Abstract
White sucker (Catostomus commersonii) is a large-bodied benthic fish species that is found across a broad geographic region in North America. Often overlooked, white suckers are an integral component of aquatic ecosystems in their role as the dominant nearshore benthivore in many lakes. Few detailed field investigations on habitat use and thermal occupancy of white sucker exist, limiting our ability to predict the risk of habitat loss from development and climate warming for this cool-water species. Here we investigated seasonal depth, temperature and spatial occupancy patterns of white suckers in a lake located in northern Ontario, Canada. Using a combination of positioning acoustic telemetry and environmental data, we determined depth and space use patterns, seasonal temperature preference indices, and the affinity of white sucker to the lake bottom (i.e., benthic habitat) over a year long period. We found that the white suckers were consistently observed in shallow waters (< 10 m depth) and near the lake bottom across all seasons but were positioned slightly deeper in the winter. The tagged white suckers showed a strong temperature selection preference for thermal habitat between 10 and 16 °C during the open-water seasons and tended to avoid cold (< 6 °C) thermal habitat. Space use patterns, calculated using kernel utilization distributions, and daily movement rates were surprisingly consistent across all seasons, with regular occupancy of only some nearshore areas. This study highlights a highly restrictive pattern of habitat use by white sucker that is consistent across seasons, suggesting that this generalist species may be more vulnerable to anthropogenic disturbance than previously thought.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Many different fish species are integral to healthy functional ecosystems, but some of these are often overlooked in research and management as their perceived commercial or recreational value is minimal relative to other species. One such species is the white sucker (Catostomus commersonii), a benthivorous fish that can be commonly found in rivers and lakes across the majority of North America, from as southern as Georgia, USA (33°N) to the northern extent of its’ distribution in the Mackenzie River delta (Northwest Territories, Canada; 68°N) (Scott and Crossman 1973; Chu et al. 2016; Brownscombe et al. 2023). In many boreal lakes, white suckers represent the largest fish biomass in a given system (Trippel and Harvey 1987). Yet despite being an important component of fish communities, information on white sucker behaviour and ecology is limited. Instead, much of the research focus for this species has been related to their role as environmental monitors to evaluate the effects of toxic chemicals (Gibbons et al. 1998; McMaster et al. 2020), acidic deposition (Somers and Harvey 1984; Trippel and Harvey 1987), and diseases (Colavecchia et al. 2006; Blazer et al. 2017) due to its large geographic distribution and generally robust populations.
White suckers are rarely captured for direct human consumption in recreational fisheries but are regularly trapped for use as bait in the pursuit of gamefish species (Olson 1963; Margenau 2007; Begley et al. 2017). Additionally, white suckers are often used as bait in commercial fisheries such as the Maine lobster (Homarus americanus) fishery (Begley et al. 2017) or used in fish meal for walleye (Sander vitreus) aquaculture (Mejri et al. 2019). Our current understanding of the ecological significance of white suckers lies in their role as one of the only large-bodied benthivores in the nearshore areas of Boreal lakes. In this role, they can be competitors with other fish species, such as brook trout (Salvelinus fontinalis) (Brodeur et al. 2001), as well as an important food source for other animals such as river otters (Lontra canadensis) (Reid et al. 1994), bald eagles (Haliaeetus leucocephalus) (Todd et al. 1982), northern pike (Esox lucius) and walleye (Bertolo and Magnan 2005). White sucker are also known for their large spawning runs into small tributaries during the spring (Cooke et al. 2005; Doherty et al. 2010), thereby promoting the movement of resources essential to other biota in the form of resource subsidies as fish move from lakes to tributaries to spawn (Jones and Mackereth 2016; Jones and McKenzie 2023).
Temperature is a key component for habitat occupancy patterns of fish, as ectothermic species depend on suitable thermal habitats to effectively regulate biological processes such as metabolism (Brett and Groves 1979; Schulte 2015). There has been extensive research investigating the thermal habitat use of fish species with considerable economic value. For example, Christie and Regier (1988) estimated the sustained yield of four commercially valuable fish species (lake trout Salvelinus namaycush, lake whitefish Coregonus clupeaformis, walleye, and northern pike) in the Laurentian Great Lakes, using the amount of thermal habitat available to each species. More recent estimates of thermal occupancy in Boreal Shield lakes include top predators, such as brook trout (Cote et al. 2020; Smith et al. 2020), lake trout (Plumb and Blanchfield 2009; Ridgway et al. 2023), and northern pike (Guzzo et al. 2016). In contrast, the temperature preferences of underappreciated fish species, such as white sucker, are generalized based on thermal guilds (Hasnain et al. 2013) or unclear due to a lack of recent field observations (see Coutant 1977, Wismer and Christie 1987 and Hasnain et al. 2013 for past studies). Furthermore, past studies have not taken advantage of modern technology, such as acoustic telemetry, to follow individuals and observe white sucker temperature occupancy patterns. White suckers are often categorized as a cool-water species and are therefore assumed to prefer temperatures around 15–25 °C (Huff and Thomas 2014). An earlier study by Cincotta and Stauffer (1984) estimated the optimal thermal habitat for white suckers to be 14.1 to 21.2°C, whereas lab and field observations suggest a final temperature preferendum of 23.4°C (Hasnain et al. 2013).
Dimictic north temperate lakes show strong thermal stratification that can result in severe reductions in the availability of resources and suitable thermal habitat for fish depending on their preferences. The seasonal shifts in thermal habitat can provide opportunities for fish species of different thermal guilds to access the necessary resources for growth and reproduction through both spatial and temporal niche partitioning (Guzzo et al. 2016; Bloomfield et al. 2022). The warming of the epilimnion can lead cool- and cold-water fish species towards deeper waters and away from productive nearshore habitats, thus limiting the growth potential of these fish (Plumb and Blanchfield 2009; Pierce et al. 2013). For example, Guzzo et al. (2017) found that lake trout heavily relied on energy from littoral regions during the spring to sustain growth and continued to make metabolically costly forays into the nearshore habitat from deeper waters throughout summer to obtain high quality resources such as littoral prey fish. Unfortunately, we lack these types of detailed data on seasonal habitat use by white suckers, limiting our ability to predict seasonal shifts in fish thermoregulatory behaviour and the potential for habitat loss in the face of climate change.
The trophic ecology of white suckers is often characterized as a generalist feeder that forages in benthic nearshore habitats for detritus and invertebrates (Logan et al. 1991; Saint-Jacques et al. 2000), leading to a relatively low trophic position compared to piscivorous gamefish in similar ecosystems (Kidd et al. 1995; Johnston et al. 2019). White suckers have been observed to consume small-bodied prey, including both zoobenthos and zooplankton, during the growing season (Saint-Jacques et al. 2000). Logan et al. (1991) observed that the majority of white sucker diet was comprised of benthic taxa found in the shallow nearshore zone of lakes (epilimnion), although they also consumed benthos from the metalimnion and hypolimnion. These studies suggest that white suckers can utilize resources from a wide range of available habitats in lakes, which is extremely beneficial in the face of seasonal changes in the lake thermal profile that result in fluctuations in the availability of suitable habitat and associated resources. While previous studies have provided valuable insight into the trophic ecology of white suckers (Trippel and Harvey 1987; Logan et al. 1991; Saint-Jacques et al. 2000), very little is known about the seasonal patterns of white sucker behaviour and habitat use.
