Abstract
Background
Obesity and metabolic syndrome (MetS) have been implicated as rising risk factors for the development of colorectal cancers. A rapid increase in the prevalence of obesity and severe obesity among Hispanic patients in the United States may present substantially increased risk for advanced colorectal neoplasia in this population. Currently, there is very little research in this area.
Aims
We sought to identify metabolic risk factors for advanced adenomas (AA) in Hispanic Americans.
Methods
We retrospectively reviewed data from the Los Angeles General (LAG) Medical Center of asymptomatic Hispanic patients above 45 years of age who underwent their first colonoscopies following a positive screening FBT. Patient demographics, metabolic characteristics, as well as colon polyp size and histology were recorded. Polyps were classified as adenomas or AA (including both high-risk adenomas and high-risk serrated polyps). Relative risk for AA was assessed by multivariate logistical regression analyses.
Results
Of the 672 patients in our study, 41.4% were male, 67% had adenomas, and 16% had AA. The mean BMI was 31.2 kg/m2. The mean HDL-C was 49.5 mg/dL (1.28 mmol/L) and the mean triglyceride level was 151 mg/dL. 44.6% had diabetes and 64.1% had hypertension. When comparing patients with AA to patients with no adenoma, male sex, BMI > 34.9 kg/m2, and elevated fasting triglyceride levels were associated with an increased risk of AA. FIB-4 ≥1.45 was also associated with an increased risk of AA in males. There was no significant difference in the risk of AA with diabetes, hypertension, FIB-4 score, LDL-C level, and HDL-C level.
Conclusions
Hispanic patients with a positive FBT were observed to have a high incidence of AA. Class II obesity (BMI ≥ 35 kg/m2), elevated triglyceride levels were identified as risk factors among males in our study. Early interventions to address these modifiable risk factors in at-risk populations, such as multi-disciplinary weight management programs for the treatment of obesity and related co-morbidities, could potentially lead to risk reduction and CRC prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer (CRC) is the third leading cause of cancer-related deaths in U.S. [1]. Over the past two decades, there have been notable declines in overall CRC incidence and mortality largely due to the widespread implementation of screening processes, including colonoscopy, CT colonography, fecal immunohistochemistry tests (FIT) and fecal occult blood tests (FOBT) [2]. However, the largest CRC declines have been in non-Hispanic Whites, whereas there has been a less notable decline in CRC among the Hispanic population [3]. Some research suggests that obesity and metabolic syndrome (MetS) may contribute to early onset of CRC [4,5,6]. Obesity is more prevalent within the Hispanic population (45%) compared to the general U.S. adult population (40%) [2]. Closely linked to obesity, MetS—comprising of insulin resistance, central obesity, dyslipidemia, and elevated blood pressure—is also highly prevalent in the Hispanics [7]. We speculate that obesity and elements of MetS may impact the development of CRC in the Hispanic population. Our study sought to assess the association of obesity and elements of MetS with advanced adenoma (AA)—a precursor to CRC—in Hispanic patients after a positive FBT.
Materials and Methods
Study Design and Population
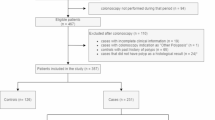
This is a retrospective study of Hispanic patients at least 45 years of age who underwent a diagnostic colonoscopy between December 2016 and December 2020 following a positive FBT at Los Angeles General Medical Center (LAG), formerly known as Los Angeles County + University of Southern California Medical Center. LAG is a large urban safety-net hospital and serves as a major referral center for uninsured and underinsured patients in Los Angeles County. Patients at average-risk for colon cancer are most commonly screened using FBT, either FIT or FOBT, in this underserved population as it is non-invasive and inexpensive, with studies showing higher adherence rates with its use in safety-net populations as compared to screening colonoscopies [8]. If positive, the patient is referred for colonoscopy.
Using the electronic medical record (EMR), patients scheduled for an outpatient colonoscopy with an indication which included the term “fecal immunohistochemistry test,” “FIT,” “fecal occult blood test,” “FOBT,” “FBT,” or “fecal blood test” were screened to ensure they met study criteria. Patients who self-identified as Hispanic in the demographics section on EMR and had no prior colonoscopy were included. Patients with poor quality bowel preparation, history of colorectal cancer, first degree relative with colorectal cancer, or prior colectomy were excluded from the study. Approval from the institutional review board was obtained and all research was performed in accordance with relevant guidelines and regulations.
