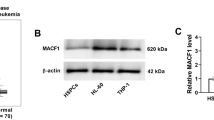
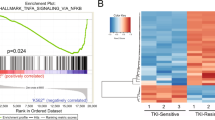
Summary
Stathmin 1 (STMN1) is a microtubule-destabilizing protein highly expressed in hematological malignancies and involved in proliferation and differentiation. Although a previous study found that the PML–RARα fusion protein, which contributes to the pathophysiology of acute promyelocytic leukemia (APL), positively regulates STMN1 at the transcription and protein activity levels, little is known about the role of STMN1 in APL. In this study, we aimed to investigate the STMN1 expression levels and their associations with laboratory, clinical, and genomic data in APL patients. We also assessed the dynamics of STMN1 expression during myeloid cell differentiation and cell cycle progression, and the cellular effects of STMN1 silencing and pharmacological effects of microtubule-stabilizing drugs on APL cells. We found that STMN1 transcripts were significantly increased in samples from APL patients compared with those of healthy donors (all p < 0.05). However, this had no effect on clinical outcomes. STMN1 expression was associated with proliferation- and metabolism-related gene signatures in APL. Our data confirmed that STMN1 was highly expressed in early hematopoietic progenitors and reduced during cell differentiation, including the ATRA-induced granulocytic differentiation model. STMN1 phosphorylation was predominant in a pool of mitosis-enriched APL cells. In NB4 and NB4-R2 cells, STMN1 knockdown decreased autonomous cell growth (all p < 0.05) but did not impact ATRA-induced apoptosis and differentiation. Finally, treatment with paclitaxel (as a single agent or combined with ATRA) induced microtubule stabilization, resulting in mitotic catastrophe with repercussions for cell viability, even in ATRA-resistant APL cells. This study provides new insights into the STMN1 functions and microtubule dynamics in APL.




Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Rowley JD, Golomb HM, Vardiman J, Fukuhara S, Dougherty C, Potter D (1977) Further evidence for a non-random chromosomal abnormality in acute promyelocytic leukemia. Int J Cancer 20:869–872
Melnick A, Licht JD (1999) Deconstructing a disease: RARalpha, its fusion partners, and their roles in the pathogenesis of acute promyelocytic leukemia. Blood 93:3167–3215
Avvisati G, Lo Coco F, Mandelli F (2001) Acute promyelocytic leukemia: clinical and morphologic features and prognostic factors. Semin Hematol 38:4–12
Reiter A, Lengfelder E, Grimwade D (2004) Pathogenesis, diagnosis and monitoring of residual disease in acute promyelocytic leukaemia. Acta Haematol 112:55–67
Wang ZY, Chen Z (2008) Acute promyelocytic leukemia: from highly fatal to highly curable. Blood 111:2505–2515
Sanz MA, Grimwade D, Tallman MS, Lowenberg B, Fenaux P, Estey EH, Naoe T, Lengfelder E, Buchner T, Dohner H, Burnett AK, Lo-Coco F (2009) Management of acute promyelocytic leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood 113:1875–1891
Zada AA, Geletu MH, Pulikkan JA, Muller-Tidow C, Reddy VA, Christopeit M, Hiddemann WD, Behre HM, Tenen DG, Behre G (2006) Proteomic analysis of acute promyelocytic leukemia: PML-RARalpha leads to decreased phosphorylation of OP18 at serine 63. Proteomics 6:5705–5719
Machado-Neto JA, Saad ST, Traina F (2014) Stathmin 1 in normal and malignant hematopoiesis. BMB Rep 47:660–665
Mistry SJ, Atweh GF (2001) Stathmin inhibition enhances okadaic acid-induced mitotic arrest: a potential role for stathmin in mitotic exit. J Biol Chem 276:31209–31215