In this study, our main objective was to investigate seasonal depth, temperature, and habitat occupancy patterns of white suckers in a southern boreal lake. We used data collected from a positioning telemetry system over one year and continuous lake temperature profiles to characterize seasonal differences in depth and temperature selection. Given the broad range of temperature preference values found in the literature, we assessed white sucker thermal preferences in each season using temperature selection indices across the available thermal habitat to quantify selection and avoidance. Likewise, given the limited information available on white sucker habitat use in lakes, we evaluated their space use to determine whether there were specific areas of the lake that were preferred by white suckers and whether space use was season dependent. Understanding the seasonal behaviour and habitat use of white suckers fills an important knowledge gap that could allow for future predictions of the impacts of lake warming on this ecologically important species.
Methods
Study site and limnological data collection
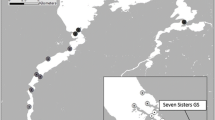
Our study site is Turkey Lake (84.42°W, 47.05°N), a 52 ha lake on the Boreal Shield in the highlands east of Lake Superior and approximately 58 km north of Sault Ste Marie, Ontario, Canada. This lake is situated in the Turkey Lakes watershed, a study area operated and used by various departments of the Canadian federal government since 1979 (Webster et al. 2021). The lake consists of a large primary basin with two smaller bays, one in the northwest and the other in the southeast portions of the lake (Fig. 1). Turkey Lake has a maximum depth of approximately 34 m with an average depth of 12 m. This oligotrophic lake is dimictic as it undergoes two periods of thermal stratification throughout the year (Jeffries et al. 1988). The fish community of Turkey Lake consists of white sucker, lake trout, brook trout, burbot (Lota lota), and various minnow species (e.g., northern redbelly dace Chrosomus eos, lake chub Couesius plumbeus, and emerald shiner Notropis atherinoides) (Kelso 1985).
Map of the telemetry array in Turkey Lake, Ontario, Canada from Richter et al. (2024). A total of 29 VR2Tx acoustic receivers (yellow) and two reference tags (pink) were deployed throughout the lake. The black dot in the inset map is the location of Turkey Lake at a broader geographic scale. Water temperature profile data were collected from the temperature loggers located over the deepest part of the lake using the temperature sensor string (star)
We collected lake bathymetry data using a sonar unit (Humminbird Helix 7; 455 and 1200 kHz modes) mounted on a small boat, which we drove in straight-line transects across the lake. We obtained full coverage of the entire lake by conducting transects that overlapped with one another and were spaced 20 – 100 m apart depending on the settings of the unit. We used the ReefMaster software (https://reefmaster.com.au/) to develop a bathymetric map (resolution = 17 m) to determine the lake's physical boundaries and to be able to associate a lake depth with each fish position observed.
Temperature data were collected year-round using an SDI-12 Temperature Sensor String from Campbell Scientific (https://www.campbellsci.ca/). The temperature sensor string was placed at the deepest part of the main central basin of Turkey Lake and suspended underneath a floating raft (Fig. 1). The series consisted of 18 temperature sensors, with the first sensor located 0.5 m underneath the lake's surface. After the first sensor, sensors were spaced at 1 m intervals until a depth of 12.5 m, at which the spacing between sensors increased to approximately 5 m intervals. Temperature data were collected every 10 min and were used to develop daily thermal profiles by linear interpolation. Seasons were delineated using daily mean surface water temperatures (defined as all depths < 6 m) and, along with daily photos of ice conditions from trail cameras (Stealth Cam DS4K – STC-DS4KTM), the dates for ice-on and ice-off which signify 100% ice coverage conditions (Guzzo et al. 2017). We defined spring as the period after ice-off and before the daily mean surface water temperature exceeded 15°C. Summer was when the mean surface water temperature was greater than 15°C, and fall occurred when mean surface water temperatures fell below 15°C until ice-on. Winter was when the lake was covered with ice (ice-on to ice-off).
Fish tagging and telemetry
White suckers were captured using trap nets that were set at locations that were suitable for effective deployment when the surface water temperature was < 15 °C between October 26th and 28th, 2021. A total of ten white suckers were implanted with one of two types of pressure-sensing (depth) acoustic transmitters (V9P-2 × and V13P-2x, Innovasea Systems Inc., Bedford, Nova Scotia, Canada). The choice of the transmitter was dependent on the size of the fish (mean weight: 1257 g; range: [610, 1700 g]), with all transmitters weighing < 2% of the fish's body mass (Hubbard et al. 2021). The transmission interval for the tags used in our study were 240 to 360 s.
The surgical implantation of the transmitters generally followed protocols established in previous studies (see Blanchfield et al. 2005) and more specific details of the surgery procedures can be found in Richter et al. 2024, and the care and use of the fish complied with the Animal Care Committee of Fisheries and Oceans Canada (GWACC-130 – Fish Tagging Protocol). Once captured, the fish were transported to a well-oxygenated holding tank until surgery. The fish were immobilized using Smith-Root electric fish handling gloves (Ward et al. 2017; Tuononen et al. 2022), weighed (nearest g; A&D model SK-20KWP) and measured (to the nearest mm) for fork length and total length. All surgical instruments were sterilized using betadine (10% povidone-iodine), and lidocaine (to a max of 10 mg/kg based on GWACC-105) was injected subcutaneously at the incision site before surgery. An incision of approximately 20 – 30 mm, depending on the size of the transmitter, was made off-centre (~ 5 – 10 mm from midline) on the ventral side of the fish to insert the transmitter in the body cavity. Depending on the incision size, one to three interrupted sutures using absorbable monofilament 3–0 diameter (Ethicon coded Z761D) were used to close the incision. The fish were placed in another aerated cooler for observation and recovery post-surgery. Once the fish resumed normal swimming behaviour and appeared healthy, they were released back into the lake (mean recovery time: ~ 18 min). We were unable to sex the white suckers during the tagging event as it was outside of the spawning period.
Data collection via acoustic telemetry began in October 2021 and continued until December 2022. The fine-scale positioning system consisted of 29 underwater omnidirectional acoustic receivers (VR2Tx, 69 Hz) (Fig. 1). The receivers detect the acoustic signals emitted from the surgically implanted transmitters and record the acoustic data (fish ID, depth, and acceleration depending on transmitter type) along with the date and time of detection. VR2Tx receivers have built-in sync tags to improve positioning accuracy and allow internal clock synchronization during data processing. In addition, reference tags (transmission interval: [540, 660 s]) were also deployed at two locations in Turkey Lake to improve positioning accuracy. More details about the receiver deployment in Turkey Lake can be found in Richter et al. 2024. The spatial data processing was undertaken by Innovasea Systems Inc., which involved using the raw detection data to solve for spatial positions of the tagged fish by using the Fathom Position software (https://www.innovasea.com/fish-tracking/products/fathom-software/).
