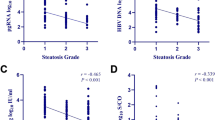
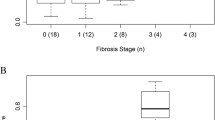
Abstract
Background
There are limited data on noninvasive methods to identify hepatic steatosis in coexisting hepatitis B virus (HBV) infection.
Aims
To evaluate the diagnostic performance of noninvasive serum-based scores to detect steatosis using two distinct chronic HBV cohorts with liver histology evaluation.
Methods
Chronic HBV cohorts with untreated HBV mono-infection (N = 302) and with treated HBV–HIV (N = 92) were included. Liver histology was scored centrally. Four serum-based scores were calculated: hepatic steatosis index (HSI), nonalcoholic fatty liver disease Liver Fat Score (NAFLD-LFS), visceral adiposity index (VAI), and triglyceride glucose (TyG) index. Optimal cutoffs (highest sensitivity + specificity) to detect ≥ 5% HS, stratified by cohort, were evaluated.
Results
HBV–HIV (vs. HBV mono-infected) patients were older (median 50 vs. 43 years), and a higher proportion were male (92% vs. 60%), were black (51% vs. 8%), had the metabolic syndrome (41% vs. 25%), and suppressed HBV DNA (< 1000 IU/mL; 82% vs. 9%). Applying optimal cutoffs, the area under the receiver operator curve for detecting ≥ 5% steatosis in HBV-only and HBV–HIV, respectively, was 0.69 and 0.61 for HSI, 0.70 and 0.76 for NAFLD-LFS, 0.68 and 0.64 for TyG, and 0.68 and 0.69 for VAI. The accuracy of optimal cutoffs ranged from 61% (NAFLD-LFS) to 67% (TyG) among HBV-only and 56% (HSI) to 76% (NAFLD-LFS) among HBV–HIV. Negative predictive values were higher than positive predictive values for all scores in both groups.
Conclusion
The relative utility of scores to identify steatosis in chronic HBV differs by co-infection/anti-HBV medication status. However, even with population-specific cutoffs, several common serum-based scores have only moderate utility. ClinicalTrials.gov NCT01924455.


Similar content being viewed by others
Abbreviations
- HBV:
-
Hepatitis B virus
- HIV:
-
Human immunodeficiency virus
- cART:
-
Combination antiretroviral therapy
- AIDS:
-
Acquired immunodeficiency syndrome
- TDF:
-
Tenofovir disoproxil fumarate
- FTC:
-
Emtricitabine
- 3TC:
-
Lamivudine
- HBRN:
-
Hepatitis B Research Network
- NIH:
-
National Institutes of Health
- NIDDK:
-
National Institute of Diabetes and Digestive and Kidney Diseases
- HBsAg:
-
Hepatitis B surface antigen
- HBsAb:
-
Anti-hepatitis B surface antibody
- HBeAg:
-
Hepatitis B envelop antigen
- HBeAb:
-
Anti-hepatitis B envelop antibody
- DNA:
-
Deoxyribonucleic acid
- HDV:
-
Hepatitis delta virus
- HCV:
-
Hepatitis C virus
- RNA:
-
Ribonucleic acid
- BMI:
-
Body mass index
- HAI:
-
Histologic activity index
- AST:
-
Aspartate aminotransferase
- ALT:
-
Alanine aminotransferase
- ALP:
-
Alkaline phosphatase
- NASH:
-
Nonalcoholic steatohepatitis
- IQR:
-
Interquartile range
- NAFLD-LFS:
-
Nonalcoholic fatty liver disease Liver Fat Score
- VAI:
-
Visceral adiposity index
- TyG:
-
Triglyceride glucose index
- HDL:
-
High-density lipoprotein
- LDL:
-
Low-density lipoprotein
- CAP:
-
Continuous attenuated parameter
- VCTE:
-
Vibration-controlled transient elastography
- NRTI:
-
Nucleoside reverse transcriptase inhibitor
- PI:
-
Protease inhibitor
References
Younossi ZM, Stepanova M, Younossi Yet al Epidemiology of chronic liver diseases in the USA in the past three decades. Gut 2020;69:564–568. https://doi.org/10.1136/gutjnl-2019-318813. Epub 2019 Jul 31
Younossi Z, Anstee QM, Marietti M et al. Global burden of NAHEPATIC STEATOSIS and NASH: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol. 2018;15:11–20
Unalp-Arida A, Ruhl CE. Noninvasive fatty liver markers predict liver disease mortality in the U.S. population. Hepatology. 2016;63:1170–83.