Machado-Neto JA, de Melo CP, Favaro P, Lazarini M, Lorand-Metze I, Costa FF, Olalla Saad ST, Traina F (2014) Stathmin 1 is involved in the highly proliferative phenotype of high-risk myelodysplastic syndromes and acute leukemia cells. Leuk Res 38:251–257
Machado-Neto JA, de Melo CP, Favaro P, Lazarini M, da Silva Santos Duarte A, Lorand-Metze I, Costa FF, Saad ST, Traina F, (2015) Stathmin 1 inhibition amplifies ruxolitinib-induced apoptosis in JAK2V617F cells. Oncotarget 6:29573–29584
Carrour TL, Assou S, Tondeur S, Lhermitte L, Lamb N, Reme T, Pantesco V, Hamamah S, Klein B, De Vos J (2010) Amazonia!: An Online Resource to Google and Visualize Public Human whole Genome Expression Data. The Open Bioinformatics Journal 4:5–10
Sanz MA, Lo Coco F, Martin G, Avvisati G, Rayon C, Barbui T, Diaz-Mediavilla J, Fioritoni G, Gonzalez JD, Liso V, Esteve J, Ferrara F, Bolufer P, Bernasconi C, Gonzalez M, Rodeghiero F, Colomer D, Petti MC, Ribera JM, Mandelli F (2000) Definition of relapse risk and role of nonanthracycline drugs for consolidation in patients with acute promyelocytic leukemia: a joint study of the PETHEMA and GIMEMA cooperative groups. Blood 96:1247–1253
Rego EM, Kim HT, Ruiz-Arguelles GJ, Undurraga MS, Uriarte Mdel R, Jacomo RH, Gutierrez-Aguirre H, Melo RA, Bittencourt R, Pasquini R, Pagnano K, Fagundes EM, Chauffaille Mde L, Chiattone CS, Martinez L, Meillon LA, Gomez-Almaguer D, Kwaan HC, Garces-Eisele J, Gallagher R, Niemeyer CM, Schrier SL, Tallman M, Grimwade D, Ganser A, Berliner N, Ribeiro RC, Lo-Coco F, Lowenberg B, Sanz MA (2013) Improving acute promyelocytic leukemia (APL) outcome in developing countries through networking, results of the International Consortium on APL. Blood 121:1935–1943
Lin X, Qiao N, Shen Y, Fang H, Xue Q, Cui B, Chen L, Zhu H, Zhang S, Chen Y, Jiang L, Wang S, Li J, Wang B, Chen B, Chen Z, Chen S (2021) Integration of Genomic and Transcriptomic Markers Improves the Prognosis Prediction of Acute Promyelocytic Leukemia. Clin Cancer Res 27:3683–3694
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A 102:15545–15550
Kohl M, Wiese S, Warscheid B (2011) Cytoscape: software for visualization and analysis of biological networks. Methods Mol Biol 696:291–303
Maere S, Heymans K, Kuiper M (2005) BiNGO: a Cytoscape plugin to assess overrepresentation of gene ontology categories in biological networks. Bioinformatics 21:3448–3449
Lanotte M, Martin-Thouvenin V, Najman S, Balerini P, Valensi F, Berger R (1991) NB4, a maturation inducible cell line with t(15;17) marker isolated from a human acute promyelocytic leukemia (M3). Blood 77:1080–1086
Duprez E, Benoit G, Flexor M, Lillehaug JR, Lanotte M (2000) A mutated PML/RARA found in the retinoid maturation resistant NB4 subclone, NB4-R2, blocks RARA and wild-type PML/RARA transcriptional activities. Leukemia 14:255–261
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Vicari HP, Lima K, Gomes RDC, Fernandes DC, da Silva JCL, Rodrigues Junior MT, Barroso de Oliveira AS, Dos Santos RN, Andricopulo AD, Coelho F, Costa-Lotufo LV, Machado-Neto JA (2021) Synthetic cyclopenta[b]indoles exhibit antineoplastic activity by targeting microtubule dynamics in acute myeloid leukemia cells. Eur J Pharmacol 894
Belletti B, Baldassarre G (2011) Stathmin: a protein with many tasks. New biomarker and potential target in cancer. Expert Opin Ther Targets 15:1249–1266
Hanash SM, Strahler JR, Kuick R, Chu EH, Nichols D (1988) Identification of a polypeptide associated with the malignant phenotype in acute leukemia. J Biol Chem 263:12813–12815
Machado-Neto JA, Rodrigues Alves APN, Fernandes JC, Coelho-Silva JL, Scopim-Ribeiro R, Fenerich BA, da Silva FB, Scheucher PS, Simoes BP, Rego EM, Traina F (2017) Paclitaxel induces Stathmin 1 phosphorylation, microtubule stability and apoptosis in acute lymphoblastic leukemia cells. Heliyon 3
