Abstract
Background and Aim
Nonalcoholic fatty liver disease (NAFLD) affects 15–40 % of the general population; 10–20 % of those patients have a more severe form of the disease known as nonalcoholic steatohepatitis (NASH). Cytokeratin-18 (CK18), released during apoptosis and one of the most studied biomarkers in NASH, can be measured by a number of commercially available kits. We compared serum measurements of the CK18 M30 from two different kits using the same cohort to evaluate the reliability between two test kits.
Methods
We measured serum levels of CK18 M30 from 185 patients with biopsy-proven NAFLD from a single center from 2009 to 2015, using two different ELISA kits, Test 1 (T1) and Test 2 (T2). Advanced fibrosis was defined as fibrosis stages 3–4 and NASH defined by NAS score ≥5.
Results
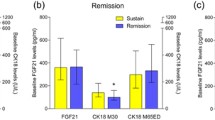
Mean age was 50.2 years (SD 12.6), 61.1 % male and 87 % White; 49.6 % had NASH and 32.2 % advanced fibrosis. There was no significant correlation between measurements from the two kits (p = 0.86, r = 0.01). While T2 predicted NASH and advanced fibrosis, T1 did not. The area under ROC curve for the prediction of NASH was 0.631 for T2 versus 0.500 for T1.
Conclusions
Measurements from two different CK18 M30 test kits did not correlate with each other. One kit showed statistically significantly higher levels of CK18 M30 in patients with advanced fibrosis and NASH, while the other kit did not. With the increasing use of CK18 as a biomarker in NASH, it is important to standardize the different kits as it could greatly bias the results.

Similar content being viewed by others
Abbreviations
- NAFLD:
-
Nonalcoholic liver disease
- NASH:
-
Nonalcoholic steatohepatitis
- CK18:
-
Cytokeratin 18
- AUROC:
-
Area under the receiver operating characteristic curve
- NAFL:
-
Nonalcoholic fatty liver, simple steatosis
- HR:
-
Hazard ratio
References
Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther. 2011;34:274–285.
Browning JD, Szczepaniak LS, Dobbins R, et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology. 2004;40:1387–1395.
Williams CD, Stengel J, Asike MI, et al. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: a prospective study. Gastroenterology. 2011;140:124–131.
Younossi ZM, Stepanova M, Afendy M, et al. Changes in the prevalence of the most common causes of chronic liver diseases in the United States From 1988 to 2008. Clin Gastroenterol Hepatol. 2011;9:524–530.
Adams LA, Sanderson S, Lindor KD, Angulo P. The histological course of nonalcoholic fatty liver disease: a longitudinal study of 103 patients with sequential liver biopsies. J Hepatol. 2005;42:132–138.
Teli MR, James OF, Burt AD, Bennett MK, Day CP. The natural history of nonalcoholic fatty liver: a follow-up study. Hepatology. 1995;22:1714–1719.
Starley BQ, Calcagno CJ, Harrison SA. Nonalcoholic fatty liver disease and hepatocellular carcinoma: a weighty connection. Hepatology. 2010;51:1820–1832.
Bugianesi E, Leone N, Vanni E, et al. Expanding the natural history of nonalcoholic steatohepatitis: from cryptogenic cirrhosis to hepatocellular carcinoma. Gastroenterology. 2002;123:134–140.
Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology. 2012;142:1592–1609.
Ong JP, Younossi ZM. Epidemiology and natural history of NAFLD and NASH. Clin Liver Dis. 2007;11:1–16.
Singh S, Allen AM, Wang Z, Prokop LJ, Murad MH, Loomba R. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin Gastroenterol Hepatol. 2015;13:643.e9–654.e9.
McPherson S, Hardy T, Henderson E, Burt AD, Day CP, Anstee QM. Evidence of NAFLD progression from steatosis to fibrosing-steatohepatitis using paired biopsies: implications for prognosis and clinical management. J Hepatol. 2014;62:1148–1155.