Lee YB, Ha Y, Chon YE et al. Association between hepatic steatosis and the development of hepatocellular carcinoma in patients with chronic hepatitis B. Clin Mol Hepatol. 2019;25:52–64
Chan AW, Wong GL, Chan HY et al. Concurrent fatty liver increases risk of hepatocellular carcinoma among patients with chronic hepatitis B. J Gastroenterol Hepatol. 2017;32:667–676
Suliman I, Abdelgelil N, Kassamali F, Hassanein TI. The effects of hepatic steatosis on the natural history of HBV infection. Clin Liver Dis. 2019;23:433–450
Khalili M, King W, Kleiner D, et al HBV-HIV Cohort Study of the Hepatitis B Research Network. Fatty Liver Disease in a Prospective North American Cohort of Adults with HIV and Hepatitis B Coinfection. Clin Infect Dis. 2020 Sep 1:ciaa1303. https://doi.org/10.1093/cid/ciaa1303. Online ahead of print.
Khalili M, King W, Kleiner D. Nonalcoholic Fatty Liver Disease (NAFLD) and its Clinical and Metabolic Correlates in a Prospective North American Cohort with HIV-HBV Coinfection on Combined Anti-Retroviral Therapy (cART). Hepatology. 2019;70:1362A–1363A
Shi FY, Jing L, Wenjun C et al. Fatty liver disease index: a simple screening tool to facilitate diagnosis of nonalcoholic fatty liver disease in the Chinese population. Dig Dis Sci. 2013;58:3326–3334. https://doi.org/10.1007/s10620-013-2774-y.
Demir K, Akyuz F, Ozdil S et al. What is the reason of elevated alanine aminotransferase level in HBeAg negative patients with low viremia: NAFLD or chronic hepatitis? Ann Hepatol. 2007;6:92–96
Fricker ZP, Pedley A, Massaro JM. Liver fat is associated with markers of inflammation and oxidative stress in analysis of data from the Framingham heart study. Clin Gastroenterol Hepatol. 2019;17:1157–1164
Li Y, Liu L, Wang B, Wang J, Chen D. Simple steatosis is a more relevant source of serum inflammatory markers than omental adipose tissue. Clin Res Hepatol Gastroenterol. 2014;38:46–54
Otgonsuren M, Estep MJ, Hossain N et al. Single non-invasive model to diagnose non-alcoholic fatty liver disease (NAHEPATIC STEATOSIS) and non-alcoholic steatohepatitis (NASH). J Gastroenterol Hepatol. 2014;29:2006–13
Sviklāne L, Olmane E, Dzērve Z et al. Fatty liver index and hepatic steatosis index for prediction of non-alcoholic fatty liver disease in type 1 diabetes. J Gastroenterol Hepatol. 2018;33:270–276
Gastaldelli A, Kozakova M, Højlund K et al RISC Investigators. Fatty liver is associated with insulin resistance, risk of coronary heart disease, and early atherosclerosis in a large European population. Hepatology. 2009;49:1537–44.
Paris R, Giral P, Khan JF et al LIDO Study Group. Fatty liver is an independent predictor of early carotid atherosclerosis. J Hepatol. 2016;65:95–102.
Kozakova M, Palombo C, Eng MP, et al RISC Investigators. Fatty liver index, gamma-glutamyltransferase, and early carotid plaques. Hepatology. 2012;55:1406–15.
Kowdley KV, Wang CC, Welch S, Roberts H, Brosgart CL. Prevalence of chronic hepatitis B among foreign-born persons living in the United States by country of origin. Hepatology. 2012;56:422–433
World Health Organization Hepatitis B Fact Sheet. https://www.who.int/news-room/fact-sheets/detail/hepatitis-b. Accessed November, 2020.
Schwenzer NF, Springer F, Schraml C et al. Noninvasive assessment and quantification of liver steatosis by ultrasound, computed tomography and magnetic resonance. J Hepatol. 2009;51:433–445
Bril F, Ortiz-Lopez C, Lomonaco R et al Clinical value of liver ultrasound for the diagnosis of nonalcoholic fatty liver disease in overweight and obese patients. Liver Int. 2015;35:2139–46.
