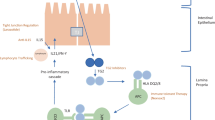
Abstract
Celiac disease is a common autoimmune disorder of the small intestine, triggered by an immunological response to the gluten present in wheat, barley, and rye in individuals who are genetically at risk. A key to reducing the complications of this disease is early diagnosis, preferably in childhood, and consuming a lifelong gluten-free diet once diagnosis is confirmed. Yet, the diagnosis of celiac disease is often considerably delayed, exposing patients to needless suffering and morbidity. It is also difficult to confirm histologically if dietary gluten has been restricted prior to obtaining a diagnostic biopsy, a significant problem given the current growing popularity of gluten-free diets. Furthermore, failure to understand or follow current guidelines means physicians may recommend patients commence the gluten-free diet before initiating referral to a gastroenterologist. Finally, adding further confusion, pediatric guidelines in Europe support a diagnosis based on serology rather than on histology, whereas those based in North America do not. The purpose of this review is to discuss these issues and other controversies in the diagnosis of celiac disease and to consider ways to optimize diagnosis across the lifespan.
Similar content being viewed by others
References
Kang JY, Kang AH, Green A, et al. Systematic review: worldwide variation in the frequency of coeliac disease and changes over time. Aliment Pharmacol Ther. 2013;38:226–245. https://doi.org/10.1111/apt.12373.
Fasano A, Berti I, Gerarduzzi T, et al. Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study. Arch Intern Med. 2003;163:286–292.
Rubio-Tapia A, Ludvigsson JF, Brantner TL, et al. The prevalence of celiac disease in the United States. Am J Gastroenterol. 2012;107:1538–1544. https://doi.org/10.1038/ajg.2012.219. (quiz 37, 45).
Husby S, Koletzko S, Korponay-Szabo IR, et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. J Pediatr Gastroenterol Nutr. 2012;54:136–160. https://doi.org/10.1097/MPG.0b013e31821a23d0.
Hill ID, Dirks MH, Liptak GS, et al. Guideline for the diagnosis and treatment of celiac disease in children: recommendations of the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr. 2005;40:1–19.
Hill ID, Fasano A, Guandalini S, et al. NASPGHAN clinical report on the diagnosis and treatment of gluten-related disorders. J Pediatr Gastroenterol Nutr. 2016;63:156–165. https://doi.org/10.1097/MPG.0000000000001216.
Ludvigsson JF, Bai JC, Biagi F, et al. Diagnosis and management of adult coeliac disease: guidelines from the British Society of Gastroenterology. Gut. 2014;63:1210–1228. https://doi.org/10.1136/gutjnl-2013-306578.
Murch S, Jenkins H, Auth M, et al. Joint BSPGHAN and Coeliac UK guidelines for the diagnosis and management of coeliac disease in children. Arch Dis Child. 2013;98:806–811. https://doi.org/10.1136/archdischild-2013-303996.
Rubio-Tapia A, Hill ID, Kelly CP, et al. ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol. 2013;108:656–676. https://doi.org/10.1038/ajg.2013.79. (quiz 77).
Hill P, Austin A, Forsyth J, Holmes G. British Society of Gastroenterology guidelines on the management of coeliac disease. Gut. 2015;64:691–692.
Giersiepen K, Lelgemann M, Stuhldreher N, et al. Accuracy of diagnostic antibody tests for coeliac disease in children: summary of an evidence report. J Pediatr Gastroenterol Nutr. 2012;54:229–241. https://doi.org/10.1097/MPG.0b013e318216f2e5.
Ribes-Koninckx C, Mearin ML, Korponay-Szabo IR, et al. Coeliac disease diagnosis: ESPGHAN 1990 criteria or need for a change? Results of a questionnaire. J Pediatr Gastroenterol Nutr. 2012;54:15–19. https://doi.org/10.1097/MPG.0b013e31822a00bb.
Beltran L, Koenig M, Egner W, et al. High-titre circulating tissue transglutaminase-2 antibodies predict small bowel villous atrophy, but decision cut-off limits must be locally validated. Clin Exp Immunol. 2014;176:190–198. https://doi.org/10.1111/cei.12249.
Saginur M, AlRefaee AM, Spady DW, et al. Antitissue transglutaminase antibody determination versus upper endoscopic biopsy diagnosis of paediatric celiac disease. Paediatrics Child Health. 2013;18:246–250.
Rajani S, Huynh HQ, Shirton L, et al. A Canadian study toward changing local practice in the diagnosis of pediatric celiac disease. Can J Gastroenterol Hepatol. 2016;2016:6234160. https://doi.org/10.1155/2016/6234160.
