Abstract
Background
Food-specific immunoglobulin G4 (FS-IgG4) is associated with eosinophilic esophagitis (EoE); however, it is not clear whether production is limited to the esophagus.
Aims
To assess FS-IgG4 levels in the upper gastrointestinal tract and plasma and compare these with endoscopic disease severity, tissue eosinophil counts, and patient-reported symptoms.
Methods
We examined prospectively banked plasma, throat swabs, and upper gastrointestinal biopsies (esophagus, gastric antrum, and duodenum) from control (n = 15), active EoE (n = 24), and inactive EoE (n = 8) subjects undergoing upper endoscopy. Patient-reported symptoms were assessed using the EoE symptom activity index (EEsAI). Endoscopic findings were evaluated using the EoE endoscopic reference score (EREFS). Peak eosinophils per high-power field (eos/hpf) were assessed from esophageal biopsies. Biopsy homogenates and throat swabs were normalized for protein content and assessed for FS-IgG4 to milk, wheat, and egg.
Results
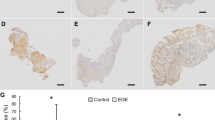
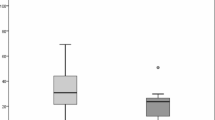
Median FS-IgG4 for milk and wheat was significantly increased in the plasma, throat swabs, esophagus, stomach, and duodenum of active EoE subjects compared to controls. No significant differences for milk- or wheat-IgG4 were observed between active and inactive EoE subjects. Among the gastrointestinal sites sampled, FS-IgG4 levels were highest in the esophagus. Esophageal FS-IgG4 for all foods correlated significantly across all sites sampled (r ≥ 0.59, p < 0.05). Among subjects with EoE, esophageal FS-IgG4 correlated significantly with peak eos/hpf (milk and wheat) and total EREFS (milk). EEsAI scores and esophageal FS-IgG4 levels did not correlate.
Conclusions
Milk and wheat FS-IgG4 levels are elevated in plasma and throughout the upper gastrointestinal tract in EoE subjects and correlate with endoscopic findings and esophageal eosinophilia.




Similar content being viewed by others
Abbreviations
- AMS:
-
Avoidance, modification, and slow eating
- BCA:
-
Bicinchoninic acid assay
- CI:
-
Confidence interval
- CTAB:
-
Cetrimonium bromide
- EEsAI:
-
Eosinophilic esophagitis symptom activity index
- EGID:
-
Eosinophilic gastrointestinal disease
- EoD:
-
Eosinophilic duodenitis
- EoE:
-
Eosinophilic esophagitis
- EoG:
-
Eosinophilic gastritis
- eos/hpf:
-
Eosinophils per high-power field
- EREFS:
-
Endoscopic reference score
- FS-IgG4:
-
Food-specific immunoglobulin G4
- GERD:
-
Gastroesophageal reflux disease
- GI:
-
Gastrointestinal
- H&E:
-
Hematoxylin and eosin
- IBS:
-
Irritable bowel syndrome
- PPI:
-
Proton pump inhibitor
- Q1:
-
Quartile 1
- Q3:
-
Quartile 3
- VDQ:
-
Visual dysphagia question
- yrs:
-
Years
References
Muir A, Falk GW. Eosinophilic Esophagitis: A Review. JAMA. 2021;326(13):1310–1318.
O’Shea KM, Aceves SS, Dellon ES et al. Pathophysiology of Eosinophilic Esophagitis. Gastroenterology. 2018;154(2):333–345.
Arias A, Gonzalez-Cervera J, Tenias JM, Lucendo AJ. Efficacy of dietary interventions for inducing histologic remission in patients with eosinophilic esophagitis: a systematic review and meta-analysis. Gastroenterology. 2014;146(7):1639–1648.
Clayton F, Fang JC, Gleich GJ et al. Eosinophilic esophagitis in adults is associated with IgG4 and not mediated by IgE. Gastroenterology. 2014;147(3):602–609.
Loizou D, Enav B, Komlodi-Pasztor E et al. A pilot study of omalizumab in eosinophilic esophagitis. PLoS One. 2015;10(3):e0113483.
Simon D, Cianferoni A, Spergel JM et al. Eosinophilic esophagitis is characterized by a non-IgE-mediated food hypersensitivity. Allergy. 2016;71(5):611–620.
