Summary
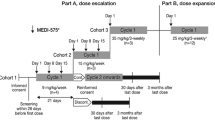
Alofanib is a small-molecule allosteric extracellular FGFR2 inhibitor. We report safety and preliminary efficacy from the first-in-human phase 1b study of alofanib in heavily pretreated patients with advanced gastric cancer. The standard dose-escalation design 3+3 aimed to establish the maximum tolerated dose (MTD) or recommended phase 2 dose (RP2D). Alofanib was administered daily intravenously 5 days on, 2 days off. There were five dose levels (50-350 mg/m2). All patients received alofanib until disease progression or unacceptable toxicity. 21 patients were enrolled. Patients were predominantly male (71%), 67% had 2 and more metastatic sites, including liver metastases (43%), 19% had ECOG PS 2, and were heavily pretreated (86% had previous 2 and more treatment lines). During dose escalation, no dose-limiting toxicities were observed, and MTD was not defined. 15 (71.4%) patients had at least one adverse event associated with the treatment (TRAE). Grade 3 or higher TRAEs were observed in 6 patients (28.6%). The most common TRAEs included reactions immediately after administration, diarrhea, thrombocytopenia, arthralgia, and headache. The median progression-free survival and overall survival was 3.63 (95% CI 1.58–5.68) and 7.0 (95% CI 3.82–10.18) months, respectively. The 6- and 12-month overall survival rates were 57.1% and 33.3%. Disease control rate was 68% with one durable partial response. The MTD has not been reached and dose of 350 mg/m2, 5 days on, 2 days off has been declared as RP2D. Alofanib showed acceptable tolerability and preliminary signs of clinical activity in the late-line treatment of metastatic gastric cancer. (ClinicalTrials.gov identifier: NCT04071184).

Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Figueiredo C, Camargo MC, Leite M, Fuentes-Pananá EM, Rabkin CS, Machado JC (2017) Pathogenesis of Gastric Cancer: Genetics and Molecular Classification. Curr Top Microbiol Immunol 400:277–304. https://doi.org/10.1007/978-3-319-50520-6_12.Erratum.In:CurrTopMicrobiolImmunol.2017;400:E1
Seo S, Park SJ, Ryu MH et al (2017) Prognostic impact of fibroblast growth factor receptor 2 gene amplification in patients receiving fluoropyrimidine and platinum chemotherapy for metastatic and locally advanced unresectable gastric cancers. Oncotarget 8(20):33844–33854
Kim HS, Kim JH, Jang HJ (2019) Pathologic and prognostic impacts of FGFR2 amplification in gastric cancer: a meta-analysis and systemic review. J Cancer 10(11):2560–2567
Ahn S, Lee J, Hong M et al (2016) FGFR2 in gastric cancer: protein overexpression predicts gene amplification and high H-index predicts poor survival. Mod Pathol 29:1095–1103
Schrumpf T, Behrens HM, Haag J, Krüger S, Röcken C (2022) FGFR2 overexpression and compromised survival in diffuse-type gastric cancer in a large central European cohort. PLoS One 17(2)
Sengal AT, Smith D, Rogers R, Snell CE, Williams ED, Pollock PM (2021) Fibroblast Growth Factor Receptor 2 Isoforms Detected via Novel RNA ISH as Predictive Biomarkers for Progestin Therapy in Atypical Hyperplasia and Low-Grade Endometrial Cancer. Cancers (Basel) 13(7):1703
Ranieri D, Rosato B, Nanni M, Magenta A, Belleudi F, Torrisi MR (2016) Expression of the FGFR2 mesenchymal splicing variant in epithelial cells drives epithelial-mesenchymal transition. Oncotarget 7(5):5440–60
Sengal AT, Bonazzi V, Kondrashova O et al (2021) Targeting FGFR2c isoform, a novel therapeutic target with FGFR inhibitor in endometrial cancer [abstract]. In: Proceedings of the AACR-NCI-EORTC Virtual International Conference on Molecular Targets and Cancer Therapeutics; 2021 Oct 7-10. Philadelphia (PA): AACR; Mol Cancer Ther 2021;20(12 Suppl): Abstract nr LBA020
Pierce KL, Deshpande AM, Stohr BA et al (2014) FPA144, a humanized monoclonal antibody for both FGFR2-amplified and nonamplified, FGFR2b-overexpressing gastric cancer patients. JCO 32(15_suppl):e15074
Epstein RJ, Tian LJ, Gu YF (2021) 2b or Not 2b: How Opposing FGF Receptor Splice Variants Are Blocking Progress in Precision Oncology. J Oncol 30(2021):9955456
Krook MA, Reeser JW, Ernst G et al (2021) Fibroblast growth factor receptors in cancer: genetic alterations, diagnostics, therapeutic targets and mechanisms of resistance. Br J Cancer 124(5):880–892
