Abstract
Introduction
Limited data exists on the effectiveness of organized outreach campaigns on CRC screening completion for patients who are newly eligible for such screening.
Methods
We conducted an analysis of an existing clinical trial dataset of a publicly funded safety-net health system serving low-income populations.
Results
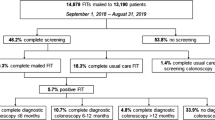
A total of 619 patients aged 50–51 received the outreach intervention and 3108 patients aged greater than 51 years old who had no prior history of FIT testing similarly received the outreach intervention. Patients newly eligible for FIT were more likely to complete a FIT test compared with older patients who had yet to complete a FIT test (58.3% vs 40.5%, p < 0.001).
Conclusion
Patients who are newly eligible for colorectal cancer screening are more likely to respond to outreach interventions than older patients without a prior history of FIT, indicating newly eligible patients across diverse populations may benefit from targeted outreach intervention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Organized outreach using mailed fecal immunochemical testing (FIT) has been demonstrated to be an effective outreach strategy to improve colorectal cancer (CRC) screening; however, the effectiveness is significantly higher in patients with a history of FIT completion compared to those with no prior FIT completion [1,2,3]. As such, in resource-limited settings, population health initiatives at times have targeted outreach to patients with a track record of FIT completion while deferring patients who have yet to complete their first FIT to clinics and their primary care providers [4, 5].
Among the group who have not completed FIT are those who are newly eligible for screening. Limited data exists on the effectiveness of organized outreach campaigns on CRC screening completion for patients who are newly eligible for such screening. We conducted an analysis of an existing clinical trial dataset to examine the effectiveness of organized outreach using mailed fecal immunochemical tests (FITs) to patients newly eligible for CRC screening.
Methods
The clinical intervention was conducted in the San Francisco Health Network (SFHN), a publicly funded safety-net health system serving low-income populations. In the trial (NCT02613260), patients from eight participating primary care clinics aged 50–75 years not up-to-date with CRC screening were randomly assigned to receive multicomponent screening interventions or usual care. The intervention included a mailed postcard and call, followed by a mailed FIT kit, and a reminder phone call if the FIT kit was not returned. Usual care was performed at the provider’s discretion and may have included tailored educational materials and coaching. Participants were enrolled between January 2016 and August 2017. The clinical outcome was screening participation at one year. The primary outcome compared patients newly eligible for screening (50–51 years old) to older patients (≥ 52 years old) without prior history of FIT completion after having received multicomponent outreach intervention. For our secondary outcomes, we analyzed screening participation for newly eligible patients who received outreach intervention compared to those receiving usual care. We also compared the FIT screening rate for older patients (> 52 years old) with a prior history of FIT to patients newly eligible for FIT screening. This trial was approved by the University of California San Francisco Institutional Review Board (IRB, 14–14,861).
Results
There were 619 patients aged 50–51 who received the outreach intervention and 3108 patients aged greater than 51 years old who had no prior history of FIT testing who similarly received the outreach intervention. Patients newly eligible for FIT were more likely to complete a FIT test compared with older patients who had yet to complete a FIT test (58.3% vs 40.5%, p < 0.001). The relative risk ratio between age cohorts (1.43 [1.31, 1.57]) is constant across all demographic subgroups including race (Caucasian:1.43 [1.18, 1.72], Black:1.58 [1.28, 1.94], Other:1.29 [1.088, 1.517]), primary language (English: 1.40 [1.24, 1.57], Spanish: 1.40 [1.18, 1.66]), gender (Male: 1.44 [1.27, 1.63], and insurance type (Medi-Cal: 1.32 [1.16, 1.50]). Older patients with a prior history of FIT were more likely to complete FIT screening compared to newly eligible patients aged 50–51 years (74.6% vs 58.3%, p < 0.001). In addition, newly eligible FIT-naive patients in the intervention were more likely to complete FIT screening compared to newly eligible patients in the usual care group (58.3% vs. 42.3%, p < 0.001).