The fish telemetry data, which included 710,002 unique positions for the tagged white suckers, were subject to multiple filters before analyses. We excluded location data that were outside the study area based on the spatial (i.e., shoreline) and depth (i.e., lake depth + 2 m buffer) profiles of Turkey Lake (9.6%). Also, we included only the data collected in 2022 as our “fall” seasonal period and omitted data from fall 2021 (13.3%) to allow for continuous daily data that included the transition from summer to fall. Therefore, the telemetry data that was used in this study ranged from December 5th, 2021 to December 8th, 2022. The omission of the fall 2021 data also included all data collected from one fish (ID 8835) which was a suspected mortality or tag failure due to a cease in detections (last detection on November 11th, 2021). We excluded data from two other fish after suspected mortality or tag failure events (see Figure S1) based on a cease in detections followed by a lack of vertical and horizontal movement (> 2 weeks). In addition, the hyperbolic positioning error (HPE), a unitless measure of spatial certainty, was calculated for each position, and positions with an HPE > 20 were removed from the dataset (7.9%) (Smith 2013; Rodrigues et al. 2022). The spatial data from the detections were converted from latitude and longitude to Universal Transverse Mercator (UTM) to ensure that both spatial and depth data were measured in the same units (m) as the amount of thermal habitat used and available were measured in volume (m3) (Charles et al. 2017).
Data analysis
Depth occupancy patterns of the white suckers were evaluated using the mean daily depth across individual fish. A linear mixed-effects model was used to test whether the observed fish depths of the telemetry-based detections differed among seasons. The model was parameterized using fish depth (log-transformed to achieve normality) as the response variable and season as the fixed effect. Fish ID nested within date was considered for the random effect. The significance of the effect of season on fish depth was evaluated using a t-test using Satterthwaite's approximation for computing the degrees of freedom (Satterthwaite 1946). Marginal and conditional variation explained (R2) were also calculated for the model to determine the amount of variance explained by the fixed and random effects (Nakagawa and Schielzeth 2013). We used the lme4 R package (Bates et al. 2014) to parameterize the linear mixed-effects model using maximum likelihood. The lmerTest (Kuznetsova et al. 2017) and MuMIn R packages (Barton and Barton 2019) were used to analyze the linear mixed-effects model.
We investigated the temperature preferences of tagged white suckers using temperature selection indices that relate the fish use of a given habitat to the amount of available habitat in each category (Pierce et al. 2013; Harrison et al. 2016). We associated water temperature to fish positions by using the depth information collected by the pressure-sensing transmitters and the daily temperature-depth profile of the lake under the assumption that the temperature-depth profile was constant throughout the lake. While we acknowledge that there may be some spatial variation in the thermal regime, however, these variations should be minimal or modest based on lake size and the heavily forested shorelines (Fee et al. 1996; Cott et al. 2015). For our study, we categorized thermal habitat by 2 °C bins/intervals. The temperature selection indices for each habitat category (\({\widehat{w}}_{i}\)) were calculated following the equation provided by Rogers and White (2007),
Where \({u}_{i+}\) is the number ofcgory i across all fish, \({\pi }_{i}\) is the proportion of the available habitat for category i, and \({u}_{++}\) was the total number of detections for all fish (Pierce et al. 2013). The daily temperature-depth profiles were used to calculate the daily available habitat volume for each thermal habitat category. The seasonal proportion of available habitat for each category was quantified as the mean of the daily available habitat volume estimates for each habitat category within a season divided by the total habitat volume for the lake. Temperature selection indices were calculated for all thermal habitat categories with available habitat for each season. The Bonferonni-corrected 95% confidence interval values (α = 0.05) for the selection indices were calculated by following the standard error equation provided by Rogers and White (2007) and Pierce et al. (2013). While acknowledging the inherent autocorrelation often associated with telemetry positional data, the effects are mitigated by the complete receiver coverage of the lake and the collection of the telemetry data at regular, but random, intervals from the tagged fish. The telemetry positional data are representative of white sucker habitat use patterns.
As white sucker is a benthic fish species, we investigated their association with the lake bottom and benthic habitat. We evaluated the density of detections between fish depth and lake depth using kernel density estimation (Guzzo et al. 2016). We calculated the core (50%) and broad (95%) fish-lake depth niches for each season using the ks package in R (Duong et al. 2022). Inverse frequency weighting based on the number of detections for each fish was used to account for differences in the number of recorded positions by individual white suckers in the pooled data for the KUD parameterization. The distance to lake bottom for each tagged sucker was calculated as the difference between fish and lake depth for each observed telemetry position, and seasonal differences among the distance to lake bottom distributions were tested using the Kruskal–Wallis test. In the presence of significant differences in the distance to the lake bottom among seasons, we used Dunn's test to determine which pairwise groups differed using the Bonferroni method to account for multiple comparisons (Rice 1989). Additionally, we calculated the proportion of detections that were ≤ 1 m from the lake bottom for each season.
The space use of the Turkey Lake white suckers was evaluated using kernel density estimation to generate kernel utilization distributions (KUDs) (Worton 1989). These KUDs generate probability densities that an animal is found across numerous discrete evaluation points throughout the study area (Charles et al. 2017). KUDs are often used to represent the home ranges and core areas (95% and 50% isopleths, respectively) of individuals or populations (Guzzo et al. 2016). We calculated the home range and core area at the species level for each season using all the available telemetry positions from all observed fish. To account for the unequal number of detections across the white suckers, we used inverse frequency weighting based on the number of detections for each fish in the season. Therefore, individuals with more detections during a season had less weight whereas individuals with fewer detections had a greater weight in the estimation of the KUD. We then compared KUD estimates among the seasons descriptively to determine whether the amount of white sucker space use and its spatial distribution differ seasonally. We evaluated potential changes in the location of white sucker space use by calculating the overlap among seasonal home ranges and core areas. Overlap of home ranges and core areas across seasons were calculated for each season pair and presented as the proportion of area overlap relative to the mean area of the two seasons. We also calculated the spatial overlap among individual- and group-level KUDs across the seasons to quantify seasonal space use for individuals relative to the total area used by all tagged individuals. The ks package in R was used to parameterize white sucker KUDs (Duong et al. 2022). Along with the KUD estimates for space use, we also calculated daily horizontal and vertical movement rates of the white suckers as the total daily movement across all observed positions within the day to determine whether there are any seasonal differences in white sucker movement behaviour. We used the Kruskal–Wallis test to determine whether movement rates significantly differed among seasons.
Results
There were 461,532 detections across the nine tagged white suckers after filtering, with telemetry data analyses covering four consecutive seasons, beginning on the first day of winter 2021 (Dec. 5) and ending on the final day of fall 2022 (Dec. 7). Telemetry data were available for nine white suckers in winter 2021, six in spring 2022, and five in both the summer and fall seasons of 2022. The depth occupancy of white suckers in Turkey Lake was consistent across seasons, with fish generally occupying the upper 10 m portion of the water column (Fig. 2). During the summer, when optimal thermal habitat was available (range: [14.1, 21.2°C]; Cincotta and Stauffer 1984), white sucker mean depth was generally bounded by the depth range of the optimal thermal habitat, with tagged fish tending to reside closer to the cooler end of the optimal habitat thermal range (i.e., 14 °C) (Fig. 2). Season had a significant effect on the depth occupancy of white sucker, however, there was no significant difference in depth occupancy between the winter and fall seasons (Table 1). On average, white suckers were at their deepest in the winter (mean: 6.10 m) and were shallower during the other seasons (3.78, 4.85, and 4.95 m for mean depths in spring, summer, and fall, respectively; Fig. 2). Quantitatively, there was higher variability in white sucker depth occupancy patterns in the spring and fall relative to the summer and winter seasons (Figure S2). The marginal R2 value indicated that the proportion of variation explained by the fixed effect, season, was 0.100 while the combination of fixed and random effects accounted for 0.824 of the variance explained (R2conditional).
