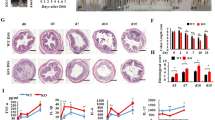
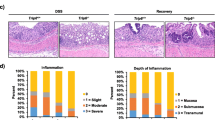
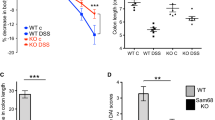
Abstract
Background
In order for vitamin D to signal and regulate inflammatory pathways, it must bind to its receptor (VDR) which must heterodimerize with the retinoid X receptor alpha (RXRα). Although the role that vitamin D signaling plays in the development and progression of colitis, a disease characterized by excessive inflammation, has been suggested, little research has been done on determining the role that RXRα plays in acute colitis development.
Aims
This study sought to determine the effects that reduced availability of RXRα would have on the development of acute murine colitis. Expression of inflammatory markers, VDR and RXRα were investigated to determine if the reduction in expression of RXRα in RXRα+/− mice would result in increased inflammatory signaling and receptor downregulation as compared to their wild-type littermates.
Methods
An acute murine model of colitis, the axozymethane (AOM) and dextran sulfate sodium (DSS) model was utilized in wild-type and RXRα+/− mice. Gross manifestations of colitis measured included weight loss and colitis score. Immunblots and real-time PCR were performed for inflammatory markers and receptor expression.
Results
RXRα+/− mice induced with AOM/DSS colitis demonstrated increased gene expression of Snail and Snail2, transcription factors downstream of inflammatory mediators, as compared to their wild-type littermates.
Conclusions
This demonstrates the importance of RXRα in regulating inflammation in acute colitis and also identifies RXRα expression as a new consideration when developing successful interventions for acute colitis due to the requirement of numerous receptors for RXRα.




Similar content being viewed by others
References
Heyman RA, Mangelsdorf DJ, Dyck JA, Stein RB, et al. 9-cis retinoic acid is a high affinity ligand for the retinoid X receptor. Cell. 1992;68:397–406.
Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, et al. The nuclear receptor superfamily: the second decade. Cell. 1995;83:835–839.
Carlberg C, Polly P. Gene regulation by vitamin D3. Crit Rev Eukaryot Gene Expr. 1998;8:19–42.
Kurokawa R, Yu VC, Naar A, Kyakumoto S, et al. Differential orientations of the DNA-binding domain and carboxy-terminal dimerization interface regulate binding site selection by nuclear receptor heterodimers. Genes Dev. 1993;7:1423–1435.
Mackey SL, Heymont JL, Kronenberg HM, Demay MB. Vitamin D receptor binding to the negative human parathyroid hormone vitamin D response element does not require the retinoid x receptor. Mol Endocrinol. 1996;10:298–305.
Demay MB, Kiernan MS, DeLuca HF, Kronenberg HM. Sequences in the human parathyroid hormone gene that bind the 1,25-dihydroxyvitamin D3 receptor and mediate transcriptional repression in response to 1,25-dihydroxyvitamin D3. Proc Natl Acad Sci USA. 1992;89:8097–8101.
Haussler MR, Haussler CA, Jurutka PW, Thompson PD, et al. The vitamin D hormone and its nuclear receptor: molecular actions and disease states. J Endocrinol. 1997;154:S57–S73.
Muto A, Kizaki M, Yamato K, Kawai Y, et al. 1,25-Dihydroxyvitamin D3 induces differentiation of a retinoic acid-resistant acute promyelocytic leukemia cell line (UF-1) associated with expression of p21(WAF1/CIP1) and p27(KIP1). Blood. 1999;93:2225–2233.
Liu M, Lee MH, Cohen M, Bommakanti M, et al. Transcriptional activation of the Cdk inhibitor p21 by vitamin D3 leads to the induced differentiation of the myelomonocytic cell line U937. Genes Dev. 1996;10:142–153.