Paige JS, Bernstein GS, Heba E, et al A Pilot Comparative Study of Quantitative Ultrasound, Conventional Ultrasound, and MRI for Predicting Histology-Determined Steatosis Grade in Adult Nonalcoholic Fatty Liver Disease. AJR Am J Roentgenol. 2017;208:W168-W177.
Siddiqui MS, Vuppalanchi R, Van Natta ML et al NASH Clinical Research Network. Vibration-Controlled Transient Elastography to Assess Fibrosis and Steatosis in Patients With Nonalcoholic Fatty Liver Disease. Clin Gastroenterol Hepatol. 2019;17:156–163.
Karlas T, Petroff D, Garnov N et al. Non-invasive assessment of hepatic steatosis in patients with NAFLD using controlled attenuation parameter and 1H-MR spectroscopy. PLoS One. 2014;9:e91987
Bedogni G, Bellentani S, Miglioli L et al. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006;2:33
Calori G, Lattuada G, Ragogna F et al. Fatty liver index and mortality: the Cremona study in the 15th year of follow-up. Hepatology. 2011;54:145–152
Lee JH, Kim D, Kim HJ. Hepatic steatosis index: a simple screening tool reflecting nonalcoholic fatty liver disease. Dig Liver Dis. 2010;42:503–508
Bedogni G, Kahn, Bellentani S, Tiribelli C, A simple index of lipid over accumulation is a good marker of liver steatosis. BMC Gastroenterol. 2010;25;10:98.
Cuthbertson DJ, Weickert MO, Lythgoe D et al. External validation of the fatty liver index and lipid accumulation product indices, using 1H-magnetic resonance spectroscopy, to identify hepatic steatosis in healthy controls and obese, insulin-resistant individuals. Eur J Endocrinol. 2014;171:561–569
Stern C, Castera L. Non-invasive diagnosis of hepatic steatosis. Hepatol Int. 2017;11:70–78
Festi D, Schiumerini R, Marzi L et al. Review article: the diagnosis of non-alcoholic fatty liver disease—availability and accuracy of non-invasive methods. Aliment Pharmacol Ther. 2013;37:392–400
Zhang Z, Wang G, Kang K, Wu G, Wang P. Diagnostic accuracy and clinical utility of a new noninvasive index for hepatic steatosis in patients with hepatitis B virus infection. Sci Rep. 2016;6:32875
Ooi GJ, Earnest A, Kemp WW, Burton PR, Laurie C, Majeed A, Johnson N, McLean C, Roberts SK, Brown WA. Evaluating feasibility and accuracy of non-invasive tests for nonalcoholic fatty liver disease in severe and morbid obesity. Int J Obes (Lond). 2018;42:1900–1911
Fedchuk L, Nascimbeni F, Pais R et al. LIDO Study Group. Performance and limitations of steatosis biomarkers in patients with nonalcoholic fatty liver disease. Aliment Pharmacol Ther. 2014;40:1209–22.
Kotronen A, Peltonen M, Hakkarainen A et al. Prediction of non-alcoholic fatty liver disease and liver fat using metabolic and genetic factors. Gastroenterology. 2009;137:865–872
Wlazlo N, van Greevenbroek MM, Ferreira I, Bravenboer B, Stehouwer CD. The diagnosis of non-alcoholic fatty liver disease. Aliment Pharmacol Ther. 2012;35:204–5; author reply205–6.)
Simental-Mendía LE, Simental-Mendía E, Rodríguez-Hernández H et al. The product of triglycerides and glucose as biomarker for screening simple steatosis and NASH in asymptomatic women. Ann Hepatol. 2016;15:715–20.
Petta S, Amato M, Cabibi D et al. Visceral adiposity index is associated with histological findings and high viral load in patients with chronic hepatitis C due to genotype 1. Hepatology. 2010;52:1543–1552
Musso G, Cassader M, Gambino R. Diagnostic accuracy of adipose insulin resistance index and visceral adiposity index for progressive liver histology and cardiovascular risk in nonalcoholic fatty liver disease. Hepatology. 2012;56:788–789
Amato MC, Giordano C, Galia M et al.; for the AlkaMeSy Study Group. Visceral adiposity index (VAI): a reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care. 2010;33:920–922
Guerrero-Romero F, Simental-Mendia LE, Gonzalez-Ortiz M, et al. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison with the euglycemic hyperinsulinemic clamp. J Clin Endocrinol Metab. 2010;95:3347–51.