Daily depth occupancy of tagged white suckers (n = 9) over time in Turkey Lake. The individual black points are the mean daily depths occupied by the individual tagged white suckers, and the shaded region represents the mean daily depth ± SE across all individuals. The vertical dashed lines represent the start of different seasons (Winter 2021, Spring 2022, Summer 2022, and Fall 2022 chronologically) across the study period. The expected depth occupancy during the summer, based on white sucker temperature preferences [14.1, 21.2 °C], is presented with the gray lines
As the availability of thermal habitat fluctuated across seasons, temperature selection indices varied among temperature intervals (Table S1; Fig. 3). Whereas white sucker did not show any thermal preference in the winter (\({\widehat{w}}_{i}=1\)) (Table S1), during the spring they displayed a strong preference for thermal habitats with a range from 12.0 °C to 16.0 °C and avoided thermal habitats below 10 °C. Tagged white suckers occupied a wide range of thermal habitats during the summer but only showed an affinity for thermal habitats from 14.0 °C to 16.0 °C, and avoided the coldest available habitat (< 6.0 °C) during the summer. White suckers tended to select for cooler thermal habitats during the fall relative to the summer, preferring temperatures from 10.0 to 14.0 °C (Table S1). Over the entire open-water period (spring, summer, fall), white suckers showed a strong preference for thermal habitats from 10.0 to 16.0 °C and displayed no preference for warmer habitats (18.0 – 22.0°C; Fig. 3).
Seasonal temperature preference indices across available temperature bins (2 °C) for acoustically-tagged white suckers in Turkey Lake (Ontario) across seasons. The error bars for each temperature interval represent the Bonferroni corrected 95% confidence intervals around the mean estimate (wi ± zα/2ISE, α = 0.05) and * indicates a significant temperature selection ratio that differs from 1. A preference index > 1 (dashed line) indicates a selection preference for that temperature bin, and a preference index < 1 is indicative of avoidance of that temperature bin. The expected optimal thermal habitat, based on existing literature, for white suckers [14.1, 21.2 °C] is presented using the shaded gray area
White suckers were strongly associated with the lake bottom, particularly for areas of the lake shallower than 10 m depth (Fig. 4). The majority of white sucker telemetry positions were within 1 m of the lake bottom with a high affinity to benthic habitat during the winter, summer and fall seasons (84.4 – 87.8%) and a notably smaller proportion during the spring (73.0%). Although fish-lake depth occupancy patterns across seasons appeared consistent (Fig. 4), distance to the lake bottom differed significantly among the seasons (KW χ2 = 1878.19, df = 3, p < 0.001). There were significant differences among all pairwise groups of seasons according to the Dunn's Test (p < 0.001). The tagged white suckers were closest to the lake bottom in the fall, followed by winter and summer, and were positioned furthest away from the lake bottom during spring.
The depth occupancy of white suckers relative to the lake depth in Turkey Lake. Colour indicates the density of telemetry observations. The points represent individual white sucker telemetry observations, and the dashed line is the 1:1 relationship between fish and lake depth. The coloured regions indicate the seasonal mean core (50% contour) and broad (95% contour) fish-lake depth habitat niches based kernel density estimates
Space use by the tagged white suckers did not appear to differ considerably among seasons in Turkey Lake based on a descriptive comparison (Fig. 5). Across the year, white sucker home range (95% KUD) was greatest in the spring (18.4 ha) and least in the fall (14.6 ha; Table 2). This same pattern did not hold for core area (50% KUD) estimates, which were largest in the winter (4.0 ha) and smallest during the summer (2.0 ha) (Table 2). White sucker seasonal home ranges and core areas were comprised almost entirely of lake habitat with a depth ≤ 10 m, with a range of [0.93, 1.00] for the proportion of ≤ 10 m habitat in core areas and [0.92, 0.96] for that in home ranges. White sucker home ranges and core areas were consistently observed in similar areas of the lake across seasons (Fig. 5). The home ranges included the majority of the northwestern bay of the lake along with the southeastern bay and most of the southern shore of the main central (Fig. 5). Large portions of the north shore of the central basin were not encompassed by the 95% KUD estimates of home range in any season.
The proportion of overlap among seasonal home ranges relative to the pairwise mean home range was high across all season pairs, ranging from 0.74 to 0.82 (Table 3). White sucker core areas during the spring, summer, and fall seasons showed high levels of proportional overlap (0.36 – 0.55), while the winter seasonal core area showed the least overlap with the other seasons (0.11– 0.18). White suckers exhibited strong localized space use across all seasons as the mean proportions of overlap between individual- and group-level core areas ranged from 0.12 to 0.22 indicating that individual white suckers used a small portion of the total area used by all the white suckers (Fig. 6). Seasonal means of daily horizontal movement rates ranged from 2.10 km/day to 3.16 km/day (overall mean ± SD: 2.6 ± 0.5 km/day) and differed significantly among seasons (KW χ2 = 224.25, df = 4, p < 0.001). Tagged white suckers had higher movement rates in the spring and summer relative to the winter and fall (Table 2; Fig. 6). Vertical movement rates also differed among seasons (KW χ2 = 222.19, df = 4, p < 0.001) and were lower in the winter (25.0 m/day) relative to the spring, summer, and fall (median: 41.8 m/day, respectively) (Table 2).
Discussion
In this study, we investigated seasonal patterns of habitat use, temperature selection and activity by white suckers, a common freshwater species across North America that is often overlooked, despite their dominance in terms of relative fish biomass in many Boreal Shield lakes (Kelso 1985; Trippel and Harvey 1987). White suckers were consistently observed in shallow habitats (< 10 m depth) and rarely used cold, deeper waters, apart from winter, when they tended towards slightly deeper depths. Often categorized as a cool-water fish species, the Turkey Lake white suckers had a wide temperature preference range [10.0, 18.0°C] during the open water season. The white suckers exhibited a strong affinity for nearshore benthic habitat across the year-long study period. Although the total daily distance travelled by white suckers averaged 2–3 km, equivalent to almost twice the length of the study lake (max length ~ 1.6 km), they exhibited consistent spatial habitat use patterns across all seasons, leaving large portions of nearshore areas of the lake unexplored.
