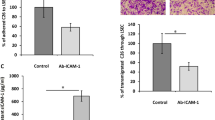
Summary
Liver metastasis is the major obstacle for prolonging the survival of colon cancer patients. Low-molecular-weight heparin (LMWH), a common drug for venous thromboembolism, has displayed beneficial effects in improving the survival of cancer patients, though the mechanism remains unclear. This study aimed to investigate the effects of LMWH on hepatic metastasis of colon cancer and its underlying molecular mechanism by targeting the interaction of the chemokine receptor CXCR4 and its ligand CXCL12 (formerly known as stromal cell-derived factor 1α, SDF-1α), as the CXCR4-CXCL12 axis has been shown to regulate the interaction of cancer cells and stroma. Experimental results revealed that LMWH (Enoxaparin, 3500–5500 Da) inhibited the CXCL12-stimulated proliferation, adhesion and colony formation of human colon cancer HCT-116 cells that highly expressed CXCR4. Interestingly, LMWH or an anti-CXCR4 blocking antibody diminished the migrating and invading abilities of HCT116 cells stimulated by the recombinant CXCL12 protein or liver homogenates which contained endogenous CXCL12 protein. Although LMWH did not significantly inhibit the growth of subcutaneous colon tumors, it significantly suppressed the formation of hepatic metastasis established by intrasplenic injection of colon cancer cells in nude Balb/c mice and also downregulated the expression of CXCL12 in hepatic sinusoidal endothelial cells. The results suggest that LMWH inhibits the formation of hepatic metastasis of colon cancer by disrupting the interaction of CXCR4 and CXCL12, supporting that perioperative administration of LMWH may help to prevent the seeding and subsequent growth of hepatic metastases of colon cancer cells.






Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ (2009) Cancer Statistics, 2009. CA Cancer J Clin 59:225–249. doi:10.3322/caac.20006
Mayo SC, Pawlik TM (2009) Current management of colorectal hepatic metastasis.Expert Rev. Gastroenterol Hepatol 3:131–144. doi:10.1586/egh.09.8
Rossi D, Zlotnik A (2000) The biology of chemokines and their receptors. Annu Rev Immunol 18:217–242
Ottaiano A, di Palma A, Napolitano M, Pisano C, Pignata S, Tatangelo F et al (2005) Inhibitory effects of anti-CXCR4 antibodies on human colon cancer cells. Cancer Immunol Immunother 54:781–791. doi:10.1007/s00262-004-0636-3
Zeelenberg IS, Ruuls-Van Stalle L, Roos E (2003) The chemokine receptor CXCR4 is required for outgrowth of colon carcinoma micrometastases. Cancer Res 63:3833–3839
Taichman RS, Cooper C, Keller ET, Pienta KJ, Taichman NS, McCauley LK (2002) Use of the stromal cell derived factor-1/CXCR4 pathway in prostate cancer metastasis to bone. Cancer Res 62:1832–1837
Smith MC, Luker KE, Garbow JR, Prior JL, Jackson E, Piwnica-Worms D, Luker GD (2004) CXCR4 regulates growth of both primary and metastatic breast cancer. Cancer Res 64:8604–8612. doi:10.1158/0008-5472.CAN-04-1844
Koshiba T, Hosotani R, Miyamoto Y, Ida J, Tsuji S, Nakajima S et al (2000) Expression of stromal cell-derived factor 1 and CXCR4 ligand receptor system in pancreatic cancer: a possible role for tumor progression. Clin Cancer Res 6:3530–3535
Marchesi F, Monti P, Leone BE, Zerbi A, Vecchi A, Piemonti L, Mantovani A, Allavena P (2004) Increased survival, proliferation, and migration in metastatic human pancreatic tumor cells expressing functional CXCR4. Cancer Res 64:8420–8427. doi:10.1158/0008-5472.CAN-04-1343
Phillips RJ, Burdick MD, Lutz M, Belperio JA, Keane MP, Strieter RM (2003) The stromal derived factor-1/CXCL12-CXC chemokine receptor 4 biological axis in non-small cell lung cancer metastases. Am J Respir Crit Care Med 167:1676–1686. doi:10.1164/rccm.200301-071OC
Kijima T, Maulik G, Ma PC, Tibaldi EV, Turner RE, Rollins B, Sattler M, Johnson BE, Salgia R (2002) Regulation of cellular proliferation, cytoskeletal function, and signal transduction through CXCR4 and c-Kit in small cell lung cancer cells. Cancer Res 62:6304–6311
