Abstract
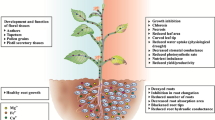
This work aimed to study the process of stress adaptation in root and leaves of different developmental stages (apex, middle and basal regions) of Pluchea sagittalis (Lam.) Cabrera plants grown under exposure to five Pb levels (0, 200, 400, 600 and 1000 μM) for 30 days. Pb concentration and content in roots, stems, and leaves of different developmental stages increased with external Pb level. Consumption of nutrient solution, transpiration ratio, leaf fresh weight, leaf area, and shoot length decreased upon addition of Pb treatments. However, dry weight of shoot parts and roots did not decrease upon addition of Pb treatments. Based on index of tolerance, the roots were much more tolerant to Pb than shoots. δ-aminolevulinic acid dehydratase activity was decreased by Pb treatments, whereas carotenoid and chlorophyll concentrations were not affected. Lipid peroxidation and hydrogen peroxide concentration both in roots and leaves increased with increasing Pb levels. Pb treatments increased ascorbate peroxidase activity in all plant parts, while superoxide dismutase activity increased in leaves and did not change in roots. Catalase activity in leaves from the apex shoot was not affected by Pb, but in other plant parts it was increased. Pb toxicity caused increase in non-protein thiol groups concentration in shoot parts, whereas no significant difference was observed in roots. Both root and shoot ascorbic acid concentration increased with increasing Pb level. Therefore, it seems that Pb stress triggered an efficient defense mechanism against oxidative stress in P. sagittalis but its magnitude was depending on the plant organ and of their physiological status. In addition, these results suggest that P. sagittalis is Pb-tolerant. In conclusion, P. sagittalis is able to accumulate on average 6730 and 550 μg Pb g−1 dry weight, respectively, in the roots and shoot, a physiological trait which may be exploited for the phytoremediation of contaminated soils and waters.





Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126
Angelone M, Bini C (1992) Trace elements concentrations in soils and plants of western Europe. In: Adriano DC (ed) Biogeochemistry of trace metals. Lewis Publishers, Boca Raton, pp 19–60
Bradford MM (1976) A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Calgaroto NS, Castro GY, Cargnelutti D, Pereira LB, Gonçalves JF, Rossato LV, Antes FG, Dressler VL, Flores EMM, Schetinger MRC, Nicoloso FT (2010) Antioxidant system activation by mercury in Pfaffia glomerata plantlets. Biometals 23:295–305
Cargnelutti D, Tabaldi LA, Spanevello RM, Jucoski GO, Battisti V, Redin M, Linares CEB, Dressler VL, Flores EMM, Nicoloso FT, Morsch VM, Schetinger MRC (2006) Mercury toxicity induces oxidative stress in growing cucumber seedlings. Chemosphere 65:999–1006
Cunningham SD, Berti WR (2000) Phytoextraction and phytostabilization: technical, economic and regulatory considerations of the sil-lead issue. In: Terry N, Banuelos G (eds) Phytoremediation of contaminated soil and water. Lewis Publication, Florida, pp 359–376
De Marco A, Roubelakis-Angelakis KA (1996) The complexity of enzymatic control f hydrogen peroxide concentration may affect the regeneration potential of plant protoplast. Plant Physiol 110:137–145
Ellman GL (1959) Tissue sulphydryl groups. Arch Biochem Biophys 82:70–77
El-Moshaty FIB, Pike SM, Novacky AJ, Sehgal OP (1993) Lipid peroxidation and superoxide production in cowpea (Vigna unguiculata) leaves infected with tobacco ringspot virus or southern bean mosaic virus. Physiol Mol Plant Pathol 43:109–119