Demographic and Clinical Data
Demographics and Patient History
Demographic information, including sex and country of origin, were self-reported and obtained from the EMR. If country of origin was not recorded in the EMR, it was labeled as unknown. Medication lists and any obesity-related medical co-morbidities including diabetes, hypertension, and dyslipidemia at the time of colonoscopy were reviewed. Family history and social history, including alcohol and tobacco use, were self-reported and extracted from the EMR.
Laboratory Values and Clinical Information
The date of a positive FBT was documented. All laboratory and other clinical data were collected from the date of colonoscopy or within twelve months of colonoscopy. Clinical data included height and weight, used to calculate body mass index (BMI). Laboratory data of interest included fasting glucose (FG), glycated hemoglobin (HbA1c), low density lipoprotein (LDL-C), high density lipoprotein (HDL-C), triglycerides (TG), total cholesterol, aspartate aminotransaminase (AST), alanine aminotransferase (ALT), albumin, and platelet count. Metabolic syndrome was defined as having any three of the following:
-
Obesity: BMI ≥ 30 kg/m2.
-
Low HDL-C: HDL-C < 40 mg/dL in males, < 50 mg/dL in females.
-
Elevated TG: TG > 150 mg/dL or prescription of an anti-hyperlipidemic medication.
-
Impaired FG: FG > 110 mg/dL, diagnosis of type 2 diabetes, or an anti-diabetic prescription.
-
Hypertension: Elevated systolic blood pressure (SBP > 130 mmHg) or diastolic blood pressure (DBP > 85 mmHg), diagnosis of hypertension, or an anti-hypertensive prescription.
Colonoscopy and Polyp Data
Patients were referred for a colonoscopy by their referring providers after a documented positive FBT. All colonoscopies were performed by gastroenterology fellows with attending supervision. Data obtained from the colonoscopy reports included procedure date, quality of bowel preparation, number of polyps, size and location of largest polyp, endoscopic appearance and histology of largest polyp, and method of removal of largest polyp. Any extracted polyps were placed in a standard formalin solution and reviewed by an attending pathologist. Extracted polyps were classified as adenomas or as AA (including both high risk adenomas and high-risk serrated polyps). High risk features of adenomatous polyps include size > 10 mm, high-grade dysplasia, and villous or tubulovillous histology. Features of high-risk serrated polyps include size > 10 mm, presence of high-grade dysplasia, proximal colon location, or > 3 non-advanced serrated adenomas.
Statistical Analysis
Patient demographics were reported as mean (standard deviation) for continuous variables, frequency and percentage for categorical variables. Variables distribution between no adenoma vs. high risk group were compared using t-test or Chi-square test when appropriate. Multivariable poisson regression with robust variance were used for modeling outcomes (1) Any adenoma vs. normal; (2) High risk adenoma vs. normal. Linearity assumptions for continuous variables were checked by LOESS plot and fractional polynomial test. Continuous variables were tested using splines, categorization by clinical meaningful cutoffs, transformations, and final model was chosen based on smallest model Akaike information criterion (AIC) and Bayesian information criterion (BIC values). With prior research indicating lipids acting differently based on sex, additional analysis were conducted by different sex cohort. Significance level was set as 0.05, 2-sided. All analysis were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA.).
Results
Demographics
Our study included 672 patients of Hispanic origin who underwent colonoscopy after a positive FBT. No adenoma was present in only 224 (33%) of patients with the remaining 67% having at least one. AA was found in 105 patients (16%). The 343 patients (51%) with adenomas which are not AA were not included in the analyses. The summative demographic data of all patients, of patients with AA, and of patients with no adenoma are shown in Table 1.
Amongst the total patient cohort, the mean age was 58.9 years and 41.4% were male. The mean BMI was 31.2 kg/m2. 44.6% of the study population had diabetes with a mean Hb A1c 6.6%, and 64.1% had hypertension. Statins were used by 51.6% of patients. Mean HDL-C, LDL-C, and triglyceride level were 49.5 mg/dL (1.28 mmol/L), 93.1 mg/dL(2.41 mmol/L) and 151 mg/dL (3.91 mmol/L) respectively.