Ley TJ, Miller C, Ding L, Raphael BJ, Mungall AJ, Robertson A, Hoadley K, Triche TJ Jr, Laird PW, Baty JD, Fulton LL, Fulton R, Heath SE, Kalicki-Veizer J, Kandoth C, Klco JM, Koboldt DC, Kanchi KL, Kulkarni S, Lamprecht TL, Larson DE, Lin L, Lu C, McLellan MD, McMichael JF, Payton J, Schmidt H, Spencer DH, Tomasson MH, Wallis JW, Wartman LD, Watson MA, Welch J, Wendl MC, Ally A, Balasundaram M, Birol I, Butterfield Y, Chiu R, Chu A, Chuah E, Chun HJ, Corbett R, Dhalla N, Guin R, He A, Hirst C, Hirst M, Holt RA, Jones S, Karsan A, Lee D, Li HI, Marra MA, Mayo M, Moore RA, Mungall K, Parker J, Pleasance E, Plettner P, Schein J, Stoll D, Swanson L, Tam A, Thiessen N, Varhol R, Wye N, Zhao Y, Gabriel S, Getz G, Sougnez C, Zou L, Leiserson MD, Vandin F, Wu HT, Applebaum F, Baylin SB, Akbani R, Broom BM, Chen K, Motter TC, Nguyen K, Weinstein JN, Zhang N, Ferguson ML, Adams C, Black A, Bowen J, Gastier-Foster J, Grossman T, Lichtenberg T, Wise L, Davidsen T, Demchok JA, Shaw KR, Sheth M, Sofia HJ, Yang L, Downing JR, Eley G (2013) Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N Engl J Med 368:2059–2074
Jiang W, Huang S, Song L, Wang Z (2018) STMN1, a prognostic predictor of esophageal squamous cell carcinoma, is a marker of the activation of the PI3K pathway. Oncol Rep 39:834–842
Li M, Yang J, Zhou W, Ren Y, Wang X, Chen H, Zhang J, Chen J, Sun Y, Cui L, Liu X, Wang L, Wu C (2017) Activation of an AKT/FOXM1/STMN1 pathway drives resistance to tyrosine kinase inhibitors in lung cancer. Br J Cancer 117:974–983
Carlos J, Lima K, Costa-Lotufo LV, Leitao A, Machado-Neto JA (2021) AD80, a multikinase inhibitor, exhibits antineoplastic effects in acute leukemia cellular models targeting the PI3K/STMN1 axis. Invest New Drugs
Tang J, Luo Y, Wu G (2020) A glycolysis-related gene expression signature in predicting recurrence of breast cancer. Aging (Albany NY) 12:24983–24994
Xu Z, Xu L, Liu L, Li H, Jin J, Peng M, Huang Y, Xiao H, Li Y, Guan H (2021) A Glycolysis-Related Five-Gene Signature Predicts Biochemical Recurrence-Free Survival in Patients With Prostate Adenocarcinoma. Front Oncol 11
Iancu-Rubin C, Gajzer D, Tripodi J, Najfeld V, Gordon RE, Hoffman R, Atweh GF (2011) Down-regulation of stathmin expression is required for megakaryocyte maturation and platelet production. Blood 117:4580–4589
Ramlogan-Steel CA, Steel JC, Fathallah H, Iancu-Rubin C, Atweh GF (2021) Stathmin 1 deficiency induces erythro-megakaryocytic defects leading to macrocytic anemia and thrombocythemia in Stathmin 1 knock out mice. Blood Cells Mol Dis 87
Chappell J, Dalton S (2013) Roles for MYC in the establishment and maintenance of pluripotency. Cold Spring Harb Perspect Med 3
Holmfeldt P, Brannstrom K, Stenmark S, Gullberg M (2006) Aneugenic activity of Op18/stathmin is potentiated by the somatic Q18–>e mutation in leukemic cells. Mol Biol Cell 17:2921–2930
Holmfeldt P, Sellin ME, Gullberg M (2010) Upregulated Op18/stathmin activity causes chromosomal instability through a mechanism that evades the spindle assembly checkpoint. Exp Cell Res 316:2017–2026
Esposito MT, Zhao L, Fung TK, Rane JK, Wilson A, Martin N, Gil J, Leung AY, Ashworth A, So CW (2015) Synthetic lethal targeting of oncogenic transcription factors in acute leukemia by PARP inhibitors. Nat Med 21:1481–1490
Faraoni I, Compagnone M, Lavorgna S, Angelini DF, Cencioni MT, Piras E, Panetta P, Ottone T, Dolci S, Venditti A, Graziani G, Lo-Coco F (2015) BRCA1, PARP1 and gammaH2AX in acute myeloid leukemia: Role as biomarkers of response to the PARP inhibitor olaparib. Biochim Biophys Acta 1852:462–472