Discussion
Organized outreach to individuals newly eligible for FIT screening, those aged 50–51, had higher rates of FIT completion compared to outreach to FIT-naive individuals ≥ 52 years of age. This result was consistent throughout all demographic subtypes analyzed, including across gender, race/ethnicity, primary language, and insurance type, indicating newly eligible patients across diverse populations may benefit from broad outreach intervention. Overall, our results demonstrate that patients who are newly eligible for colorectal cancer screening are more likely to respond to outreach interventions than older patients without a prior history of FIT. Therefore, health systems should consider mailed FIT invitations to individuals newly eligible for CRC screening, given that newly eligible patients represent a subset of patients that may benefit dramatically from targeted screening efforts. Additionally, concerted outreach to newly eligible patients may extend the durability and cost-effective strategy of these outreach efforts [3]. In a previous analysis of patients with no prior FIT testing, the FIT completion rate was ~ 15% in the ensuing year in the absence of any intervention [6]. The substantial FIT completion rate of (~ 40%) in newly eligible patients without the outreach intervention highlights the relative potential of newly eligible patients to engage in FIT screening.
While these patients were studied prior to the recent pandemic, the adoption of telehealth and asynchronous population health services since the emergence of COVID-19 have facilitated outreach interventions [7]. The increased utilization of these remote health services, including the incorporation of text reminders and online communication portals, will provide an avenue for economical and accessible colorectal cancer screening interventions targeted toward specific populations [8, 9].
There are several limitations. We are not aware of how often newly eligible patients seek out information from their primary care provider as a result of multicomponent outreach. These newly eligible patients may be responsive to other prompts, although the provision of FIT kits by mail by virtue of reducing barriers will still result in higher terminal screening rates. For older patients who have no history of FIT completion, we do not have data on whether they had been previously offered FIT or the number of FIT offerings. It is likely that they had been offered to screen and the lack of test completion suggests that high-touch or individualized education is needed. In conclusion, our results suggest that patients newly eligible for FIT screening are more likely to complete FIT testing after outreach and should be considered a subgroup particularly responsive to CRC screening interventions. Tailored interventions could then be dedicated to individuals who have not had a history of FIT test completion.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to the presence of Protected Health Information (PHI) but are available from the corresponding author on reasonable request.
References
Levin TR, Corley DA, Jensen D et al. Gastroenterology. 2018;155:1383–1391.e5.
Issaka RB, Avila P, Whitaker E, Bent S, Somsouk M. Prev Med. 2019;118:113–121.
Somsouk M, Mannalithara A, Garcia D et al. J Natl Cancer Inst. 2019;112:305–313.
Chiu HM, Yen AM, Chiu SY et al. Cancer. 2015;121:3221–3229.
Bretthauer M, Løberg M, Wieszczy P et al. N Engl J Med. 2022;387(17):1547–1556.
Burns S, Somsouk M. Dig Dis Sci. 2022;10:1–3.
Patel SY, Mehrotra A, Huskamp HA, Uscher-Pines L, Ganguli I, Barnett ML. JAMA Intern Med. 2021;181:388–391.
Balzora S, Issaka RB, Anyane-Yeboa A, Gray II DM, May FP. Gastrointest Endosc. 2020;92:946–950.
Gupta S, Coronado GD, Argenbright K et al. CA: Cancer J Clin. 2020;70:283–298.
Acknowledgments
Not Applicable
Author information
Authors and Affiliations
Contributions
SB: drafting of the manuscript, analysis, and interpretation of data; statistical analysis, JW: statistical analysis; revision of manuscript, MS: study concept and design; acquisition of data; analysis and interpretation of data; revision of the manuscript; study supervision.
Corresponding author
Ethics declarations
Conflict of interests
The authors have not disclosed any conflict of interests.
Ethical approval and consent to participate
Research involving human participants, human material, or human data, must have been performed in accordance with the Declaration of Helsinki and was approved by the University of California San Francisco Institutional Review Board (IRB, 14–14861). Informed consent to participate in the study was obtained from participants (or their parent or legal guardian in the case of children under 16).
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Burns, S., Wang, J. & Somsouk, M. Effect of Mailed Fecal Immunochemical Test Outreach for Patients Newly Eligible for Colorectal Cancer Screening. Dig Dis Sci 68, 2315–2317 (2023). https://doi.org/10.1007/s10620-023-07925-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-023-07925-1