Among patients with AA, the mean age was 59.3 years, 60.0% were male, and the mean BMI was 32.2 kg/m2. 44.8% of patients with AA had diabetes with a mean Hb A1c of 6.5%, and 73.3% had hypertension. Mean HDL-C was 46.4 mg/dL (1.20 mmol/L), mean LDL-C was 85.1 mg/dL (2.20 mmol/L), and mean triglyceride level was 160.6 mg/dL (4.16 mmol/L). In patients with no adenoma, the mean age was 57.7 years, 45.5% were male, and the mean BMI was 31.3 kg/m2. In this group, 57.6% had hypertension, 40.6% had diabetes with a mean Hb A1c 6.5%. Mean HDL-C was 51.5 mg/dL (1.33 mmol/L), mean LDL-C 99.9 mg/dL (2.59 mmol/L), and mean triglyceride was 139.1 mg/dL (3.60 mmol/L). When comparing patients with AA to patients with no adenoma, those with AA were predominantly male, had significantly more hypertension, significantly higher statin use, significantly higher HDL-C levels, and significantly lower LDL-C levels.
Metabolic Determinants and Advanced Adenoma
Using multivariate regression analysis, we compared the 105 patients with AA to the 224 patients with no adenoma to determine the relative risk for BMI, various metabolic determinants, and FIB-4. The analyses of metabolic risk factors for AA are shown in Table 2; Fig. 1.
When comparing patients with AA to patients with no adenoma, male sex (RR 2.146, 95% CI 1.460–3.156, p = 0.0001), BMI > 34.9 kg/m2 (RR 1.788, 95% CI 1.034–3.0942, p = 0.04) and elevated triglyceride levels (per 10-unit increase, RR 1.016, 95% CI 1.00–1.0283, p = 0.02) were associated with an increased risk of AA. There was no significant difference in the risk of AA with diabetes, hypertension, FIB-4 score, LDL-C level, and HDL-C level.
Sex-Based Differences in Metabolic Determinants and Advanced Adenoma
Using multivariate regression analysis, we compared males with AA to males with no adenoma. The analyses of metabolic risk factors for AA in males are shown in Table 3; Fig. 1. Multivariate regression analysis also compared females with AA to females with no adenoma. The analyses for metabolic risk factors for AA in females are shown in Table 4; Fig. 1.
In males, BMI > 34.9 kg/m2 (RR 1.974, 95% CI 1.162–3.352, p = 0.01) almost doubled AA risk. Elevated triglyceride level (per 10-unit increase, RR 1.015, 95% CI 1.0016–1.0291, p = 0.03) and FIB-4 score greater than 1.45 (RR 1.574, 95% CI 1.066–2.324, p = 0.02) also significantly increased the risk for AA in males. Other metabolic determinants including diabetes, hypertension, HDL-C, and LDL-C levels were not associated with increased risk of AA in males. In females, none of the observed metabolic factors, BMI, nor FIB-4 score were associated with increased risk of AA.
Discussion
In this retrospective study of the metabolic risk-factors of AA in Hispanic patients after positive FBT, we found that male sex, class II obesity, and elevated triglyceride level was associated with an increased risk of AA. When stratifying by sex, we found that obesity and elevated triglyceride level posed an increased risk of AA in males. On the other hand, these metabolic risks were not significant in females with AA. Overall, there was a high prevalence of obesity, metabolic syndromes, and advanced hepatic fibrosis in our study population. Our study sought to better understand the association of adenoma and individual metabolic risk factors in Hispanic patients, a cohort that is currently underrepresented in research.
Metabolic Determinants of AA
Several studies have provided evidence that the components of metabolic syndrome—including insulin resistance, obesity, and dyslipidemia—are associated with an increased risk of developing colorectal cancer in Non-Hispanic Whites and Asians [2, 9,10,11,12,13,14]. The focus on separate elements of metabolic syndrome enhances our understanding of how these various components contribute to risk of colorectal adenoma development, both individually and together as a syndrome. Identification of key metabolic risk factors for colorectal adenoma and malignancy provides valuable information for risk stratification, and targets for possible risk reduction.
In line with other studies, we found an increased risk of AA in patients with class II obesity in our study population. Approximately 52% of patients in our study had obesity, with a mean BMI of 31.2 kg/m2. Based on CDC data (2017–2018) [15], Hispanic adults in the United States had a higher prevalence of obesity (44.8%) compared to non-Hispanic whites (42.2%).