Fernandez-Banares F, Alsina M, Modolell I, et al. Are positive serum-IgA-tissue-transglutaminase antibodies enough to diagnose coeliac disease without a small bowel biopsy? Post-test probability of coeliac disease. J Crohn’s Colitis. 2012;6:861–866. https://doi.org/10.1016/j.crohns.2012.01.016.
Wolf J, Hasenclever D, Petroff D, et al. Antibodies in the diagnosis of coeliac disease: a biopsy-controlled, international, multicentre study of 376 children with coeliac disease and 695 controls. PLoS ONE. 2014;9:e97853. https://doi.org/10.1371/journal.pone.0097853.
Schirru E, Jores RD, Congia M. Prudence is necessary in the application of the new ESPGHAN criteria for celiac disease omitting duodenal biopsy: a case report. Eur J Gastroenterol Hepatol. 2014;26:679–680. https://doi.org/10.1097/MEG.0000000000000096.
Elitsur Y, Sigman T, Watkins R, et al. Tissue transglutaminase levels are not sufficient to diagnose celiac disease in north american practices without intestinal biopsies. Dig Dis Sci. 2017;62:175–179. https://doi.org/10.1007/s10620-016-4354-4.
De Leo L, Quaglia S, Ziberna F, et al. Serum anti-tissue transglutaminase antibodies detected during febrile illness may not be produced by the intestinal mucosa. J Pediatr. 2015;166:761–763. https://doi.org/10.1016/j.jpeds.2014.12.005.
Wolf J, Petroff D, Richter T, et al. Validation of antibody-based strategies for diagnosis of pediatric celiac disease without biopsy. Gastroenterology. 2017;153:410–419. https://doi.org/10.1053/j.gastro.2017.04.023. (e17).
Trovato CM, Montuori M, Anania C, et al. Are ESPGHAN “biopsy-sparing” guidelines for celiac disease also suitable for asymptomatic patients? Am J Gastroenterol. 2015;110:1485–1489. https://doi.org/10.1038/ajg.2015.285.
Smarrazzo A, Misak Z, Costa S, et al. Diagnosis of celiac disease and applicability of ESPGHAN guidelines in Mediterranean countries: a real life prospective study. BMC Gastroenterol. 2017;17:17. https://doi.org/10.1186/s12876-017-0577-x.
Paul SP, Harries SL, Basude D. Barriers to implementing the revised ESPGHAN guidelines for coeliac disease in children: a cross-sectional survey of coeliac screen reporting in laboratories in England. Arch Dis Child. 2017;102:942–946. https://doi.org/10.1136/archdischild-2016-312027.
Benelli E, Carrato V, Martelossi S, et al. Coeliac disease in the ERA of the new ESPGHAN and BSPGHAN guidelines: a prospective cohort study. Arch Dis Child. 2016;101:172–176. https://doi.org/10.1136/archdischild-2015-309259.
Efthymakis K, Serio M, Milano A, et al. Application of the biopsy-sparing ESPGHAN guidelines for celiac disease diagnosis in adults: a real-life study. Dig Dis Sci. 2017;62:2433–2439. https://doi.org/10.1007/s10620-017-4672-1.
Di Tola M, Marino M, Goetze S, et al. Identification of a serum transglutaminase threshold value for the noninvasive diagnosis of symptomatic adult celiac disease patients: a retrospective study. J Gastroenterol. 2016;51:1031–1039. https://doi.org/10.1007/s00535-016-1188-y.
Zanini B, Magni A, Caselani F, et al. High tissue-transglutaminase antibody level predicts small intestinal villous atrophy in adult patients at high risk of celiac disease. Dig Liver Dis. 2012;44:280–285. https://doi.org/10.1016/j.dld.2011.10.013.
Casella S, Zanini B, Lanzarotto F, et al. Celiac disease in elderly adults: clinical, serological, and histological characteristics and the effect of a gluten-free diet. J Am Geriatr Soc. 2012;60:1064–1069. https://doi.org/10.1111/j.1532-5415.2012.03997.x.
Rubio-Tapia A, Rahim MW, See JA, et al. Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am J Gastroenterol. 2010;105:1412–1420. https://doi.org/10.1038/ajg.2010.10.
Lebwohl B, Granath F, Ekbom A, et al. Mucosal healing and mortality in coeliac disease. Aliment Pharmacol Ther. 2013;37:332–339. https://doi.org/10.1111/apt.12164.
Lebwohl B, Murray JA, Rubio-Tapia A, et al. Predictors of persistent villous atrophy in coeliac disease: a population-based study. Aliment Pharmacol Ther. 2014;39:488–495. https://doi.org/10.1111/apt.12621.
Silvester JA, Kurada S, Szwajcer A, et al. Tests for serum transglutaminase and endomysial antibodies do not detect most patients with celiac disease and persistent villous atrophy on gluten-free diets: a meta-analysis. Gastroenterology. 2017;153:689–701. https://doi.org/10.1053/j.gastro.2017.05.015. (e1).