Geminder H, Sagi-Assif O, Goldberg L, Meshel T, Rechavi G, Witz IP, Ben-Baruch A (2001) A possible role for CXCR4 and its ligand, the CXC chemokine stromal cell-derived factor-1, in the development of bone marrow metastases in neuroblastoma. J Immunol 167:4747–4757
Schimanski CC, Bahre R, Gockel I, Müller A, Frerichs K, Hörner V et al (2006) Dissemination of hepatocellular carcinoma is mediated via chemokine receptor CXCR4. Br J Cancer 95:210–217. doi:10.1038/sj.bjc.6603251
Liu H, Pan Z, Li A, Fu S, Lei Y, Sun H, Wu M, Zhou W (2008) Roles of chemokine receptor 4 (CXCR4) and chemokine ligand 12 (CXCL12) in metastasis of hepatocellular carcinoma cells. Cell Mol Immunol 5:373–378. doi:10.1038/cmi.2008.46
Sutton A, Friand V, Brulé-Donneger S, Chaigneau T, Ziol M, Sainte-Catherine O et al (2007) Stromal cell-derived factor-1/chemokine (C-X-C motif) ligand 12 stimulates human hepatoma cell growth, migration, and invasion. Mol Cancer Res 5:21–33. doi:10.1158/1541-7786.MCR-06-0103
Kim J, Takeuchi H, Lam ST, Turner RR, Wang HJ, Kuo C, Foshag L, Bilchik AJ, Hoon DS (2005) Chemokine receptor CXCR4 expression in colorectal cancer patients increases the risk for recurrence and for poor survival. J Clin Oncol 23:2744–2753. doi:10.1200/JCO.2005.07.078
Ottaiano A, Franco R, Aiello Talamanca A, Liguori G, Tatangelo F, Delrio P et al (2006) Overexpression of both CXC chemokine receptor 4 and vascular endothelial growth factor proteins predicts early distant relapse in stage II-III colorectal cancer patients. Clin Cancer Res 12:2795–2803. doi:10.1158/1078-0432.CCR-05-2142
Schimanski CC, Schwald S, Simiantonaki N, Jayasinghe C, Gönner U, Wilsberg V et al (2005) Effect of chemokine receptors CXCR4 and CCR7 on the metastatic behavior of human colorectal cancer. Clin Cancer Res 11:1743–1750. doi:10.1158/1078-0432.CCP-04-1195
Proudfoot AE (2006) The biological relevance of chemokine-proteoglycan interactions. Biochem Soc Trans 34(Pt 3):422–426
Robert F (2010) The potential benefits of low-molecular-weight heparins in cancer patients. J Hematol Oncol doi:10.1186/1756-8722-3-3
Ludwig RJ (2009) Therapeutic use of heparin beyond anticoagulation. Curr Drug Discov Technol 6:281–289
Hostettler N, Naggi A, Torri G, Ishai-Michaeli R, Casu B, Vlodavsky I, Borsig L (2007) P-selectin- and heparanase-dependent antimetastatic activity of non-anticoagulant heparins. FASEB J 21:3562–3572. doi:10.1096/fj.07-8450com
Niers TM, Klerk CP, DiNisio M, Van Noorden CJ, Büller HR, Reitsma PH, Richel DJ (2007) Mechanisms of heparin induced anti-cancer activity in experimental cancer models. Crit Rev Oncol Hematol 61:195–207. doi:10.1016/j.critrevonc.2006.07.007
Simka M (2007) Anti-metastatic activity of heparin is probably associated with modulation of SDF-1-CXCR4 axis. Med Hypotheses 69:709. doi:10.1016/j.mehy.2007.01.008
Harvey JR, Mellor P, Eldaly H, Lennard TW, Kirby JA, Ali S (2007) Inhibition of CXCR4-mediated breast cancer metastasis: a potential role for heparinoids? Clin Cancer Res 13:1562–1570. doi:10.1158/1078-0432.CCR-06-1987
Mellor P, Harvey JR, Murphy KJ, Pye D, O'Boyle G, Lennard TW, Kirby JA, Ali S (2007) Modulatory effects of heparin and short-length oligosaccharides of heparin on the metastasis and growth of LMD MDA-MB 231 breast cancer cells in vivo. Br J Cancer 97:761–768. doi:10.1038/sj.bjc.6603928
Zou X, Qiao H, Jiang X, Dong X, Jiang H, Sun X (2008) Downregulation of developmentally regulated endothelial cell locus-1 inhibits the growth of colon cancer. J Biomed Sci doi:10.1007/s11373-008-9284-5
Zhou J, Hu L, Cui Z, Jiang X, Wang G, Krissansen GW, Sun X (2010) Interaction of SDF-1α and CXCR4 plays an important role in pulmonary cellular infiltration in retinoic acid syndrome. Int J Hematol 91:293–302. doi:10.1007/s12185-009-0488-x
Ouyang N, Ke S, Eagleton N, Xie Y, Chen G, Laffins B, Yao H, Zhou B, Tian Y (2010) Pregnane X receptor suppresses proliferation and tumourigenicity of colon cancer cells. Br J Cancer 102:1753–1761