The association between obesity and colorectal adenoma and malignancy has been established previously in numerous studies [16]. In some, waist circumference, visceral obesity, and waist to hip ratio were also found to be independent risk factors for colorectal adenoma [10, 17, 18]. Another study found a significant positive association between BMI and colorectal cancer risk with any 1 kg/m2 increase in BMI conferring additional colorectal cancer risk (HR 1.03 for males, HR 1.02 for females) [19]. Furthermore, in both males and females, BMI is significantly associated with higher rates of mortality in colon cancer [12]. Our study adds to this data, confirming these findings in Hispanic patients.
The mechanism linking obesity and metabolic syndromes with CRC remains unclear. Insulin resistance, a key feature to the pathophysiology of MetS, has been implicated in colonic carcinogenesis by promoting cell proliferation, and inhibiting apoptosis through hyperinsulinemia and the release of insulin like growth factors [20]. A significant association has been found between the prevalence of colorectal adenomas and increasing levels of glucose, levels of hemoglobin A1c, and C-peptide [21]. Diabetes was not associated with an increased risk of AA in our study, although this may be due to the lower mean Hgb A1c levels noted in our patient population (mean Hgb A1c 6.6 ± 1.5). In one study, it has been shown that poor glycemic control in patients with type 2 diabetes mellitus, defined as Hgb A1c ≥ 7.5%, independently predicts a higher tendency to develop adenomatous polyps and an increased risk for colorectal cancer compared to well-controlled diabetes [22]. While our study found higher incidences of insulin resistance in our Hispanic patients, we did not find an association with increased incidence of AA.
In our study, the presence of elevated triglyceride levels were associated with an increased risk of AA in males, while the presence of hypertension, HDL-C, and LDL-C had no association with AA. Similarly, mixed results have been noted in other studies. One large study revealed that the prevalence of colorectal adenoma was higher in individuals with higher triglyceride, total cholesterol, and blood pressure levels [13]. However, in another study, there were mixed results, showing a statistically significant increase in both non-neoplastic polyps and AA in patients with hypertension [23]. Another systematic review and a meta-analysis of colonoscopy studies in Asia between 2000 and 2014 showed that people with adenoma were more likely to have unfavorable cholesterol profiles at the time of colonoscopy than those without adenoma [24].
Sex Based Differences in AA Risk Factors
Our study identified more AA in males (23%) compared to females (11%) after a positive FBT. Males are also known to have a higher incidence and mortality of CRC than women [25]. In addition, the sensitivity and positive predictive value of fecal occult blood tests are higher among men than among women [26]. Furthermore, a large cross-sectional study reported that a larger proportion of women exhibited flat- and depressed-type colorectal neoplasia, often seen in right-sided colonic malignancies, whereas men had a higher percentage of polypoid neoplasia, which are more easily detectable [27].
Prevalence of AA and Obesity in Hispanic Patients
In our study, 67% and 16% of patients with positive FBT were found to have any colorectal adenomas and AA, respectively, on colonoscopy. In a recent systematic review of adenoma detection by colonoscopy in individuals with positive FIT, the pooled adenoma detection rate was 47.8% (44.1–51.6) and the pooled AA detection rate was 25.3% (22–29) [28]. While some literature suggests that there are no significant differences between non-Hispanic whites and Hispanics in the United States [29], our study demonstrates higher than expected incidences of adenoma, but lower than expected AA in our Hispanic cohort. In another study that examined a colorectal neoplasia in FIT positive patients in a predominantly Hispanic community, AA detection rate was 11.1% [30]. Similarly, prior studies report AA prevalence in U.S. Hispanics ranging from 3.5 to 12.7% [31,32,33]. While lower than expected, our study’s AA rates are consistent with other studies that examine the Hispanic population.
We calculated FIB-4 scores to assess the proportion of patients with underlying fibrosis, which may be attributable to NAFLD given the known association with obesity, metabolic syndrome, and Hispanic ethnicity [34]. Our study has shown an association of AA with FIB-4 ³1.45 (³F2 fibrosis) in male Hispanic patients. Several studies have also demonstrated that NAFLD is a risk factor for colorectal adenoma, but this has been more studied in Asian populations [35,36,37]. Our study identified an association between hepatic fibrosis and colorectal neoplasia in the Hispanic population, highlighting the importance of emphasizing CRC screening in these patients.