Barbuti AM, Chen ZS (2015) Paclitaxel Through the Ages of Anticancer Therapy: Exploring Its Role in Chemoresistance and Radiation Therapy. Cancers (Basel) 7:2360–2371
Acknowledgements
The authors thank John De Vos and Tanguy Le Carrour for led the initiative Amazonia! (http://amazonia.transcriptome.eu), providing a tool for analysis of genomic data. The authors also thank Professor Francisco Garcia Soriano (Internal Medicine Department, School of Medicine, University of São Paulo) for kindly providing the olaparib used in this study.
Funding
This study was supported by grant #2019/23864–7, #2017/24993–0, #2017/23117–1, #2020/12842–0 and #2019/01700–2 from the São Paulo Research Foundation (FAPESP), and grant #402587/2016–2 form the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Contributions
H.P.V and J.L.C.-S. designed, executed and analyzed the experiments and prepared the manuscript. D.A.P.-M., A.R.L.-A, K.L., J.C.L.S., and L.V.C.-L. provided inputs and participated in experiments. P.S.S. participated in flow cytrometry experiments and data analysis. L.C.K., R.A.M., R.B.B., K.P., E.N., E.M.F., F.K., L.L.F.-P., and E.M.R contributed recruiting patients and collecting data. F.T. and J.A.M.-N. supervised and participated in overall design of study, experiments and analyses, and prepared the manuscript. All authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Ethical approval
All procedures used were approved by the Ethics Committee of the Medical School of Ribeirao Preto, University of Sao Paulo, and by the National Commission of Ethics in Research, National Health Council, Ministry of Health (CONEP) (Registry# 12920; Process number: 13496/2005; CAAE: 155.0.004.000–05). Informed consent was obtained from all patients and approved by the Research Ethics Board. All methods were carried out in accordance with the approved guidelines and to the Declaration of Helsinki.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
Hugo Passos Vicari declares that he has no conflict of interest. Juan Luiz Coelho-Silva declares that he has no conflict of interest. Diego A. Pereira-Martins declares that he has no conflict of interest. Antônio Roberto Lucena-Araujo declares that he has no conflict of interest. Keli Lima declares that she has no conflict of interest. Jean Carlos Lipreri da Silva declares that he has no conflict of interest. Priscila Santos Scheucher declares that she has no conflict of interest. Luisa C. Koury declares that she has no conflict of interest. Raul A. de Melo declares that he has no conflict of interest. Rosane Bittencourt declares that she has no conflict of interest. Katia Pagnano declares that she has no conflict of interest. Elenaide Nunes declares that she has no conflict of interest. Evandro M. Fagundes declares that he has no conflict of interest. Fabio Kerbauy declares that he has no conflict of interest. Lorena Lobo de Figueiredo-Pontes declares that she has no conflict of interest. Leticia Veras Costa-Lotufo declares that she has no conflict of interest. Eduardo Magalhães Rego declares that he has no conflict of interest. Fabiola Traina declares that she has no conflict of interest. João Agostinho Machado-Neto declares that he has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vicari, H.P., Coelho-Silva, J.L., Pereira-Martins, D.A. et al. STMN1 is highly expressed and contributes to clonogenicity in acute promyelocytic leukemia cells. Invest New Drugs 40, 438–452 (2022). https://doi.org/10.1007/s10637-021-01197-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-021-01197-0