The thermal habitat preferences of white suckers are not well characterized from wild populations, limiting our understanding of the ecological niche of this underappreciated fish species. White suckers are cool-water fish based on classifications of thermal fish guilds in the literature (Coker et al. 2001; McMeans et al. 2020). However, they tended to select for colder temperatures in our study (< 16 °C), even when warmer thermal habitats were available. Relative to past studies investigating temperature preferences of white suckers (Wismer and Christie 1987), the results from our study suggest that white suckers prefer colder waters than previously documented. Hasnain et al. (2013) estimated that the final temperature preferendum for white suckers is 23.4 °C based on a literature review that included both field and lab observations (Wismer and Christie 1987). Summer water temperatures in Turkey Lake reached a maximum of 24.6 °C; however, warm waters (> 18 °C) were not preferentially selected by tagged white suckers in our study when they were available. The utilization of colder than expected thermal habitat could promote energy conservation in the white suckers due to lower metabolic costs (Shuter et al. 2012). Alternatively, white sucker habitat selection is driven by the availability of optimal benthic habitat, and associated resources, rather than thermal habitat. The contrast in white sucker temperature preference among studies highlights the need to conduct field studies that evaluate ecological patterns for fish species across different ecosystems and latitudes to have a better understanding of population-level differences in fish habitat use patterns.
Fish that inhabit temperate ecosystems often display seasonal habitat use patterns due to the dynamic fluxes in environmental conditions (Blanchfield et al. 2009; Croft-White et al. 2023). The inter-season variability in temperature preference of white suckers could be attributed to the seasonal changes in the availability of different thermal habitats. However, for many of the other metrics we examined, such as depth occupancy, space use (i.e. home range and core area sizes), and movement rates, the white suckers exhibited remarkably consistent patterns across all seasons. Previous research focused on the movement of white suckers in the Toronto Harbour of Lake Ontario (3,813 ha of 1.83 million ha; Ontario, Canada) found that white suckers exhibited consistent habitat use patterns in spatial and depth habitat use in the Outer Harbor regions, albeit with some seasonal variability (Midwood et al. 2019; Brownscombe et al. 2023). However, this telemetry array in the urban Toronto Harbour covered a larger area relative to our study at Turkey Lake and did not have complete coverage of the entire study area by using a fine-scale array. In contrast, a recent study in a small boreal lake (8.4 ha) on the seasonal patterns of habitat use by lake whitefish, a cold-water benthic feeder, showed variation in seasonal movement rates and much greater diversity of depth occupancy by individual fish in winter (Rodrigues et al. 2022). We did observe that white suckers were at slightly greater depths during the winter months compared to the open water period of the year, but overall their habitat selection and behaviour remained similar across seasons.
The spatial location of home ranges and core areas within Turkey Lake were virtually unchanged throughout the year. The high overlap in home range area (> 70% across seasons) demonstrate that white sucker exhibit high site fidelity to certain areas of the lake throughout the year. Our findings are similar to those from Doherty et al. (2010), who found that adult white suckers in the Saint John River watershed in New Brunswick, Canada also displayed strong site fidelity across seasons except for the spawning period in the spring. The home range of the white suckers in our study was considerably lower than the size of the entire lake, and at most the tagged fish used ~ 35% of the lake area across all seasons. The areas most frequently selected by white sucker included the northwestern and southeastern bays while avoiding areas such as the north shore of the main basin. There were some white sucker detections along the north shore, indicating infrequent forays in these areas, but the tagged fish did not spend a large amount of time in certain habitats. Previous studies have made no indications that there is restricted habitat availability within Turkey Lake (see Kelso 1985; Jeffries et al. 1988) yet white suckers appear highly selective in their habitat use patterns. A recent study found that benthivorous brook trout shift habitat use in winter to areas that are more profitable for foraging (Pépino et al. 2024). We did not find this type of seasonal shift in habitat use by white suckers in Turkey Lake, despite the fact that their seasonal home ranges were almost exclusively comprised of nearshore habitat ([92.1, 96.2%]) that encompassed 39.1 to 51.5% of the total available nearshore habitat. Possible explanations for the selection of specific nearshore habitat and high site fidelity could include the clustering of resources in certain areas of the lake or the overall limited habitat availability for white sucker in this ecosystem. Further research into the abiotic and biotic requirements for white sucker habitat could help determine the amount of available suitable habitat. White sucker tended to occupy areas that were shallow and relatively flat (i.e., northwest and southeastern bays) while avoiding the main basin that is characterized by deep waters and steeper nearshore slopes. The avoidance of certain areas was particularly surprising given that white suckers displayed high rates of total daily movement (2–3 km/day) that were greater than the length of Turkey Lake. The high site fidelity and lack of seasonal habitat use patterns contrast observations of other white sucker populations (Brownscombe et al. 2023) and other temperate fish species, such as lake trout and smallmouth bass Micropterus dolomieu (Bloomfield et al. 2022; Blanchfield et al. 2023). Likewise, tagged white suckers in our study remained highly active during the winter based on their space use and movement patterns, in contrast to the reduction in activity most commonly observed during winter conditions for fishes in Boreal Shield lakes (reviewed in Marsden et al. 2021).
White suckers and other catostomid species are known for their spawning migrations from lakes and larger bodies of water into small tributaries during spring freshets following post ice-off conditions (Cooke et al. 2005). Therefore, we expected the movement and space use of the tagged white suckers to be the greatest during the spring to reflect their spawning behaviour (Midwood et al. 2019). Consistent with this expectation, white sucker home range size, horizontal movement rates, and distance from the lake bottom were greatest during the spring compared to all other seasons. Together, these findings suggest that in the spring, white suckers are more mobile and spend more time involved in movements not associated with feeding (i.e. close to the lake bottom). These behavioural changes exhibited by white suckers in the spring are consistent with what one would expect for spawning fishes, and all tagged individuals in our study were of sufficient size to be sexually mature (Scott and Crossman 1973). From a foraging perspective, an increase in the movement of white suckers during the spring following the winter could be in response to the increased availability of suitable thermal habitat and associated resources after a prolonged period of suboptimal thermal conditions and limited resources.
A primary goal of this study was to investigate seasonal fish movement and habitat use within the lake proper, and as such, we did not monitor adjoining tributaries as both the primary inlet and outlet were shallow (< 0.5 m depth) and challenging to navigate due to boulders and woody debris. However, we observed that two of the six tagged white suckers disappeared and reappeared from the Turkey Lake telemetry array in late May, and another tagged fish was lost near the inlet at the same time, which is likely indicative of these fish moving into a tributary to spawn. Based on the position of Turkey Lake within the watershed (see Webster et al. 2021), we would expect white suckers to spawn in the upstream tributary that connects Little Turkey Lake and Turkey Lake. The slight increase in spatial use of the lake area surrounding the inlet during the spring could indicate spawning activity in this area or movement into the tributary. On the other hand, three other tagged fish were consistently detected in Turkey Lake during the spring, which raises more questions about white sucker spawning behaviour, such as the possibility of some fish either spawning within the lake or perhaps skip spawning (Rideout and Tomkiewicz 2011).