Strengths and Limitations
Our study has some limitations as a single-center retrospective study. The information was obtained via chart review of each patient history, reflective of what was updated in the patient’s EMR. The data of waist circumferential in our study population was not recorded in EMR. This may underestimate the proportion of patients with metabolic syndrome. Additionally, the fecal blood test (FBT) used as an initial screening for colon cancer in our study population, often overestimates the prevalence of colonic neoplasia.
Several strengths to our study include the fact that our study population represents an underserved Hispanic population in Los Angeles’s largest safety-net hospital. Our study also adds to existing knowledge by focusing on the increasing Hispanic population in the United States and their metabolic risk factors in the development of colon neoplasia. There is limited research in this area with a focus on Hispanic patients. In addition, the use of FBT as the screening method of choice in our study is important, as FBTs are widely used as the initial screening method of choice at safety-net medical centers in an effort to improve screening rates given its cost-effectiveness, reductions in colonoscopy-related complications, and ease of completion [38].
Conclusion and Implications
Hispanic patients with a positive FBT were observed to have a higher-than-expected incidence of adenomas, but a lower-than-expected incidence of AA, with class II obesity (BMI ≥ 35 kg/m2) and elevated triglyceride identified as risk factors for AA in Hispanic males. Our study highlights the importance of identifying and characterizing risk factors for colorectal neoplasia in the rapidly increasing Hispanic. Early interventions to address these modifiable risk factors in vulnerable populations, such as multi-disciplinary weight management programs for the treatment of obesity and related co-morbidities, could potentially lead to risk reduction and disease prevention.
References
Miller KD, Ortiz AP, Pinheiro PS, Bandi P, Minihan A, Fuchs HE, Martinez Tyson D, Tortolero-Luna G, Fedewa SA, Jemal AM, Siegel RL. Cancer statistics for the US Hispanic/Latino population, 2021. CA Cancer J Clin. 2021;71(6):466–487. doi: 10.3322/caac.21695. Epub 2021 Sep 21. PMID: 34545941.
Centers for Disease Control and Prevention (CDC). Vital signs: colorectal cancer screening test use–United States, 2012. MMWR Morb Mortal Wkly Rep. 2013;62(44):881–8. PMID: 24196665; PMCID: PMC4585592.
Garcia S, Pruitt SL, Singal AG, Murphy CC. Colorectal cancer incidence among Hispanics and non-Hispanic Whites in the United States. Cancer Causes Control. 2018;29(11):1039–1046. doi: 10.1007/s10552-018-1077-1. Epub 2018 Aug 28. PMID: 30155605; PMCID: PMC6628724.
Burke CA. Colonic complications of obesity. Gastroenterol Clin North Am. 2010;39(1):47–55. doi: https://doi.org/10.1016/j.gtc.2009.12.005. PMID: 20202578.
Chan AT, Giovannucci EL. Primary prevention of colorectal cancer. Gastroenterology. 2010;138(6):2029–2043.e10. doi: https://doi.org/10.1053/j.gastro.2010.01.057. PMID: 20420944; PMCID: PMC2947820.
Bardou M, Barkun AN, Martel M. Obesity and colorectal cancer. Gut. 2013;62(6):933 – 47. doi: 10.1136/gutjnl-2013-304701. Epub 2013 Mar 12. PMID: 23481261.
Heiss G, Snyder ML, Teng Y, Schneiderman N, Llabre MM, Cowie C, Carnethon M, Kaplan R, Giachello A, Gallo L, Loehr L, Avilés-Santa L. Prevalence of metabolic syndrome among Hispanics/Latinos of diverse background: the Hispanic Community Health Study/Study of Latinos. Diabetes Care. 2014;37(8):2391–9. doi: https://doi.org/10.2337/dc13-2505. PMID: 25061141; PMCID: PMC4113166.
Inadomi JM, Vijan S, Janz NK, Fagerlin A, Thomas JP, Lin YV, Muñoz R, Lau C, Somsouk M, El-Nachef N, Hayward RA. Adherence to colorectal cancer screening: a randomized clinical trial of competing strategies. Arch Intern Med. 2012;172(7):575–82. doi: https://doi.org/10.1001/archinternmed.2012.332. PMID: 22493463; PMCID: PMC3360917.
