Abstract
Background
Impact of first-line induction therapy on medium-term outcomes in the setting of early thiopurine (TP) use in children with Crohn’s disease has not been evaluated, in particular whether choice of exclusive enteral nutrition (EEN) over corticosteroids (CS) for induction impacts clinical outcomes at 12 and 24 months.
Aims and Methods
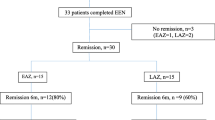
In this retrospective study, 89 children from our database with new diagnosis CD and follow-up of at least 2 years following induction with exclusive course of CS or EEN and early, dose-optimized TP (within 6 months from diagnosis) were evaluated. We compared steroid dependency (relapse <3 months of tapering first course CS or inability to wean <10 mg prednisolone), need for IFX, linear growth, and surgical resections over the first 2 years.
Results
Choice of EEN over CS induction was associated with reduced linear growth failure (7 vs. 26 %, p = 0.02), CS dependency (7 vs. 43 %, p = 0.002), and improved primary sustained response to IFX (86 vs. 68 %, p = 0.02). Combined CS/IFX-free remission and surgical resection rates were similar.
Conclusion
In the setting of early TP commencement, EEN induction is superior to CS induction for reducing growth failure, CS dependency, and loss of response to IFX over the first 2 years.
Similar content being viewed by others
References
Vernier-Massouille G, Balde M, Salleron J, et al. Natural history of pediatric Crohn’s disease: a population-based cohort study. Gastroenterology. 2008;135:1106–1113.
Peneau A, Salleron J, Fumery M, et al. P137, Long term outcome of pediatric onset Crohn’s disease: A population based study, ECCO abstracts 2012.
Van Limbergen J, Russell RK, Drummond HE, et al. Definition of phenotypic characteristics of childhood-onset inflammatory bowel disease. Gastroenterology. 2008;135:1114–1122.
Pigneur B, Seksik P, Viola S, et al. Natural history of Crohn’s disease: comparison between childhood- and adult-onset disease. Inflamm Bowel Dis. 2010;16:953–961.
Whelan G, Farmer RG, Fazio VW, et al. Recurrence after surgery in Crohn’s disease. Relationship to location of disease (clinical pattern) and surgical indication. Gastroenterology. 1985;88:1826–1833.
Castile RG, Telander RL, Cooney D, et al. Crohn’s disease in children: assessment of the progression of disease, growth, and prognosis. J Pediatr Surg. 1980;15:462–469.
Ruemmele FM, Veres G, Kolho KL, et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J Crohns Colitis. 2014;8:1179–1207.
Markowitz J, Grancher K, Kohn N, Lesser M, et al. A multicenter trial of 6-mercaptopurine and prednisone in children with newly diagnosed Crohn’s disease. Gastroenterology. 2000;119:895.
Jaspers G, Verkade H, Escher J, et al. Azathioprine maintains first remission in newly diagnosed pediatric Crohn’s disease. Inflamm Bowel Dis. 2006;12:831–836.
Waters T, Kim M, Denson L, et al. Increased effectiveness of early therapy with anti-tumor necrosis factor-α vs an immunomodulator in children with Crohn’s disease. Gastroenterology. 2014;146:383–391.
Riello L, Talbotec C, Garnier-Lengline H, et al. Tolerance and efficacy of azathioprine in pediatric Crohn’s disease. Inflamm Bowel Dis. 2011;17:2138–2143.
Boyle B, Kappelman M, Colletti R, et al. Routine use of thiopurines in maintaining remission in paediatric Crohn’s disease. World J Gastroenterol. 2014;20:9185–9190.
Cosnes J, Bourrier A, Laharie D, et al. Early administration of azathioprine vs conventional management of Crohn’s disease: a randomized controlled trial. Gastroenterology. 2013;145:758–765.
Panes J, Lopez-Sanroma A, Bermejo F, et al. Early azathioprine therapy is no more effective than placebo for newly diagnosed Crohn’s disease. Gastroenterology. 2013;145:766–774.
