Abstract
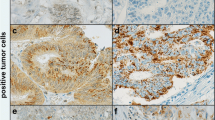
Background: CXCR4, the chemokine receptor for CXCL12, has recently been involved in the metastatic process of several neoplasms. Materials and methods: The expression of CXCR4 was evaluated by immunohistochemistry of colorectal tissue samples and by flow cytometry on Caco2, GEO, SW480, SW48, Lovo and SW620 human colon carcinoma cell lines. Correlations with pathological characteristics of the specimens were analysed with chi-square test. To verify the functional status of CXCR4, cell lines were tested in adhesion, migration, and proliferation assays. Results: We studied the expression of CXCR4 in 88 human colorectal tissues and we found that CXCR4 was expressed in >10% of epithelial cells in 50% of normal mucosae (7/14), in 55% of polyps (29/53), in all of carcinomas (16/16) and hepatic metastasis (5/5). Notably, CXCR4 was significantly over-expressed in cancerous lesions (carcinomas and metastasis) compared to non-cancerous lesions (normal mucosa and polyps) (P=0.003) and in adenomatous polyps versus hyperplastic polyps (P=0.009). The diameter of a polyp was also significantly associated with CXCR4 expression (P=0.031). SW480, SW48 and SW620 cell lines showed the highest levels of CXCR4 (60–80% of positive cells). Adhesion, migration, and proliferation increased in response to the CXCL12 chemokine. These effects were abrogated by the addition of anti-CXCR4 antibodies. Further, CXCL12 activated ERK1/2 in SW480 cells. Conclusions: These data suggest that CXCR4 might play a role in colon cancer cell properties and that anti-CXCR4 antibodies could have therapeutic effects against colorectal cancer.





Similar content being viewed by others
References
Murphy PM (2001) Chemokines and the molecular basis of cancer metastasis. N Engl J Med 345:833–835
Rollins BJ (1997) Chemokines. Blood 3:909–928
Christopherson K, Hromas R (2001) Chemokine regulation of normal and pathological immune responses. Stem Cells 19:388–396
Muller A, Homey B, Soto H et al (2001) Involvement of chemokine receptors in breast cancer metastasis. Nature 410:50–56
Bachelder RE, Wendt MA, Mercurio AM (2002) Vascular endothelial growth factor promotes breast carcinoma invasion in an autocrine manner by regulating the chemokine receptor CXCR4. Cancer Res 62:7203–7206
Kijima T, Maulik G, Ma PC et al (2002) Regulation of cellular proliferation, cytoskeletal function, and signal transduction through CXCR4 and c-Kit in small cell lung ancer cells. Cancer Res 62:6304–6311
Scotton C, Milliken D, Wilson J, Raju S, Balkwill F (2001) Analysis of CC chemokine and chemokine receptor expression in solid ovarian tumors. Br J Cancer 85:891–897
Scotton CJ, Wilson JL, Scott K et al (2002) Multiple actions of the chemokine CXCL12 on epithelial tumor cells in human ovarian cancer. Cancer Res 62:5930–5938
Koshiba T, Hosotani R, Miyamoto Y et al (2000) Expression of stromal cell-derived factor 1 and CXCR4 ligand receptor system in pancreatic cancer: a possible role for tumor progression. Clin Cancer Res 6:3530–3535
Robledo MM, Bartolome RA, Longo N et al (2001) Expression of functional chemokine receptors CXCR3 and CXCR4 on human melanoma cells. J Biol Chem 276:45098–45105
Murakami T, Maki W, Cardones AR et al (2002) Expression of CXC chemokine receptor-4 enhances the pulmonary metastatic potential of murine B16 melanoma cells. Cancer Res 62:7328–7334
Bertolini F, Dell’Agnola C, Mancuso P et al (2002) CXCR4 neutralization, a novel therapeutic approach for non-Hodgkin’s lymphoma. Cancer Res 62:3106–3112
Corcione A, Ottonello L, Tortolina G et al (2000) Stromal cell-derived factor 1 as a chemoattractant for follicular center lymphoma B cells. J Natl Cancer Inst 92:625–628
Arai J, Yasukawa M, Yakushijin Y, Miyazaki T, Fujita S (2000) Stromal cells in lymph nodes attract B lymphoma cells via production of stromal cell-derived factor-1. Eur J Haematol 64:323–332
Geminder H, Sagi-Assif O, Goldberg L et al (2001) A possible role for CXCR4 and its ligand, the CXC chemokine stromal cell-derived factor-1, in the development of bone marrow metastases in neuroblastoma. J Immunol 167:4747–4757