O’Sullivan DE, Sutherland RL, Town S, Chow K, Fan J, Forbes N, Heitman SJ, Hilsden RJ, Brenner DR. Risk Factors for Early-Onset Colorectal Cancer: A Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol. 2022;20(6):1229–1240.e5. doi: 10.1016/j.cgh.2021.01.037. Epub 2021 Jan 29. PMID: 33524598.
Kim JH, Lim YJ, Kim YH, Sung IK, Shim SG, Oh SO, Park SS, Yang S, Son HJ, Rhee PL, Kim JJ, Rhee JC, Choi YH. Is metabolic syndrome a risk factor for colorectal adenoma? Cancer Epidemiol Biomarkers Prev. 2007;16(8):1543-6. doi: https://doi.org/10.1158/1055-9965.EPI-07-0199. PMID: 17684126.
Ortiz AP, Thompson CL, Chak A, Berger NA, Li L. Insulin resistance, central obesity, and risk of colorectal adenomas. Cancer. 2012;118(7):1774–81. doi: https://doi.org/10.1002/cncr.26454. Epub 2011 Aug 25. PMID: 22009143; PMCID: PMC3262947.
Calle EE, Rodriguez C, Walker-Thurmond K, Thun MJ. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med. 2003;348(17):1625-38. doi: https://doi.org/10.1056/NEJMoa021423. PMID: 12711737.
Pyo JH, Kim ES, Chun HJ, Keum B, Jeen YT, Lee HS, Kim CD, Ryu HS, Kim YH, Lee JE. Fasting blood sugar and serum triglyceride as the risk factors of colorectal adenoma in korean population receiving screening colonoscopy. Clin Nutr Res. 2013;2(1):34–41. doi: 10.7762/cnr.2013.2.1.34. Epub 2013 Jan 29. PMID: 23429421; PMCID: PMC3572815.
Chen H, Zheng X, Zong X, Li Z, Li N, Hur J, Fritz CD, Chapman W Jr, Nickel KB, Tipping A, Colditz GA, Giovannucci EL, Olsen MA, Fields RC, Cao Y. Metabolic syndrome, metabolic comorbid conditions and risk of early-onset colorectal cancer. Gut. 2021;70(6):1147–1154. doi: https://doi.org/10.1136/gutjnl-2020-321661. Epub 2020 Oct 9. PMID: 33037055; PMCID: PMC8032822.
Hales CM, Carroll MD, Fryar CD, Ogden CL. Prevalence of Obesity and Severe Obesity Among Adults: United States, 2017–2018. NCHS Data Brief. 2020;(360):1–8. PMID: 32487284.
Li H, Boakye D, Chen X, Hoffmeister M, Brenner H. Association of Body Mass Index With Risk of Early-Onset Colorectal Cancer: Systematic Review and Meta-Analysis. Am J Gastroenterol. 2021;116(11):2173–2183. doi: https://doi.org/10.14309/ajg.0000000000001393. PMID: 34309586; PMCID: PMC8560162.
Kang HW, Kim D, Kim HJ, Kim CH, Kim YS, Park MJ, Kim JS, Cho SH, Sung MW, Jung HC, Lee HS, Song IS. Visceral obesity and insulin resistance as risk factors for colorectal adenoma: a cross-sectional, case-control study. Am J Gastroenterol. 2010;105(1):178–87. doi: https://doi.org/10.1038/ajg.2009.541. Epub 2009 Sep 15. PMID: 19755965.
Ortiz AP, Thompson CL, Chak A, Berger NA, Li L. Insulin resistance, central obesity, and risk of colorectal adenomas. Cancer. 2012;118(7):1774–81. doi: https://doi.org/10.1002/cncr.26454. Epub 2011 Aug 25. PMID: 22009143; PMCID: PMC3262947.
Matsuo K, Mizoue T, Tanaka K, Tsuji I, Sugawara Y, Sasazuki S, Nagata C, Tamakoshi A, Wakai K, Inoue M, Tsugane S; Development and Evaluation of Cancer Prevention Strategies in Japan. Association between body mass index and the colorectal cancer risk in Japan: pooled analysis of population-based cohort studies in Japan. Ann Oncol. 2012;23(2):479–90. doi: https://doi.org/10.1093/annonc/mdr143. Epub 2011 May 19. PMID: 21597097.