Wright BL, Kulis M, Guo R et al. Food-specific IgG(4) is associated with eosinophilic esophagitis. J Allergy Clin Immunol. 2016;138(4):1190–1192.
Dellon ES, Guo R, McGee SJ et al. A Novel Allergen-Specific Immune Signature-Directed Approach to Dietary Elimination in Eosinophilic Esophagitis. Clin Transl Gastroenterol. 2019;10(12):e00099.
McGowan EC, Medernach J, Keshavarz B, et al. Food antigen consumption and disease activity affect food-specific IgG4 levels in patients with eosinophilic esophagitis (EoE). Clin Exp Allergy. 2022.
Schuyler AJ, Wilson JM, Tripathi A et al. Specific IgG(4) antibodies to cow’s milk proteins in pediatric patients with eosinophilic esophagitis. J Allergy Clin Immunol. 2018;142(1):139–148.
Dilollo J, Rodriguez-Lopez EM, Wilkey L, Martin EK, Spergel JM, Hill DA. Peripheral markers of allergen-specific immune activation predict clinical allergy in eosinophilic esophagitis. Allergy. 2021;76(11):3470–3478.
Tordesillas L, Berin MC. Mechanisms of Oral Tolerance. Clin Rev Allergy Immunol. 2018;55(2):107–117.
Pyne AL, Hazel MW, Uchida AM et al. Oesophageal secretions reveal local food-specific antibody responses in eosinophilic oesophagitis. Aliment Pharmacol Ther. 2022;56(9):1328–1336.
Rosenberg CE, Mingler MK, Caldwell JM et al. Esophageal IgG4 levels correlate with histopathologic and transcriptomic features in eosinophilic esophagitis. Allergy. 2018;73(9):1892–1901.
Pope AE, Stanzione N, Naini BV et al. Esophageal IgG4: Clinical, Endoscopic, and Histologic Correlations in Eosinophilic Esophagitis. J Pediatr Gastroenterol Nutr. 2019;68(5):689–694.
Zukerberg L, Mahadevan K, Selig M, Deshpande V. Oesophageal intrasquamous IgG4 deposits: an adjunctive marker to distinguish eosinophilic oesophagitis from reflux oesophagitis. Histopathology. 2016;68(7):968–976.
Peterson K, Lin E, Saffari H et al. Food-specific antibodies in oesophageal secretions: association with trigger foods in eosinophilic oesophagitis. Aliment Pharmacol Ther. 2020;52(6):997–1007.
Hirano I, Moy N, Heckman MG, Thomas CS, Gonsalves N, Achem SR. Endoscopic assessment of the oesophageal features of eosinophilic oesophagitis: validation of a novel classification and grading system. Gut. 2013;62(4):489–495.
Dellon ES, Liacouras CA, Molina-Infante J et al. Updated International Consensus Diagnostic Criteria for Eosinophilic Esophagitis: Proceedings of the AGREE Conference. Gastroenterology. 2018;155(4):1022–1033.
Schoepfer AM, Straumann A, Panczak R et al. Development and validation of a symptom-based activity index for adults with eosinophilic esophagitis. Gastroenterology. 2014;147(6):1255–1266.
Safroneeva E, Pan Z, King E et al. Long-Lasting Dissociation of Esophageal Eosinophilia and Symptoms After Dilation in Adults With Eosinophilic Esophagitis. Clin Gastroenterol Hepatol. 2022;20(4):766–775.
Liacouras CA, Furuta GT, Hirano I et al. Eosinophilic esophagitis: updated consensus recommendations for children and adults. J Allergy Clin Immunol. 2011;128(1):3–20.
Zhan T, Ali A, Choi JG et al. Model to Determine the Optimal Dietary Elimination Strategy for Treatment of Eosinophilic Esophagitis. Clin Gastroenterol Hepatol. 2018;16(11):1730–1737.
Hiramoto B, Zalewski A, Gregory D et al. Low Prevalence of Extraesophageal Gastrointestinal Pathology in Patients with Eosinophilic Esophagitis. Dig Dis Sci. 2022;67(7):3080–3088.
Quinn L, Nguyen B, Menard-Katcher C, Spencer L. IgG4+ cells are increased in the gastrointestinal tissue of pediatric patients with active eosinophilic gastritis and duodenitis and decrease in remission. Dig Liver Dis. 2023;55(1):53–60.
Kosaka S, Tanaka F, Nakata A et al. Gastrointestinal IgG4 Deposition Is a New Histopathological Feature of Eosinophilic Gastroenteritis. Dig Dis Sci. 2022;67(8):3639–3648.