Rempel SA, Dudas S, Ge S, Gutierrez JA (2000) Identification and localization of the cytokine SDF1 and its receptor, CXC chemokine receptor 4, to regions of necrosis and angiogenesis in human glioblastoma. Clin Cancer Res 6:102–111
Barbero S, Bonavia R, Bajetto A et al (2003) Stromal cell-derived factor 1a stimulates human glioblastoma cell growth through the activation of both extracellular signal-regulated kinases 1 and 2 and Akt. Cancer Res 63:1969–1974
Schrader AJ, Lechner O, Templin M et al (2002) CXCR4/CXCL12 expression and signalling in kidney cancer. Br J Cancer 86:1250–1256
Aust G, Steinert M, Kiessling S, Kamprad M, Simchen C (2001) Reduced expression of stromal-derived factor 1 in autonomous thyroid adenomas and its regulation in thyroid derived cells. J Clin Endocrinol Metab 86:3368–3374
Hwang JH, Hwang JH, Chung HK et al (2002) CXC chemokine receptor 4 expression and function in human anaplastic thyroid cancer cells. J Clin Endocrinol Metab 88:408–416
Libura J, Drukala J, Majka M et al (2002) CXCR4-SDF-1 signaling is active in rhabdomyosarcoma cells and regulates locomotion, chemotaxis, and adhesion. Blood 100:2597–2606
Taichman RS, Cooper C, Keller ET et al (2002) Use of the stromal cell-derived factor-1/CXCR4 pathway in prostate cancer metastasis to bone. Cancer Res 62:1832–1837
Staller P, Sulitkova J, Lisztwan J et al (2003) Chemokine receptor CXCR4 downregulated by von Hippel-Lindau tumour suppressor pVHL. Nature 425:307–313
Ries LAG, Eisner MP, Kosary CL (eds) (2002) SEER cancer statistics review, 1973–1999. National Cancer Institute, Bethesda
Hujanen ES, Terranova VP (1985) Migration of tumor cells to organ-derived chemoattractants. Cancer Res 45:3517–3521
Jordan NJ, Kolios G, Abbot SE et al (1999) Expression of functional CXCR4 chemokine receptors on human colonic epithelial cells. J Clin Invest 104:1061–1069
Mitra P, Shibuta K, Mathai J et al (1999) CXCR4 mRNA expression in colon, esophageal and gastric cancers and hepatitis C infected liver. Int J Oncol 14:917–925
Dwinell MB, Eckmann L, Leopard JD, Varki NM, Kagnoff MF (1999) Chemokine receptor expression by human intestinal epithelial cells. Gastroenterology 117:359–367
Kinzler KW, Vogelstein B (1996) Lessons from hereditary colorectal cancer. Cell 87:159–170
Fearon ER, Vogelstein B (1990) A genetic model for colorectal tumorigenesis. Cell 61:759–767
Zeelenberg IS, Ruuls-Van Stalle L, Roos E (2003) The chemokine receptor CXCR4 is required for outgrowth of colon carcinoma micrometastases. Cancer Res 63:3833–3839
Brader S, Eccles SA (2004) Phosphoinositide 3-kinase signalling pathways in tumor progression, invasion and angiogenesis. Tumori 90:2–8
Fresno Vara JA, Casado E, de Castro J, Cejas P, Belda-Iniesta C, Gonzalez-Baron M (2004) PI3K/Akt signalling pathway and cancer. Cancer Treat Rev 30:193–204
Aihua Li, Michelle LV, Rakesh KS (2001) Expression of interleukin 8 and its receptors in human colon carcinoma cells with different metastatic potentials. Clin Cancer Res 7:3298–3304
Kim J, Takeuchi H, Foshag L, Bilchik A, Hoon DSB. Colorectal cancer expression of chemokine receptor CXCR4 promotes liver metastasis. ASCO 2004 annual meeting, Abstract number 3582
De Clercq E (2003) The bicyclam AMD3100 story. Nat Rev Drug Discov 2:581–587
Hendrix CW, Flexner C, MacFarland RT et al (2000) Pharmacokinetics and safety of AMD-3100, a novel antagonist of the CXCR-4 chemokine receptor, in human volunteers. Antimicrob Agents Chemother 44:1667–1673
Acknowledgements
We thank Dr Maria Vittoria Barone (NCI—Naples) for her help in chemotaxis and adhesion experiments. We also thank Antonio Barbato and Fernando Caccavello for their technical assistance in the laboratories of the Department of Surgical Pathology. This work was supported by grants from Novartis (Dr Nora Pigna and Dr Saverio Valerio) and AstraZeneca (Dr Nino Sala).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ottaiano, A., di Palma, A., Napolitano, M. et al. Inhibitory effects of anti-CXCR4 antibodies on human colon cancer cells. Cancer Immunol Immunother 54, 781–791 (2005). https://doi.org/10.1007/s00262-004-0636-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-004-0636-3