Villanueva R (eds), Cephalopod culture. Springer, New York, pp. 415–426
Zhou M, Wang AL, Xian JA (2011) Variation of free amino acid and carbohydrate concentrations in white shrimp, Litopenaeus vannamei: effects of continuous cold stress. Aquac Int 317:182–186. https://doi.org/10.1016/j.aquaculture.2011.04.033
Zuo ZR, Zheng XD, Yuan Y, Li Q (2011) Development and characterization of 12 polymorphic microsatellite loci in Octopus minor (Sasaki, 1920). Conserv Genet Resour 3:489–491. https://doi.org/10.1007/s12686-011-9386-7
Acknowledgements
The authors thank Mr. CAI Bing and Mr. CAI Hui from Mashan Fishery Company for their help in supplying O. minor samples and experiment sites. This work was supported by research grants from National Science Foundation of China (No. 31672257) and the Fundamental Research Funds for the Central Universities (No. 201822022).
Author information
Authors and Affiliations
Contributions
R.X. and X.D.Z conceived and designed the study. R.X. collected samples and performed the experimental work and data analysis. R.X. wrote the paper and X.D.Z revised the paper. All the authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards in Cephalopods (reviewed in Andrews et al. 2013).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Xu, R., Zheng, X. Hemocytes transcriptomes reveal metabolism changes and detoxification mechanisms in response to ammonia stress in Octopus minor. Ecotoxicology 29, 1441–1452 (2020). https://doi.org/10.1007/s10646-020-02279-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-020-02279-0