Jee SH, Kim HJ, Lee J. Obesity, insulin resistance and cancer risk. Yonsei Med J. 2005;46(4):449–55. doi: https://doi.org/10.3349/ymj.2005.46.4.449. PMID: 16127767; PMCID: PMC2815827.
Rampal S, Yang MH, Sung J, Son HJ, Choi YH, Lee JH, Kim YH, Chang DK, Rhee PL, Rhee JC, Guallar E, Cho J. Association between markers of glucose metabolism and risk of colorectal adenoma. Gastroenterology. 2014;147(1):78–87.e3. doi: https://doi.org/10.1053/j.gastro.2014.03.006. Epub 2014 Mar 14. PMID: 24632359.
Huang X, Fan Y, Zhang H, Wu J, Zhang X, Luo H. Association between serum HbA1c levels and adenomatous polyps in patients with the type 2 diabetes mellitus. Minerva Endocrinol. 2015;40(3):163–7. Epub 2014 Jul 8. PMID: 25003225.
Huang HE, Yang YC, Wu JS, Wang RH, Lu FH, Chang CJ. The relationship between different glycemic statuses and colon polyps in a Taiwanese population. J Gastroenterol. 2014;49(7):1145–51. doi: https://doi.org/10.1007/s00535-013-0863-5. Epub 2014 Jan 16. PMID: 24429895.
Passarelli MN, Newcomb PA. Blood Lipid Concentrations and Colorectal Adenomas: A Systematic Review and Meta-Analysis of Colonoscopy Studies in Asia, 2000–2014. Am J Epidemiol. 2016;183(8):691–700. doi: https://doi.org/10.1093/aje/kwv294. Epub 2016 Mar 24. PMID: 27013025; PMCID: PMC4832054.
Brenner H, Hoffmeister M, Arndt V, Haug U. Gender differences in colorectal cancer: implications for age at initiation of screening. Br J Cancer. 2007;96(5):828–31. doi: https://doi.org/10.1038/sj.bjc.6603628. Epub 2007 Feb 20. PMID: 17311019; PMCID: PMC2360074.
Brenner H, Haug U, Hundt S. Sex differences in performance of fecal occult blood testing. Am J Gastroenterol. 2010;105(11):2457–64. doi: https://doi.org/10.1038/ajg.2010.301. Epub 2010 Aug 10. PMID: 20700114.
Kim SE, Paik HY, Yoon H, Lee JE, Kim N, Sung MK. Sex- and gender-specific disparities in colorectal cancer risk. World J Gastroenterol. 2015;21(17):5167–75. doi: https://doi.org/10.3748/wjg.v21.i17.5167. PMID: 25954090; PMCID: PMC4419057.
Mohan BP, Khan SR, Daugherty E, Chandan S, Ponnada S, Facciorusso A, Kassab LL, Asokkumar R, Adler DG. Pooled Rates of Adenoma Detection by Colonoscopy in Asymptomatic Average Risk Individuals with Positive Fecal Immunochemical Test: A Systematic Review and Meta-Analysis. Gastrointest Endosc. 2022 Apr 9:S0016-5107(22)00258-9. doi: 10.1016/j.gie.2022.04.004. Epub ahead of print. PMID: 35413330.
Corley DA, Jensen CD, Marks AR, Zhao WK, de Boer J, Levin TR, Doubeni C, Fireman BH, Quesenberry CP. Variation of adenoma prevalence by age, sex, race, and colon location in a large population: implications for screening and quality programs. Clin Gastroenterol Hepatol. 2013;11(2):172–80. doi: 10.1016/j.cgh.2012.09.010. Epub 2012 Sep 14. PMID: 22985608; PMCID: PMC3954741.
Willauer AN, Zuckerman MJ, Alomari A, Alvarado LA, Salaiz R, Casner N, Dwivedi AK, Shokar NK. Colorectal neoplasia prevalence in a predominantly Hispanic community: Results from a colorectal cancer screening program in Texas. Am J Med Sci. 2022 Apr 6:S0002-9629(22)00140-9. doi: 10.1016/j.amjms.2022.03.013. Epub ahead of print. PMID: 35398033.
Lee KK, Jandorf L, Thélèmaque L, Itzkowitz SH. Colorectal neoplasia detection among black and Latino individuals undergoing screening colonoscopy: a prospective cohort study. Gastrointest Endosc. 2014;79(3):466–72. doi: https://doi.org/10.1016/j.gie.2013.12.020. PMID: 24528828; PMCID: PMC4403728.