While our study provides novel insight into white sucker habitat use patterns, we recognize that our findings are based on a limited number of tagged individuals from a single lake population. Future studies can address this limitation by increasing the number of tagged fish to make stronger population inferences and evaluating habitat use and behaviour patterns across multiple populations in different ecosystems. As the distribution of white suckers spans a large geographic area (i.e., the majority of North America), there may be population-level differences in temperature preferences, particularly when considering the latitudinal gradient that white suckers inhabit. The majority of field observations have occurred at slightly lower latitudes, such as southern Ontario (Corbett and Powles 1983), Wisconsin (Coutant 1977), and Michigan (Wismer and Christie 1987) (approximately 44–46° N). Our findings from Turkey Lake (84.42°W, 47.05°N) showed some differences in temperature selection from these other white sucker studies, which may suggest that more northern populations tolerate or prefer cooler temperatures than previously described. Additionally, our study focuses on white sucker telemetry data collected across one year, and therefore we are unable to investigate any potential interannual variation in white sucker habitat-use behaviour. Future research could investigate whether seasonal behaviour is consistent across years despite potential annual fluctuations in environmental conditions.
Our study focused on the habitat use and behaviour of white suckers in lakes and advances our understanding of the ecology of this underappreciated species. Specifically, our key findings include the use of cooler than expected water temperatures and evidence for spatially constrained areas of occupancy that were consistent throughout the year. The results from our study can be subsequently applied in management scenarios to ensure the health and viability of white sucker populations across North America. White suckers and other catostomid species are susceptible to many ecological concerns, including invasive species, habitat degradation, and environmental contaminants (Cooke et al. 2005), and therefore furthering our knowledge of their ecology is paramount to address current and future management issues surrounding these species. White suckers displayed a strong affinity for nearshore habitat in a small boreal lake, and the maintenance of these habitats should be considered to maintain healthy white sucker populations that are an integral component of aquatic ecosystems. White sucker activity was highly concentrated in certain areas of the lake; thus, future research is needed to understand the key drivers of white sucker habitat use and which environmental conditions are prioritized in white sucker habitat selection preferences. Under future climate change projections, the extension of the growing season will likely result in the prolonged availability of optimal thermal habitat for white suckers in cooler systems like Turkey Lake (Guzzo and Blanchfield 2017). However, if warming nearshore temperatures force white suckers into deeper areas of the lake they may come into more direct competition with other benthivorous species, such as burbot (Scott and Crossman 1973; Cott et al. 2013) and lake whitefish (Johnston et al. 2019; Rodrigues et al. 2022), and could be negatively affected due to an increase in interactions with other species and/or the disruption of established temporal niche partitioning dynamics (McMeans et al. 2020). Additional research that examines seasonal species interactions in boreal fish communities containing white suckers would further aid management efforts for this underappreciated species.
Data availability
The data collected by Fisheries and Oceans Canada are available upon request. Please contact the first author (ian.richter@dfo-mpo.gc.ca) for details about the code or data used in this study.
References
Barton K, Barton MK (2019) Package “muMIn.” Version 1(18):439
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. arXiv preprint arXiv:14065823. https://doi.org/10.48550/arXiv.1406.5823
Begley M, Coghlan SM Jr, Zydlewski J (2017) A comparison of age, size, and fecundity of harvested and reference white sucker populations. N Am J Fish Manag 37:510–523. https://doi.org/10.1080/02755947.2017.1290719
Bertolo A, Magnan P (2005) The relationship between piscivory and growth of white sucker (Catostomus commersoni) and yellow perch (Perca flavescens) in headwater lakes of the Canadian shield. Can J Fish Aquat Sci 62:2706–2715. https://doi.org/10.1139/f05-174
Blanchfield PJ, Flavelle LS, Hodge TF, Orihel DM (2005) The response of lake trout to manual tracking. Trans Am Fish Soc 134:346–355. https://doi.org/10.1577/T04-048.1
Blanchfield PJ, Tate LS, Plumb JM et al (2009) Seasonal habitat selection by lake trout (Salvelinus namaycush) in a small Canadian Shield Lake: constraints imposed by winter conditions. Aquat Ecol 43:777–787. https://doi.org/10.1007/s10452-009-9266-3
Blanchfield PJ, McKee G, Guzzo MM et al (2023) Seasonal variation in activity and nearshore habitat use of Lake Trout in a subarctic lake. Mov Ecol 11:54. https://doi.org/10.1186/s40462-023-00417-x
Blazer VS, Walsh H, Braham R et al (2017) Tumours in white suckers from Lake Michigan tributaries: pathology and prevalence. J Fish Dis 40:377–393. https://doi.org/10.1111/jfd.12520
Bloomfield EJ, Guzzo MM, Middel TA et al (2022) Seasonality can affect ecological interactions between fishes of different thermal guilds. Front Eco Evol 10:986459. https://doi.org/10.3389/fevo.2022.986459
Brett J, Groves T (1979) Physiological energetics. In: Hoar WS, Randall DF, Brett JR (eds) Fish physiology, vol 8. Academic Press, New York, pp 279–352
Brodeur P, Magnan P, Legault M (2001) Response of fish communities to different levels of white sucker (Catostomus commersoni) biomanipulation in five temperate lakes. Can J Fish Aquat Sci 58:1998–2010. https://doi.org/10.1139/f01-137
Brownscombe JW, Midwood JD, Doka SE, Cooke SJ (2023) Telemetry-based spatial–temporal fish habitat models for fishes in an urban freshwater harbour. Hydrobiol 850:1779–1800. https://doi.org/10.1007/s10750-023-05180-z
Charles C, Blanchfield PJ, Gillis DM (2017) Site fidelity of escaped rainbow trout to an experimental freshwater aquaculture facility and habitat overlap with native fish fauna. Aquacult Environ Interact 9:415–428. https://doi.org/10.3354/aei00243
Christie GC, Regier HA (1988) Measures of optimal thermal habitat and their relationship to yields for four commercial fish species. Can J Fish Aquat Sci 45:301–314. https://doi.org/10.1139/f88-036
Chu C, Lester NP, Giacomini HC et al (2016) Catch-per-unit-effort and size spectra of lake fish assemblages reflect underlying patterns in ecological conditions and anthropogenic activities across regional and local scales. Can J Fish Aquat Sci 73:535–546. https://doi.org/10.1139/cjfas-2015-0150