Leonard MM, Weir DC, DeGroote M, et al. Value of IgA tTG in predicting mucosal recovery in children with celiac disease on a gluten-free diet. J Pediatr Gastroenterol Nutr. 2017;64:286–291. https://doi.org/10.1097/MPG.0000000000001460.
Ghazzawi Y, Rubio-Tapia A, Murray JA, et al. Mucosal healing in children with treated celiac disease. J Pediatr Gastroenterol Nutr. 2014;59:229–231. https://doi.org/10.1097/MPG.0000000000000390.
Haere P, Hoie O, Schulz T, et al. Long-term mucosal recovery and healing in celiac disease is the rule—not the exception. Scand J Gastroenterol. 2016;51:1439–1446. https://doi.org/10.1080/00365521.2016.1218540.
Beg S, Ragunath K, Wyman A, et al. Quality standards in upper gastrointestinal endoscopy: a position statement of the British Society of Gastroenterology (BSG) and Association of Upper Gastrointestinal Surgeons of Great Britain and Ireland (AUGIS). Gut. 2017;66:1886–1899. https://doi.org/10.1136/gutjnl-2017-314109.
Weir DC, Glickman JN, Roiff T, et al. Variability of histopathological changes in childhood celiac disease. Am J Gastroenterol. 2010;105:207–212. https://doi.org/10.1038/ajg.2009.557.
Ravelli A, Villanacci V, Monfredini C, et al. How patchy is patchy villous atrophy?: distribution pattern of histological lesions in the duodenum of children with celiac disease. Am J Gastroenterol. 2010;105:2103–2110. https://doi.org/10.1038/ajg.2010.153.
Ofei S, Boyle B, Ediger T, et al. Adherence to endoscopy biopsy guidelines for celiac disease. J Pediatr Gastroenterol Nutr. 2015;61:440–444. https://doi.org/10.1097/MPG.0000000000000834.
Lebwohl B, Kapel RC, Neugut AI, et al. Adherence to biopsy guidelines increases celiac disease diagnosis. Gastrointest Endosc. 2011;74:103–109. https://doi.org/10.1016/j.gie.2011.03.1236.
Rostami-Nejad M, Villanacci V, Hogg-Kollars S, et al. Endoscopic and histological pitfalls in the diagnosis of celiac disease: a multicentre study assessing the current practice. Revista espanola de enfermedades digestivas: organo oficial de la Sociedad Espanola de Patologia Digestiva. 2013;105:326–333.
Latorre M, Lagana SM, Freedberg DE, et al. Endoscopic biopsy technique in the diagnosis of celiac disease: one bite or two? Gastrointest Endosc. 2015;81:1228–1233. https://doi.org/10.1016/j.gie.2014.10.024.
Taavela J, Popp A, Kurppa K, et al. Is there a role for duodenal bulb biopsies in celiac disease diagnostics? Clin Gastroenterol Hepatol. 2016;14:1510–1511. https://doi.org/10.1016/j.cgh.2016.04.014.
Stoven SA, Choung RS, Rubio-Tapia A, et al. Analysis of biopsies from duodenal bulbs of all endoscopy patients increases detection of abnormalities but has a minimal effect on diagnosis of celiac disease. Clin Gastroenterol Hepatol. 2016;14:1582–1588. https://doi.org/10.1016/j.cgh.2016.02.026.
Taavela J, Popp A, Korponay-Szabo IR, et al. A prospective study on the usefulness of duodenal bulb biopsies in celiac disease diagnosis in children: urging caution. Am J Gastroenterol. 2016;111:124–133. https://doi.org/10.1038/ajg.2015.387.
Caruso R, Marafini I, Del Vecchio Blanco G, et al. Sampling of proximal and distal duodenal biopsies in the diagnosis and monitoring of celiac disease. Dig Liver Dis. 2014;46:323–329. https://doi.org/10.1016/j.dld.2013.12.005.
Memeo L, Jhang J, Hibshoosh H, et al. Duodenal intraepithelial lymphocytosis with normal villous architecture: common occurrence in H. pylori gastritis. Mod Pathol. 2005;18:134–144. https://doi.org/10.1038/modpathol.3800404.
Marsh MN. Coeliac disease, mucosal change and IEL: doing what counts the best. Gastroenterol Hepatol Bed Bench. 2016;9:1–5.
Lahdeaho ML, Kaukinen K, Collin P, et al. Celiac disease: from inflammation to atrophy: a long-term follow-up study. J Pediatr Gastroenterol Nutr. 2005;41:44–48.
Auricchio R, Tosco A, Piccolo E, et al. Potential celiac children: 9-year follow-up on a gluten-containing diet. Am J Gastroenterol. 2014;109:913–921. https://doi.org/10.1038/ajg.2014.77.