Pais R, Charlotte F, Fedchuk L, et al. A systematic review of follow-up biopsies reveals disease progression in patients with non-alcoholic fatty liver. J Hepatol. 2013;59:550–556.
Ekstedt M, Hagström H, Nasr P, et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology. 2015;61:1547–1554.
Kramer G, Erdal H, Mertens HJMM, et al. Differentiation between cell death modes using measurements of different soluble forms of extracellular cytokeratin 18. Cancer Res. 2004;64:1751–1756.
Wieckowska A, Zein NN, Yerian LM, Lopez AR, McCullough AJ, Feldstein AE. In vivo assessment of liver cell apoptosis as a novel biomarker of disease severity in nonalcoholic fatty liver disease. Hepatology. 2006;44:27–33.
Yilmaz Y. Systematic review: caspase-cleaved fragments of cytokeratin 18—the promises and challenges of a biomarker for chronic liver disease. Aliment Pharmacol Ther. 2009;30:1103–1109.
Aida Y, Abe H, Tomita Y, et al. Serum cytokeratin 18 fragment level as a noninvasive biomarker for non-alcoholic fatty liver disease. Int J Clin Exp Med. 2014;7:4191–4198.
Younossi ZM, Jarrar M, Nugent C, et al. A novel diagnostic biomarker panel for obesity-related nonalcoholic steatohepatitis (NASH). Obes Surg. 2008;18:1430–1437.
Cao W, Zhao C, Shen C, Wang Y. Cytokeratin 18, alanine aminotransferase, platelets and triglycerides predict the presence of nonalcoholic steatohepatitis. PLoS One. 2013;8:1–8.
Anty R, Iannelli A, Patouraux S, Bonnafous S, Lavallard VJ, Amor IB. A new composite model including metabolic syndrome, alanine aminotransferase and cytokeratin-18 for the diagnosis of non-alcoholic steatohepatitis in morbidly obese patients. Aliment Pharmacol Ther. 2010;32:1315–1322.
Feldstein AE, Wieckowska A, Lopez AR, Liu YC, Zein NN, McCullough AJ. Cytokeratin-18 fragment levels as noninvasive biomarkers for nonalcoholic steatohepatitis: a multicenter validation study. Hepatology. 2009;50:1072–1078.
Chan W-K, Sthaneshwar P, Mustapha NRN, Mahadeva S. Limited utility of plasma M30 in discriminating non-alcoholic steatohepatitis from steatosis—a comparison with routine biochemical markers. PLoS One. 2014;9:e105903.
Cusi K, Chang Z, Harrison S, et al. Limited value of plasma cytokeratin-18 as a biomarker for NASH and fibrosis in patients with non-alcoholic fatty liver disease. J Hepatol. 2014;60:167–174.
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313–1321.
Yilmaz Y, Dolar E, Ulukaya E, et al. Soluble forms of extracellular cytokeratin 18 may differentiate simple steatosis from nonalcoholic steatohepatitis. World J Gastroenterol. 2007;13:837–844.
Shen J, Chan HL-Y, Wong GL-H, et al. Assessment of non-alcoholic fatty liver disease using serum total cell death and apoptosis markers. Aliment Pharmacol Ther. 2012;36:1057–1066.
Acknowledgments
This publication was supported by NIDDK Grant 1K23DK083439 to Michelle Lai, MD (PI). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIDDK or the NIH.
Author contributions
C.F.M.G.P. and M.L. were involved in the primary study design and data analysis; C.F.M.G.P., Z.G.J., T.O., and L.F. were involved in CK18 level measurements; T.L.C. and I.N. were responsible for liver pathology analysis; S.R. and N.A. were responsible for manuscript analysis and comments; all authors participated in the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Pimentel, C.F.M.G., Jiang, Z.G., Otsubo, T. et al. Poor Inter-test Reliability Between CK18 Kits as a Biomarker of NASH. Dig Dis Sci 61, 905–912 (2016). https://doi.org/10.1007/s10620-015-3916-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3916-1