Levine A, Griffiths A, Markowitz J, et al. Pediatric modification of the Montreal classification for inflammatory bowel disease: the Paris classification. Inflamm Bowel Dis. 2011;17:1314–1321.
Lee J, Escher J, Shuman M, et al. Final adult height of children with inflammatory bowel disease is predicted by parental height and patient minimum height Z-score. Inflamm Bowel Dis. 2010;16:1669–1677.
Cole TJ, Flegal KM, Nicholls D, et al. Body mass index cut offs to define thinness in children and adolescents: international survey. BMJ. 2007;335:194–202.
Hyams J, Ferry G, Mandel F, et al. Development and validation of paediatric Crohn’s disease activity index. J Pediatr Gastroenterol Nutr. 1991;12:439–447.
Hyams J, Markowitz J, Otley A, et al. Evaluation of the pediatric Crohn disease activity index: a prospective multicenter experience. J Pediatr Gastroenterol Nutr. 2005;41:416–421.
Daperno M, D’Haens G, Van Assche G, et al. Development and validation of a new, simplified endoscopic activity score for Crohn’s disease: the SES-CD. Gastrointest Endosc. 2004;60:505–512.
Schoepfer AM, Beglinger C, Straumann A, et al. Fecal calprotectin correlates more closely with the Simple Endoscopic Score for Crohn’s disease (SES-CD) than CRP, blood leukocytes, and the CDAI. Am J Gastroenterol. 2010;105:162–169.
Grover Z, Muir R, Lewindon P. Exclusive enteral nutrition induces early clinical, mucosal and transmural remission in paediatric Crohn’s disease. J Gastroenterol. 2014;49:638–645.
Van Assche G, Dignass A, Panes J, et al. The second European evidence-based consensus on the diagnosis and management of Crohn’s disease: definitions and diagnosis. J Crohns Colitis. 2010;4:7–27.
Dubinsky MC, Lamothe S, Yang HY, et al. Pharmacogenomics and metabolite measurement for 6-mercaptopurine therapy in inflammatory bowel disease. Gastroenterology. 2000;118:705–713.
Grover Z, Biron R, Carman N, et al. Predictors of response to infliximab in children with luminal Crohn’s disease. J Crohns Colitis. 2014;8:739–746.
Sazuka S, Katsuno T, Nakagawa T, et al. Concomitant use of enteral nutrition therapy is associated with sustained response to infliximab in patients with Crohn’s disease. Eur J Clin Nutr. 2012;66:1219–1223.
Hirai F, Ishihara H, Yada S, et al. Effectiveness of concomitant enteral nutrition therapy and Infliximab for maintenance treatment of Crohn’s disease in adults. Dig Dis Sci. 2013;58:1329–1334. doi:10.1007/s10620-012-2374-2.
Lambert B, Lemberg A, Leach ST, et al. Longer-term outcomes of nutritional management of Crohn’s disease in children. Dig Dis Sci. 2012;57:2171–2177. doi:10.1007/s10620-012-2232-2.
Berni Canani R, Terrin G, Borrelli O, et al. Short- and long-term therapeutic efficacy of nutritional therapy and corticosteroids in paediatric Crohn’s disease. Dig Liver Dis.. 2006;38:381–387.
Cameron F, Gerasimidis K, Papangelou A, et al. Clinical progress in the two years following a course of exclusive enteral nutrition in 109 paediatric patients with Crohn’s disease. Aliment Pharmacol Ther. 2013;37:622–629.
Conflict of interest
Dr. Zubin Grover received ANZ trustees PHD and Queensland Children Medical Research Scholarship. He is a Paediatric Gastroenterology Advisory Board member for Abbvie. A/Prof Peter Lewindon is on Advisory Board panel of Janseen and Abbvie.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Grover, Z., Lewindon, P. Two-Year Outcomes After Exclusive Enteral Nutrition Induction Are Superior to Corticosteroids in Pediatric Crohn’s Disease Treated Early with Thiopurines. Dig Dis Sci 60, 3069–3074 (2015). https://doi.org/10.1007/s10620-015-3722-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3722-9