Oberhuber G, Granditsch G, Vogelsang H. The histopathology of coeliac disease: time for a standardized report scheme for pathologists. Eur J Gastroenterol Hepatol. 1999;11:1185–1194.
Rostami K, Marsh MN, Johnson MW, et al. ROC-king onwards: intraepithelial lymphocyte counts, distribution & role in coeliac disease mucosal interpretation. Gut. 2017;66:2080–2086. https://doi.org/10.1136/gutjnl-2017-314297.
Tortora R, Capone P, Imperatore N, et al. Predictive value of “Marsh 1” type histology in subjects with suspected cealic disease. Scand J Gastroenterol. 2014;49:801–806. https://doi.org/10.3109/00365521.2014.919019.
Monten C, Bjelkenkrantz K, Gudjonsdottir AH, et al. Validity of histology for the diagnosis of paediatric coeliac disease: a Swedish multicentre study. Scand J Gastroenterol. 2016;51:427–433. https://doi.org/10.3109/00365521.2015.1101486.
Kaur Bilkhoo H, Ducruet T, Marchand V, et al. Revisiting pathological criteria for earlier diagnosis of coeliac disease. J Pediatr Gastroenterol Nutr. 2016;62:734–738. https://doi.org/10.1097/MPG.0000000000001026.
Eigner W, Wrba F, Chott A, et al. Early recognition of possible pitfalls in histological diagnosis of celiac disease. Scand J Gastroenterol. 2015;50:1088–1093. https://doi.org/10.3109/00365521.2015.1017835.
Arguelles-Grande C, Tennyson CA, Lewis SK, et al. Variability in small bowel histopathology reporting between different pathology practice settings: impact on the diagnosis of coeliac disease. J Clin Pathol. 2012;65:242–247. https://doi.org/10.1136/jclinpath-2011-200372.
Agriculture and Agri-Food Canada. Market Analysis: food intolerance products in the United States: lactose-free, gluten-free and diabetic food. October 2013. http://www.agr.gc.ca/resources/prod/internet-internet/MISB-DGSIM/ATS-SEA/PDF/6429-eng.pdf.
Moore LR. “But we’re not hypochondriacs”: the changing shape of gluten-free dieting and the contested illness experience. Soc Sci Med. 2014;105:76–83. https://doi.org/10.1016/j.socscimed.2014.01.009.
Silvester JA, Weiten D, Graff LA, et al. Is it gluten-free? Relationship between self-reported gluten-free diet adherence and knowledge of gluten content of foods. Nutrition. 2016;32:777–783. https://doi.org/10.1016/j.nut.2016.01.021.
Laurin P, Wolving M, Falth-Magnusson K. Even small amounts of gluten cause relapse in children with celiac disease. J Pediatr Gastroenterol Nutr. 2002;34:26–30.
Bruins MJ. The clinical response to gluten challenge: a review of the literature. Nutrients. 2013;5:4614–4641. https://doi.org/10.3390/nu5114614.
Sarna VK, Lundin KEA, Morkrid L, et al. HLA-DQ-Gluten tetramer blood test accurately identifies patients with and without celiac disease in absence of gluten consumption. Gastroenterology. 2017;. https://doi.org/10.1053/j.gastro.2017.11.006.
Choung RS, Larson SA, Khaleghi S, et al. Prevalence and morbidity of undiagnosed celiac disease from a community-based study. Gastroenterology. 2017;152:830–839. https://doi.org/10.1053/j.gastro.2016.11.043. (e5).
Corrao G, Corazza GR, Bagnardi V, et al. Mortality in patients with coeliac disease and their relatives: a cohort study. Lancet. 2001;358:356–361.
Paez MA, Gramelspacher AM, Sinacore J, et al. Delay in diagnosis of celiac disease in patients without gastrointestinal complaints. Am J Med. 2017;130:1318–1323. https://doi.org/10.1016/j.amjmed.2017.05.027.
Vavricka SR, Vadasz N, Stotz M, et al. Celiac disease diagnosis still significantly delayed—doctor’s but not patients’ delay responsive for the increased total delay in women. Dig Liver Dis. 2016;48:1148–1154. https://doi.org/10.1016/j.dld.2016.06.016.
Pham BN, Musset L, Chyderiotis G, et al. Celiac disease diagnosis: impact of guidelines on medical prescription in France. J Dig Dis. 2014;15:435–443. https://doi.org/10.1111/1751-2980.12158.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no conflict of interest.
Rights and permissions
About this article
Cite this article
Turner, J.M. Diagnosis of Celiac Disease: Taking a Bite Out of the Controversy. Dig Dis Sci 63, 1384–1391 (2018). https://doi.org/10.1007/s10620-018-5050-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-018-5050-3