Cincotta D, Stauffer J (1984) Temperature preference and avoidance studies of six North American freshwater fish species. Hydrobiologia 109:173–177. https://doi.org/10.1007/BF00011576
Coker G, Portt CB, Minns CK (2001) Morphological and ecological characteristics of Canadian freshwater fishes. Fisheries and Oceans Canada Burlington, Ontario
Colavecchia MV, Hodson PV, Parrott JL (2006) CYP1A induction and blue sac disease in early life stages of white suckers (Catostomus commersoni) exposed to oil sands. J Toxicol Environ Health A 69:967–994. https://doi.org/10.1080/15287390500362154
Cooke SJ, Bunt CM, Hamilton SJ et al (2005) Threats, conservation strategies, and prognosis for suckers (Catostomidae) in North America: insights from regional case studies of a diverse family of non-game fishes. Biol Conserv 121:317–331. https://doi.org/10.1016/j.biocon.2004.05.015
Corbett B, Powles P (1983) Spawning and early-life ecological phases of the white sucker in Jack Lake, Ontario. Trans Am Fish Soc 112:308–313. https://doi.org/10.1577/1548-8659(1983)112<308:SAEEPO>2.0.CO;2
Cote D, Tibble B, Curry R et al (2020) Seasonal and diel patterns in activity and habitat use by brook trout (Salvelinus fontinalis) in a small Newfoundland lake. Environ Biol Fish 103:31–47. https://doi.org/10.1007/s10641-019-00931-1
Cott PA, Johnston TA, Gunn JM (2013) Stability in life history characteristics among burbot populations across environmental gradients. Trans Am Fish Soc 142:1746–1756. https://doi.org/10.1080/00028487.2013.811101
Cott PA, Guzzo MM, Chapelsky AJ et al (2015) Diel bank migration of burbot (Lota lota). Hydrobiologia 757:3–20. https://doi.org/10.1007/s10750-015-2257-6
Coutant CC (1977) Compilation of temperature preference data. J Fish Board Can 34:739–745. https://doi.org/10.1139/f77-115
Croft-White MV, Larocque SM, Reddick DT et al (2023) Diversity of movement patterns of Longnose Gar tracked in coastal waters of western Lake Ontario. Environ Biol Fish: 1–16. https://doi.org/10.1007/s10641-023-01491-1
Doherty CA, Curry RA, Munkittrick KR (2010) Spatial and temporal movements of white sucker: implications for use as a sentinel species. Trans Am Fish Soc 139:1818–1827. https://doi.org/10.1577/T09-172.1
Duong T, Wand M, Chacon J, Gramacki A (2022) R package "ks": kernel-smoothing. R Package Version 1.13.5. https://cran.r-project.org/web/packages/ks/index.html. Accessed 5 July 2023
Fee E, Hecky R, Kasian S, Cruikshank D (1996) Effects of lake size, water clarity, and climatic variability on mixing depths in Canadian Shield lakes. Limnol Oceanogr 41:912–920. https://doi.org/10.4319/lo.1996.41.5.0912
Gibbons WN, Munkittrick KR, McMaster ME, Taylor WD (1998) Monitoring aquatic environments receiving industrial effluents using small fish species 2: Comparison between responses of trout-perch (Percopsis omiscomaycus) and white sucker (Catostomus commersoni) downstream of a pulp mill. Environ Toxicol Chemistry: an Int J 17:2238–2245. https://doi.org/10.1002/etc.5620171114
Guzzo MM, Blanchfield PJ (2017) Climate change alters the quantity and phenology of habitat for lake trout (Salvelinus namaycush) in small Boreal Shield lakes. Can J Fish Aquat Sci 74:871–884. https://doi.org/10.1139/cjfas-2016-0190
Guzzo MM, Blanchfield PJ, Chapelsky AJ, Cott PA (2016) Resource partitioning among top-level piscivores in a sub-Arctic lake during thermal stratification. J Great Lakes Res 42:276–285. https://doi.org/10.1016/j.jglr.2015.05.014
Guzzo MM, Blanchfield PJ, Rennie MD (2017) Behavioral responses to annual temperature variation alter the dominant energy pathway, growth, and condition of a cold-water predator. Proc Natl Acad Sci USA 114:9912–9917. https://doi.org/10.1073/pnas.170258411
Harrison P, Gutowsky L, Martins E et al (2016) Temporal plasticity in thermal-habitat selection of burbot Lota lota a diel-migrating winter-specialist. J Fish Biol 88:2111–2129. https://doi.org/10.1111/jfb.12990
Hasnain SS, Shuter BJ, Minns CK (2013) Phylogeny influences the relationships linking key ecological thermal metrics for North American freshwater fish species. Can J Fish Aquat Sci 70:964–972. https://doi.org/10.1139/cjfas-2012-0217
Hubbard JA, Hickie BE, Bowman J et al (2021) No long-term effect of intracoelomic acoustic transmitter implantation on survival, growth, and body condition of a long-lived stenotherm in the wild. Can J Fish Aquat Sci 78:173–183. https://doi.org/10.1139/cjfas-2020-0106
Huff A, Thomas A (2014) Lake Superior climate change impacts and adaptation. In: Prepared for the Lake Superior Lakewide action and management plan – superior work group. Available from: http://www.michigan.gov/documents/deq/Lake_Superior_Climate_Change_Impacts_and_Adaptation_445176_7.pdf
Jeffries DS, Kelso JR, Morrison IK (1988) Physical, chemical, and biological characteristics of the Turkey Lakes Watershed, central Ontario, Canada. Can J Fish Aquat Sci 45:s3–s13. https://doi.org/10.1139/f88-262
Johnston TA, Ehrman A, Hamilton GL et al (2019) Plenty of room at the bottom: niche variation and segregation in large-bodied benthivores of boreal lakes. Can J Fish Aquat Sci 76:1411–1422. https://doi.org/10.1139/cjfas-2018-0180
Jones NE, Mackereth RW (2016) Resource subsidies from adfluvial fishes increase stream productivity. Freshwater Biol 61:991–1005. https://doi.org/10.1111/fwb.12762
Jones NE, McKenzie M (2023) Spatial patterns of resource subsidies in Great Lakes tributaries from migratory fishes. J Great Lakes Res 102272:102272. https://doi.org/10.1016/j.jglr.2023.102272
Kelso JR (1985) Standing stock and production of fish in a cascading lake system on the Canadian Shield. Can J Fish Aquat Sci 42:1315–1320. https://doi.org/10.1139/f85-164
Kidd K, Hesslein R, Fudge R, Hallard K (1995) The influence of trophic level as measured by δ 15 N on mercury concentrations in freshwater organisms. Water Air Soil Pollut 80:1011–1015. https://doi.org/10.1007/BF01189756
Kuznetsova A, Brockhoff PB, Christensen RHB (2017) lmerTest package: tests in linear mixed effects models. J Stat Soft 82:1–26. https://doi.org/10.18637/jss.v082.i13
Logan C, Trippel E, Beamish F (1991) Thermal stratification and benthic foraging patterns of white sucker. Hydrobiol 213:125–132. https://doi.org/10.1007/BF00014998
Margenau TL (2007) Effects of angling with a single-hook and live bait on muskellunge survival. In: Diana JS, Margenau TL (eds) The Muskellunge Symposium: a memorial tribute to E.J. Crossman. Developments in environmental biology of fishes, vol 26. Springer, Dordrecht, pp 155–162. https://doi.org/10.1007/978-1-4020-6049-6_14
Marsden JE, Blanchfield PJ, Brooks JL et al (2021) Using untapped telemetry data to explore the winter biology of freshwater fish. Rev Fish Biol Fisheries 31:115–134. https://doi.org/10.1007/s11160-021-09634-2
McMaster ME, Tetreault GR, Clark T et al (2020) Baseline white sucker health and reproductive endpoints for use in assessment of further development in the Alberta Oil Sands. Int J Environ Impacts 3:219–237. https://doi.org/10.2495/EI-V3-N3-219-237