Sterling RK, Wahed AS, King WC, et al. and the HBV-HIV Cohort Study of the Hepatitis B Research Network. Spectrum of Liver Disease in Hepatitis B Virus (HBV) Patients Co-infected with Human Immunodeficiency Virus (HIV): Results of the HBV-HIV Cohort Study. Am J Gastroenterol. 2019;114:746–757.
Ghany MG, Perrillo R, Li R et al. Hepatitis B Research Network; Hepatitis B Research Network. Characteristics of adults in the hepatitis B research network in North America reflect their country of origin and hepatitis B virus genotype. Clin Gastroenterol Hepatol. 2015;13:183–92.
Di Bisceglie AM, Lombardero M, Teckman et al. Research Network HBRN. Determination of hepatitis B phenotype using biochemical and serological markers. J Viral Hepat. 2017;24:320–329.
NIAAA. What is "low-risk" drinking? National Institute on Alcohol Abuse and Alcoholism. http://rethinkingdrinking.niaaa.nih.gov/IsYourDrinkingPatternRisky/WhatsLowRiskDrinking.asp, Accessed July 15, 2016
Kleiner DE, Brunt EM. Nonalcoholic fatty liver disease: pathologic patterns and biopsy evaluation in clinical research. Semin Liver Dis. 2012;32:3–13
Chon YE, Jung KS, Kim SU, Park JY, Park YN, Kim DY, Ahn SH, Chon CY, Lee HW, Park Y, Han KH. Controlled attenuation parameter (CAP) for detection of hepatic steatosis in patients with chronic liver diseases: a prospective study of a native Korean population. Liver Int. 2014;34:102–109
Xu L, Lu W, Li P et al. A comparison of hepatic steatosis index, controlled attenuation parameter and ultrasound as noninvasive diagnostic tools for steatosis in chronic hepatitis B. Dig Liver Dis. 2017;49:910–917
Borman MA, Ladak F, Crotty P et al. The Fatty Liver Index has limited utility for the detection and quantification of hepatic steatosis in obese patients. Hepatol Int. 2013;7:592–599
Prati D, Taioli E, Zanella A et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med. 2002;137:1–10
Funding
This study was funded by NIDDK (R01-DK94818) as an ancillary study (NCT01924455) of the Hepatitis B Research Network to Dr. Richard K. Sterling. Dr. Sulkowski was partially supported by K24DA034621. Dr. Khalili was partially supported by K24AA022523
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
Richard Sterling has received research grant from Abbott, AbbVie, Gilead, and Roche and serves on the data safety and monitoring board for Pfizer and Baxter. Mandana Khalili is a recipient of research grant (to her institution) from Gilead Sciences Inc. and Intercept Pharmaceuticals, and she has served as consultant for Gilead Sciences Inc. Raymond Chung has research grants (to institution) from Gilead, AbbVie, BMS, Merck, Boehringer, Roche, and Janssen. Mauricio Lisker-Melman serves on the speaker bureau for AbbVie, Gilead Sciences Inc., and SimplySpeaking. Mamta K. Jain has received research funding from Gilead Sciences, Janssen Pharmaceuticals, Merck, and GlaxoSmithKline. She has served on the scientific advisory board for Gilead Sciences. David Wong, Wendy King, Marc Ghany, and David Kleiner do not have any disclosures relevant to this project.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Human and animal rights
The studies have been approved by the appropriate institutional and/or national research ethics committee and have been performed in accordance with the ethical standards as laid down in the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary file (PDF 29 kb)
Flowchart of HBRN HBV mono-infected and HBV–HIV co-infected participants.
Rights and permissions
About this article
Cite this article
Sterling, R.K., King, W.C., Khalili, M. et al. Performance of Serum-Based Scores for Identification of Mild Hepatic Steatosis in HBV Mono-infected and HBV–HIV Co-infected Adults. Dig Dis Sci 67, 676–688 (2022). https://doi.org/10.1007/s10620-021-06860-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-021-06860-3