Estrella-Gómez N, Mendoza-Cózatl D, Moreno-Sánchez R, González-Mendoza D, Zapata-Pérez O, Martínez-Hernández A, Santamaría JM (2009) The Pb-hyperaccumulator aquatic fern Salvinia minima Baker, responds to Pb2+ by increasing phytochelatins via changes in SmPCS expression and in phytochelatin synthase activity. Aquat Toxicol 91:320–328
Fatima RA, Ahmad M (2004) Certain antioxidant enzymes of Allium cepa as biomarkers for the detection of toxic heavy metals in wasterwater. Sci Total Environ 346:256–273
Gonçalves JF, Tabaldi LA, Cargnelutti D, Pereira LB, Maldaner J, Becker AG, Rossato LV, Rauber R, Bagatini MD, Bisognin DA, Schetinger MRC, Nicoloso FT (2009) Cadmium-induced oxidative stress in two potato cultivars. Biometals 22:779–792
Gopal R, Rizvi AH (2008) Excess lead alters growth, metabolism and translocation of certain nutrients in radish. Chemosphere 70:1539–1544
Gratão PL, Polle A, Lea PJ, Azevedo RA (2005) Making the life of heavy metal-stressed plants a little easier. Funct Plant Biol 32:481–494
Gupta DK, Nicoloso FT, Schetinger MRC, Rossato LV, Pereira LB, Castro GY, Srivastava S, Tripathi RD (2009) Antioxidant defense mechanism in hydroponically grown Zea mays seedlings under moderate lead stress. J Hazard Mater 172:479–484
Han Y, Huang S, Gu J, Qiu S, Chen J (2008) Tolerance and accumulation of lead by species of Iris L. Ecotoxicology 17:853–859
Hiscox JD, Israelstam GE (1979) A method for the extraction of chlorophyll from leaf tissue without maceration. Can J Bot 57:1132–1134
Horemans N, Foyer CH, Potters G, Asard H (2000) Ascorbate function and associated transport systems in plants. Plant Physiol Biochem 38:531–540
Islam E, Liu D, Li T, Yang X, Jin X, Mahmood Q, Tian S, Li J (2008) Effect of Pb toxicity on leaf growth, physiology and ultrastructure in the two ecotypes of Elsholtzia argyi. J Hazard Mater 154:914–926
Jacques-Silva MC, Nogueira CW, Broch LC, Flores EMM, Rocha JBT (2001) Diphenyl diselenide and ascorbic acid changes deposition of selenium and ascorbic acid in liver and brain of mice. Pharmacol Toxicol 88:119–125
Johnson FM (1998) The genetic effects of environmental lead. Mutat Res 410:123–140
Kratovalieva S, Cvetanovska L (2001) Influence of different Pb concentrations to some morphophysiological parameters at tomato (Lycopersicon esculentum Mill.) in experimental conditions. Maced Agr Rev 48:35–41
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Lorenzi H, Matos FJA (2002) Plantas Medicinais no Brasil: nativas e Exóticas cultivadas. Instituto Plantarum, São Paulo, p 168
Loreto F, Velikova V (2001) Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiol 127:1781–1787
Mishra S, Srivastava S, Tripathi RD, Kumar R, Seth CS, Gupta DK (2006) Lead detoxification by coontail (Ceratophyllum demersum L.) involves induction of phytochelatins and antioxidant system in response to its accumulation. Chemosphere 65:1027–1039
Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and simple assay for superoxide dismutase. J Biol Chem 244:6049–6055
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498
Morsch VM, Schetinger MRC, Martins AF, Rocha JBT (2002) Effects of cadmium, lead, mercury and zinc on δ-aminolevulinic acid dehydratase activity from radish leaves. Biol Plant 45:85–89
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Pereira LB, Tabaldi LA, Gonçalves JF, Jucoski JO, Pauletto MM, Weis SN, Nicoloso FT, Borher D, Rocha JBT, Schetinger MRC (2006) Effect of aluminum on δ-aminolevulinic acid dehydratase (ALA-D) and the development of cucumber (Cucumis sativus). Environ Exp Bot 57:106–115
Reddy AM, Kumar SG, Jyothsnakumari G, Thimmanaik S, Sudhakar C (2005) Lead induced changes in antioxidant metabolism of horsegram (Macrotyloma uniflorum (Lam.)Verdc.) and bengalgram (Cicer arietinum L.). Chemosphere 60:97–104