Tsimafeyeu I, Ludes-Meyers J, Stepanova E et al (2016) Targeting FGFR2 with alofanib (RPT835) shows potent activity in tumour models. Eur J Cancer 61:20–8
Tyulyandina A, Harrison D, Yin W et al (2017) Alofanib, an allosteric FGFR2 inhibitor, has potent effects on ovarian cancer growth in preclinical studies. Invest New Drugs 35(2):127–133
Khochenkov DA, Solomko ES, Peretolchina NM, Ryabaya OO, Stepanova EV (2015) Antiangiogenic Activity of Alofanib, an Allosteric Inhibitor of Fibroblast Growth Factor Receptor 2. Bull Exp Biol Med 160(1):84–7
Hierro C, Alsina M, Sánchez M, Serra V, Rodon J, Tabernero J (2017) Targeting the fibroblast growth factor receptor 2 in gastric cancer: promise or pitfall? Ann Oncol 28(6):1207–1216
Abou-Alfa GK, Sahai V, Hollebecque A et al (2020) Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: a multicentre, open-label, phase 2 study. Lancet Oncol 21(5):671–684
Javle M, Roychowdhury S, Kelley RK et al (2021) Infigratinib (BGJ398) in previously treated patients with advanced or metastatic cholangiocarcinoma with FGFR2 fusions or rearrangements: mature results from a multicentre, open-label, single-arm, phase 2 study. Lancet Gastroenterol Hepatol 6(10):803–815
Lapina NV, Stosman KI, Melikhova MV et al (2019) Preclinical toxicology of alofanib, an allosteric inhibitor of fibroblast growth factor receptor 2. Malignant tumours 9(3):65-70. (In Russ.) https://doi.org/10.18027/2224-5057-2019-9-3-65-70
Lordick F, Carneiro F, Cascinu S et al (2022) Gastric cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol 33(10):1005–1020
Shitara K, Doi T, Dvorkin M et al (2018) Trifluridine/tipiracil versus placebo in patients with heavily pretreated metastatic gastric cancer (TAGS): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 19(11):1437-1448
Tabernero J, Shitara K, Dvorkin M et al (2018) Overall survival results from a phase III trial of trifluridine/tipiracil versus placebo in patients with metastatic gastric cancer refractory to standard therapies (TAGS). Ann Oncol 29(Supplement 5):v122–v123. https://doi.org/10.1093/annonc/mdy208.001
Tabernero J, Shitara K, Zaanan A et al (2021) Trifluridine/tipiracil versus placebo for third or later lines of treatment in metastatic gastric cancer: an exploratory subgroup analysis from the TAGS study. ESMO Open 6(4)
Wainberg ZA, Enzinger PC, Kang YK et al (2022) Bemarituzumab in patients with FGFR2b-selected gastric or gastro-oesophageal junction adenocarcinoma (FIGHT): a randomised, double-blind, placebo-controlled, phase 2 study. Lancet Oncol 23(11):1430–1440
Acknowledgements
We thank all investigators (Vyacheslav Kurakin, Natalia Trenina, Kristina Novoselova, Nikita Volkov, Natalia Abramova, Tatyana Titova, Ilya Agranov, Sergei Iugai, Ekaterina Obarevich, Alina Khashanova, Margarita Suetina, Aiyyna Nikiforova, Maria Mukhina, and Dmitry Reznikov) participating in the study and Kristina Zakurdaeva for her support in the development of the study protocol.
Funding
This work was supported by grant from Skolkovo Foundation (no grant number).
Author information
Authors and Affiliations
Contributions
Ilya Tsimafeyeu and Sergei Tjulandin conceived and designed the study and analysis. Ilya Tsimafeyeu wrote the first draft of the manuscript. Galina Statsenko, Liubov Vladimirova, Natalia Besova, Elena Artamonova, Ivan Rykov, Anastasia Mochalova, Igor Utyashev, and Vladimir Moiseyenko enrolled patients and collected data. Grigory Raskin performed pathological studies. Svetlana Gorbacheva and Vasily Kazey performed PK studies.Evgenia Gavrilova and Nadezhda Dragun monitored the study and conducted an internal audit. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
Evgenia Gavrilova and Nadezhda Dragun are employees of Ruspharmtech LLC.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tsimafeyeu, I., Statsenko, G., Vladimirova, L. et al. A phase 1b study of the allosteric extracellular FGFR2 inhibitor alofanib in patients with pretreated advanced gastric cancer. Invest New Drugs 41, 324–332 (2023). https://doi.org/10.1007/s10637-023-01340-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-023-01340-z