McMeans BC, McCann KS, Guzzo MM et al (2020) Winter in water: differential responses and the maintenance of biodiversity. Ecol Lett 23:922–938. https://doi.org/10.1111/ele.13504
Mejri S, Tremblay R, Vandenberg G, Audet C (2019) Novel feed from invasive species is beneficial to Walleye aquaculture. N Am J Aquacult 81:3–12. https://doi.org/10.1002/naaq.10063
Midwood JD, Rous AM, Doka SE, Cooke SJ (2019) Acoustic telemetry in Toronto Harbour: assessing residency, habitat selection, and within-harbour movements of fishes over a five-year period. Fisheries and Oceans Canada, Great Lakes Laboratory for Fisheries and Aquatic Sciences. https://publications.gc.ca/collections/collection_2019/mpo-dfo/Fs97-6-3331-eng.pdf. Accessed 15 Dec 2023
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142. https://doi.org/10.1111/j.2041-210x.2012.00261.x
Olson DE (1963) Role of the white sucker in Minnesota waters. J Minnesota Acad Sci 31:68–73
Pépino M, Magnan P, Leroux R, Bertolo A (2024) Thermal habitat fragmentation in stratified lakes induces resource waves that brook charr track across seasons. Oikos e10539. https://doi.org/10.1111/oik.10539
Pierce RB, Carlson AJ, Carlson BM et al (2013) Depths and thermal habitat used by large versus small northern pike in three Minnesota lakes. Trans Am Fish Soc 142:1629–1639. https://doi.org/10.1080/00028487.2013.822422
Plumb JM, Blanchfield PJ (2009) Performance of temperature and dissolved oxygen criteria to predict habitat use by lake trout (Salvelinus namaycush). Can J Fish Aquat Sci 66:2011–2023. https://doi.org/10.1139/F09-129
Reid D, Code T, Reid A, Herrero S (1994) Food habits of the river otter in a boreal ecosystem. Can J Zool 72:1306–1313. https://doi.org/10.1139/z94-174
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225. https://doi.org/10.2307/2409177
Richter IA, Smokorowski KE, Blanchfield PJ (2024) Incorporating vertical movement of fishes in habitat use models. J Fish Bio https://doi.org/10.1111/jfb.15857
Rideout RM, Tomkiewicz J (2011) Skipped spawning in fishes: more common than you might think. Mar Coastal Fish 3:176–189. https://doi.org/10.1080/19425120.2011.556943
Ridgway MS, Bell AH, Lacombe NA et al (2023) Thermal niche and habitat use by co-occurring lake trout (Salvelinus namaycush) and brook trout (S. fontinalis) in stratified lakes. Environ Biol Fish 106:941–955. https://doi.org/10.1007/s10641-022-01368-9
Rodrigues TH, Chapelsky AJ, Hrenchuk LE et al (2022) Behavioural responses of a cold-water benthivore to loss of oxythermal habitat. Environ Biol Fish 105:1489–1507. https://doi.org/10.1007/s10641-022-01335-4
Rogers KB, White GC (2007) Analysis of movement and habitat use from telemetry data. In: Guy CS, Brown ML (eds) Analysis and interpretation of freshwater fisheries data. American Fisheries Society, Bethesda, pp 625–676
Saint-Jacques N, Harvey HH, Jackson DA (2000) Selective foraging in the white sucker (Catostomus commersoni). Can J Zool 78:1320–1331. https://doi.org/10.1139/z00-067
Satterthwaite FE (1946) An approximate distribution of estimates of variance components. Bio Bull 2:110–114. https://doi.org/10.2307/3002019
Schulte PM (2015) The effects of temperature on aerobic metabolism: towards a mechanistic understanding of the responses of ectotherms to a changing environment. J Exp Biol 218:1856–1866. https://doi.org/10.1242/jeb.118851
Scott WB, Crossman EJ (1973) Freshwater fishes of Canada. Fish Res Board Can 184:1–966
Shuter B, Finstad A, Helland I et al (2012) The role of winter phenology in shaping the ecology of freshwater fish and their sensitivities to climate change. Aquat Sci 74:637–657. https://doi.org/10.1007/s00027-012-0274-3
Smith F (2013) Understanding HPE in the VEMCO positioning system (VPS). http://vemco.com/wp-content/uploads/2013/09/understanding-hpe-vps.pdf. Accessed 10 Dec 2023
Smith DA, Jackson DA, Ridgway MS (2020) Thermal habitat of brook trout in lakes of different size. Freshw Sci 39:56–69. https://doi.org/10.1086/707488
Somers KM, Harvey HH (1984) Alteration of fish communities in lakes stressed by acid deposition and heavy metals near Wawa, Ontario. Can J Fish Aquat Sci 41:20–29. https://doi.org/10.1139/f84-002
Todd CS, Young L, Owen Jr RB, Gramlich FJ (1982) Food habits of bald eagles in Maine. J Wildl Manage: 636–645. https://doi.org/10.2307/3808554
Trippel EA, Harvey HH (1987) Abundance, growth, and food supply of white suckers (Catostomus commersoni) in relation to lake morphometry and pH. Can J Zool 65:558–564. https://doi.org/10.1139/z87-086
Tuononen EI, Cooke SJ, Lédée EJ et al (2022) Interactions of a temperate North American fish community with a very low head hydropower facility in Ontario, Canada. River Res Appl 38:657–669. https://doi.org/10.1002/rra.3930
Ward TD, Brownscombe JW, Gutowsky LFG et al (2017) Electric fish handling gloves provide effective immobilization and do not impede reflex recovery of adult largemouth bass. North Am J Fish Manag 37:652–659. https://doi.org/10.1080/02755947.2017.1303558
Webster KL, Leach JA, Hazlett PW et al (2021) Turkey Lakes Watershed, Ontario, Canada: 40 years of interdisciplinary whole-ecosystem research. Hydrol Processes 35:e14109. https://doi.org/10.1002/hyp.14109
Wismer DA, Christie AE (1987) Temperature relationships of great lakes fishes: a data compilation. Great Lakes Fish Comm Spec Publ, Ann Arbor, pp 87–3
Worton BJ (1989) Kernel methods for estimating the utilization distribution in home-range studies. Ecology 70:164–168. https://doi.org/10.2307/1938423
Acknowledgements
We would like to thank Evan Timusk, Rick Elsner, Gabrielle Perugini, Reagan Davis, and Marko Danailovski for data collection and Murray Mackay for leadership in the deployment and operation of the Turkey Lake climate raft and lake temperature data. The Fisheries and Oceans Canada Animal Care Committee approved this study's collection, handling, and surgical methods (Protocol number: OPA-ACC-2022-21; GWACC-130 – Fish tagging protocol) . Thank you to Innovasea Systems Inc. for their support and data analysis of the telemetry system. Funding was provided by Fisheries and Oceans Canada. We thank the reviewers for constructive comments that have improved this manuscript.
Funding
Open access funding provided by Fisheries & Oceans Canada library.
Author information
Authors and Affiliations
Contributions
PB and KS conceived the whole-ecosystem research program. All authors contributed to the data collection. IR and PB contributed to the conception and design of the study. IR lead the writing and data analysis. PB and KS contributed support for the methodology, interpretation of the results, and the writing of the manuscript (review and editing).
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Richter, I.A., Smokorowski, K.E. & Blanchfield, P.J. Seasonal habitat use of white sucker Catostomus commersonii in a small Boreal lake. Environ Biol Fish (2024). https://doi.org/10.1007/s10641-024-01581-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10641-024-01581-8