Rucinska R, Waplak S, Gwázdz EA (1999) Free radical formation and activity of antioxidant enzymes in lupin roots exposed to lead. Plant Physiol Biochem 37:187–194
Sampanpanish P, Pongsapich W, Khaodhiar S, Khan E (2006) Chromium removal from soil by phytoremediation with weed plant species in Thailand. Water Air Soil Poll 6:191–206
Sassa S (1982) Delta-aminolevulinic acid dehydratase assay. Enzyme 28:133–145
Scarano G, Morelli E (2002) Characterization of cadmium- and lead phytochelatin complexes formed in a marine microalga in response to metal exposure. Biometals 15:145–151
Sharma P, Dubey RS (2005) Lead toxicity in plants. Braz J Plant Physiol 17:35–52
Shigeoka S, Ishikawa T, Tamoi M, Miyagawa Y, Takeda T, Yabuta Y, Yoshimura K (2002) Regulation and function of ascorbate peroxidase isoenzymes. J Exp Bot 53:1305–1319
Singh PR, Tripathi RD, Sinha SK, Maheshwari R, Srivastava HS (1997) Response of higher plants to lead contaminated environment. Chemosphere 34:2467–2493
Smirnoff N (1996) The function and metabolism of ascorbic acid in plants. Ann Bot 78:661–669
Tamás L, Simonovicová M, Huttová J, Mistrík I (2004) Aluminum stimulated hydrogen peroxide production of germinating barley seeds. Environ Exp Bot 51:281–288
Van Assche F, Clijsters H (1990) Effects of metal on enzyme activity in plants. Plant Cell Environ 13:195–206
Verma S, Dubey RS (2003) Lead toxicity induces lipid peroxidation and alters the activities of antioxidant enzymes in growing rice plants. Plant Sci 164:645–655
Wang C, Tian Y, Wang X, Geng J, Jiang J, Yu H, Wang C (2010) Lead-contaminated soil induced oxidative stress, defense response and its indicative biomarkers in roots of Vicia faba seedlings. Ecotoxicology 19:1130–1139
Wierzbicka M (1987) Lead accumulation and its translocation barriers in roots of Allium cepa L.—autoradiographic and ultrastructural studies. Plant Cell Environ 10:17–26
Wierzbicka M (1998) Lead in the apoplast of Allium cepa L. root tips—ultrastructural studies. Plant Sci 133:105–119
Wierzbicka M (1999) Comparison of lead tolerance in Allium cepa with other plant species. Environ Pollut 104:41–52
Wu L, Antonovics J (1976) Experimental ecological genetics in Plantago. II. Lead tolerance in Plantago lanceolata and Cynodon dactylon from a roadside. Ecology 57:205–208
Wu C, Chen X, Tang J (2005) Lead accumulation in weed communities with various species. Comm Soil Sci Plant Anal 36:1891–1902
Xiang C, Oliver DJ (1998) Glutathione metabolic genes coordinately respond to heavy metals and jasmonic acid in Arabidopsis. Plant Cell 10:1539–1550
Yang HM, Zhang XY, Wang GX (2004) Effets of heavy metals on stomatal movements in Broad Bean leaves. Russ J Plant Physl 51:516–520
Zhou Y, Huang S, Yu S, Gu J, Zhao J, Han Y, Fu J (2010) The physiological response and sub-cellular localization of lead and cadmium in Iris pseudacorus L. Ecotoxicology 19:69–76
Zhu Z, Wei G, Li J, Qian Q, Yu J (2004) Silicon alleviates salt stress and increases antioxidant enzymes activity in leaves of salt-stressed cucumber (Cucumis sativus L.). Plant Sci 167:527–533
Acknowledgments
The authors thank Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação e Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and Fundação de Amparo à Pesquisa de Estado do Rio Grande do Sul (FAPERGS) for support to this research.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Rossato, L.V., Nicoloso, F.T., Farias, J.G. et al. Effects of lead on the growth, lead accumulation and physiological responses of Pluchea sagittalis . Ecotoxicology 21, 111–123 (2012). https://doi.org/10.1007/s10646-011-0771-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-011-0771-5