Letner D, Farris A, Khalili H, Garber J. Pollen-food allergy syndrome is a common allergic comorbidity in adults with eosinophilic esophagitis. Dis Esophagus. 2018;31(2):15.
Mahdavinia M, Bishehsari F, Hayat W, Elhassan A, Tobin MC, Ditto AM. Association of eosinophilic esophagitis and food pollen allergy syndrome. Ann Allergy Asthma Immunol. 2017;118(1):116–117.
Schroeder S, Ochkur SI, Shim KP et al. Throat-derived eosinophil peroxidase is not a reliable biomarker of pediatric eosinophilic esophagitis. J Allergy Clin Immunol Pract. 2017;5(6):1804–1805.
Saffari H, Baer K, Boynton KK, Gleich GJ, Peterson KA. Pharyngeal mucosa brushing does not correlate with disease activity in patients with eosinophilic esophagitis. J Allergy Clin Immunol Pract. 2017;5(5):1455–1456.
Smeekens JM, Baloh C, Lim N et al. Peanut-Specific IgG4 and IgA in Saliva Are Modulated by Peanut Oral Immunotherapy. J Allergy Clin Immunol Pract. 2022;10(12):3270–3275.
Wright BL, Fernandez-Becker NQ, Kambham N et al. Gastrointestinal Eosinophil Responses in a Longitudinal, Randomized Trial of Peanut Oral Immunotherapy. Clin Gastroenterol Hepatol. 2021;19(6):1151–1159.
Ravi A, Marietta EV, Alexander JA et al. Mucosal penetration and clearance of gluten and milk antigens in eosinophilic oesophagitis. Aliment Pharmacol Ther. 2021;53(3):410–417.
Ohsaki A, Venturelli N, Buccigrosso TM et al. Maternal IgG immune complexes induce food allergen-specific tolerance in offspring. J Exp Med. 2018;215(1):91–113.
Acknowledgments
We would like to thank the patients at Mayo Clinic Arizona who participated in this study. We appreciate the assistance of Sara Farmer in the Mayo Clinic Survey Research Center for her assistance with the electronic questionnaires and database. We would also like to thank the members of the esophageal disease group (Marcelo Vela, MD, Francisco Ramirez, MD, and David Fleischer, MD), the allied health staff in the endoscopy suite, and the pathology research core and biorepository staff who assisted with specimen collection, storage and requisition. We acknowledge the support of clinical research coordinators Shannan Avelar and Rachel Kendrick. We thank Ekaterina Safroneeva, PhD for allowing us to use the EEsAI PRO and Evan Dellon, MD, MPH for his aid in interpreting the EEsAI scores.
Funding
This work was supported by the Arizona Biomedical Research Consortium (ADHS18-198880), Donald R. Levin Family Foundation, Mayo Clinic Foundation, and Phoenix Children's Hospital Foundation. M.Y.M. is a member of the Immunology Graduate Program and is supported by the Mayo Clinic Graduate School of Biomedical Sciences. B.L.W. also reports funding from NIH (L30 AI147030) and the Consortium of Eosinophilic Gastrointestinal Disease Researchers U54AI117804 (CEGIR), which is part of the Rare Disease Clinical Research Network (RDCRN), an initiative of the Office of Rare Disease Research (ORDR). CEGIR is also supported by patient advocacy groups, including American Partnership for Eosinophilic Disorders (APFED), Campaign Urging Research for Eosinophilic Diseases (CURED), and Eosinophilic Family Coalition (EFC). As a member of the RDCRN, CEGIR is also supported by its Data Management and Coordinating Center (DMCC) (U2CTR002818).
Author information
Authors and Affiliations
Contributions
BLW designed the study. TS recruited the subjects. JLH collected the human specimens. MYM, WEL, AP, JGB, and GCP performed the data collection. MYM, ADD, and BLW analyzed the data and wrote the initial draft of the manuscript. MRB performed the statistical analysis. All authors have reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
None to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Masuda, M.Y., LeSuer, W.E., Horsley-Silva, J.L. et al. Food-Specific IgG4 Is Elevated Throughout the Upper Gastrointestinal Tract in Eosinophilic Esophagitis. Dig Dis Sci 68, 2406–2413 (2023). https://doi.org/10.1007/s10620-023-07924-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-023-07924-2