Xu R, Sun X, Tse LY, Li H, Chan PC, Xu S, Xiao W, Kung HF, Krissansen GW, Fan ST (2003) Long-term expression of angiostatin suppresses metastatic liver cancer in mice. Hepatology 37:1451–1460. doi:10.1053/jhep.2003.50244
Sun X, Liu M, Wei Y, Liu F, Zhi X, Xu R, Krissansen GW (2006) Overexpression of von Hippel-Lindau tumor suppressor protein and antisense HIF-1alpha eradicates gliomas. Cancer Gene Ther 13:428–435. doi:10.1038/sj.cgt.7700907
Li W, Gomez E, Zhang Z (2007) Immunohistochemical expression of stromal cell-derived factor-1 (SDF-1) and CXCR4 ligand receptor system in hepatocellular carcinoma. J Exp Clin Cancer Res 26:527–533
Shibuta K, Mori M, Shimoda K, Inoue H, Mitra P, Barnard GF (2002) Regional expression of CXCL12/CXCR4 in liver and hepatocellular carcinoma and cell-cycle variation during in vitro differentiation. Jpn J Cancer Res 93:789–797
Matsusue R, Kubo H, Hisamori S, Okoshi K, Takagi H, Hida K et al (2009) Hepatic stellate cells promote liver metastasis of colon cancer cells by the action of SDF-1/CXCR4 axis. Ann Surg Oncol 16:2645–2653. doi:10.1245/s10434-009-0599-x
Gassmann P, Haier J, Schlüter K, Domikowsky B, Wendel C, Wiesner U et al (2009) CXCR4 regulates the early extravasation of metastatic tumor cells in vivo. Neoplasia 11:651–661. doi:10.1593/neo.09272
Burger JA, Kipps TJ (2006) CXCR4: a key receptor in the crosstalk between tumor cells and their microenvironment. Blood 107:1761–1767. doi:10.1182/blood-2005-08-3182
Orimo A, Gupta PB, Sgroi DC, Arenzana-Seisdedos F, Delaunay T, Naeem R et al (2005) Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121:335–348. doi:10.1016/j.cell.2005.02.034
Terada R, Yamamoto K, Hakoda T, Shimada N, Okano N, Baba N, Ninomiya Y, Gershwin ME, Shiratori Y (2003) Stromal cell-derived factor-1 from biliary epithelial cells recruits CXCR4-positive cells: implications for inflammatory liver diseases. Lab Invest 83:665–672
Raman D, Baugher PJ, Thu YM, Richmond A (2007) Role of chemokines in tumor growth. Cancer Lett 256:137–165. doi:10.1016/j.canlet.2007.05.013
Sadir R, Baleux F, Grosdidier A, Imberty A, Lortat-Jacob H (2001) Characterization of the stromal cell-derived factor-1a-heparin complex. J Biol Chem 276:8288–8296. doi:10.1074/jbc.M008110200
Murphy JW, Cho Y, Sachpatzidis A, Fan C, Hodsdon ME, Lolis E (2007) Structural and functional basis of CXCL12 (stromal cell derived factor-1 alpha) binding to heparin. J Biol Chem 282:10018–10027. doi:10.1074/jbc.M608796200
Veldkamp CT, Peterson FC, Pelzek AJ, Volkman BF (2005) The monomer-dimer equilibrium of stromal-derived factor-1 (CXCL12) is altered by pH, phosphate, sulfate, and heparin. Protein Sci 14:1071–1081. doi:10.1110/ps.041219505
Hasan J, Shnyder SD, Clamp AR, McGown AT, Bicknell R, Presta M et al (2005) Heparin octasaccharides inhibit angiogenesis in vivo. Clin Cancer Res 11:8172–8179. doi:10.1158/1078-0432.CCR-05-0452
Topal B, Aerts JL, Roskams T, Fieuws S, Van Pelt J, Vandekerckhove P, Penninckx F (2005) Cancer cell dissemination during curative surgery for colorectal liver metastases. Eur J Surg Oncol 31:506–511
Acknowledgements
This study was supported in part by grants from the National Natural Scientific Foundation of China (30872987 and 30973474). The experiments were carried out at the Key Laboratory of Hepatosplenic Surgery (Harbin Medical University), Ministry of Education, China. L. Ma and H. Qiao contributed equally to this work.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PPT 914 kb)
Rights and permissions
About this article
Cite this article
Ma, L., Qiao, H., He, C. et al. Modulating the interaction of CXCR4 and CXCL12 by low-molecular-weight heparin inhibits hepatic metastasis of colon cancer. Invest New Drugs 30, 508–517 (2012). https://doi.org/10.1007/s10637-010-9578-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-010-9578-0