Collazo TH, Jandorf L, Thelemaque L, Lee K, Itzkowitz SH. Screening Colonoscopy among Uninsured and Underinsured Urban Minorities. Gut Liver. 2015;9(4):502–8. doi: https://doi.org/10.5009/gnl14039. PMID: 25287165; PMCID: PMC4477994.
Lebwohl B, Capiak K, Neugut AI, Kastrinos F. Risk of colorectal adenomas and advanced neoplasia in Hispanic, black and white patients undergoing screening colonoscopy. Aliment Pharmacol Ther. 2012;35(12):1467–73. doi: https://doi.org/10.1111/j.1365-2036.2012.05119.x. Epub 2012 Apr 29. PMID: 22540887; PMCID: PMC3396558.
Akram G, Corcoran ED, MacRobbie A, Harrington G, Bennie M. Developing a Model for Pharmaceutical Palliative Care in Rural Areas-Experience from Scotland. Pharmacy (Basel). 2017;5(1):6. doi: https://doi.org/10.3390/pharmacy5010006. PMID: 28970418; PMCID: PMC5419387.
Shen H, Lipka S, Kumar A, Mustacchia P. Association between nonalcoholic fatty liver disease and colorectal adenoma: a systemic review and meta-analysis. J Gastrointest Oncol. 2014;5(6):440–6. doi: https://doi.org/10.3978/j.issn.2078-6891.2014.061. PMID: 25436123; PMCID: PMC4226833.
Ze EY, Kim BJ, Jun DH, Kim JG, Kang H, Lee DY. The Fatty Liver Index: A Simple and Accurate Predictor of Colorectal Adenoma in an Average-Risk Population. Dis Colon Rectum. 2018;61(1):36–42. doi: 10.1097/DCR.0000000000000973. PMID: 29219920; PMCID: PMC5728585.
Kim MC, Park JG, Jang BI, Lee HJ, Lee WK. Liver fibrosis is associated with risk for colorectal adenoma in patients with nonalcoholic fatty liver disease. Medicine (Baltimore). 2019;98(6):e14139. doi: https://doi.org/10.1097/MD.0000000000014139. PMID: 30732129; PMCID: PMC6380844.
Quintero E, Castells A, Bujanda L, Cubiella J, Salas D, Lanas Á, Andreu M, Carballo F, Morillas JD, Hernández C, Jover R, Montalvo I, Arenas J, Laredo E, Hernández V, Iglesias F, Cid E, Zubizarreta R, Sala T, Ponce M, Andrés M, Teruel G, Peris A, Roncales MP, Polo-Tomás M, Bessa X, Ferrer-Armengou O, Grau J, Serradesanferm A, Ono A, Cruzado J, Pérez-Riquelme F, Alonso-Abreu I, de la Vega-Prieto M, Reyes-Melian JM, Cacho G, Díaz-Tasende J, Herreros-de-Tejada A, Poves C, Santander C, González-Navarro A; COLONPREV Study Investigators. Colonoscopy versus fecal immunochemical testing in colorectal-cancer screening. N Engl J Med. 2012;366(8):697–706. doi: https://doi.org/10.1056/NEJMoa1108895. Erratum in: N Engl J Med. 2016;374(19):1898. PMID: 22356323.
Funding
None.
Open access funding provided by SCELC, Statewide California Electronic Library Consortium
Author information
Authors and Affiliations
Contributions
LY, OLS, and RT were involved in conception and design. All authors contributed to collection, analysis, and interpretation of data, final approval of the article, and critical revision of the article for important intellectual content. RT, LDa, PL, and OLS were involved in drafting of the article.
Corresponding author
Ethics declarations
Conflict of Interest
LY: One Legacy, Intercept, Gilead, Zydus, Madrigal.
LDi: NIH Grants UL1TR001855 and UL1TR000130.
RT, LDa, PL, OLS: No conflict of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Tamai, R., Dawit, L., Lee, P. et al. The Association of Metabolic Risk Factors with Advanced Adenomas in Hispanic Patients. Dig Dis Sci 69, 1403–1410 (2024). https://doi.org/10.1007/s10620-024-08323-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-024-08323-x