Sinkkonen L, Malinen M, Saavalainen K, Vaisanen S, et al. Regulation of the human cyclin C gene via multiple vitamin D3-responsive regions in its promoter. Nucleic Acids Res. 2005;33:2440–2451.
Tu-Yu AH, Morris RC, Ives HE. Differential modulation of fos and jun gene expression by 1,25-dihydroxyvitamin D3. Biochem Biophys Res Commun. 1993;193:161–166.
Yanagisawa J, Yanagi Y, Masuhiro Y, Suzawa M, et al. Convergence of transforming growth factor-beta and vitamin D signaling pathways on SMAD transcriptional coactivators. Science. 1999;283:1317–1321.
Levin AD, Wadhera V, Leach ST, Woodhead HJ, et al. Vitamin D deficiency in children with inflammatory bowel disease. Dig Dis Sci. 2011;56:830–836.
Ulitsky A, Ananthakrishnan AN, Naik A, Skaros S, et al. Vitamin D deficiency in patients with inflammatory bowel disease: association with disease activity and quality of life. JPEN J Parenter Enteral Nutr. 2011;35:308–316.
Su CG, Wen X, Bailey ST, Jiang W, et al. A novel therapy for colitis utilizing PPAR-gamma ligands to inhibit the epithelial inflammatory response. J Clin Invest. 1999;104:383–389.
Saubermann LJ, Nakajima A, Wada K, Zhao S, et al. Peroxisome proliferator-activated receptor gamma agonist ligands stimulate a Th2 cytokine response and prevent acute colitis. Inflamm Bowel Dis. 2002;8:330–339.
Schaefer KL, Denevich S, Ma C, Cooley SR, et al. Intestinal antiinflammatory effects of thiazolidenedione peroxisome proliferator-activated receptor-gamma ligands on T helper type 1 chemokine regulation include nontranscriptional control mechanisms. Inflamm Bowel Dis. 2005;11:244–252.
Desreumaux P, Dubuquoy L, Nutten S, Peuchmaur M, et al. Attenuation of colon inflammation through activators of the retinoid X receptor (RXR)/peroxisome proliferator-activated receptor gamma (PPARgamma) heterodimer. A basis for new therapeutic strategies. J Exp Med. 2001;193:827–838.
Volate SR, Muga SJ, Issa AY, Nitcheva D, et al. Epigenetic modulation of the retinoid X receptor alpha by green tea in the azoxymethane-Apc Min/+ mouse model of intestinal cancer. Mol Carcinog. 2009;48:920–933.
Knackstedt RW, Moseley VR, Sun S, Wargovich MJ. Vitamin D receptor and retinoid X receptor alpha status and vitamin D insufficiency in models of murine colitis. Cancer Prev Res (Phila). 2013;6:585–593.
Larriba MJ, Bonilla F, Munoz A. The transcription factors Snail1 and Snail2 repress vitamin D receptor during colon cancer progression. J Steroid Biochem Mol Biol. 2010;121:106–109.
Palmer HG, Larriba MJ, Garcia JM, Ordonez-Moran P, et al. The transcription factor SNAIL represses vitamin D receptor expression and responsiveness in human colon cancer. Nat Med. 2004;10:917–919.
Pena C, Garcia JM, Silva J, Garcia V, et al. E-cadherin and vitamin D receptor regulation by SNAIL and ZEB1 in colon cancer: clinicopathological correlations. Hum Mol Genet. 2005;14:3361–3370.
Wu Y, Deng J, Rychahou PG, Qiu S, et al. Stabilization of snail by NF-kappaB is required for inflammation-induced cell migration and invasion. Cancer Cell. 2009;15:416–428.
Barbera MJ, Puig I, Dominguez D, Julien-Grille S, et al. Regulation of Snail transcription during epithelial to mesenchymal transition of tumor cells. Oncogene. 2004;23:7345–7354.
Hotz B, Visekruna A, Buhr HJ, Hotz HG. Beyond epithelial to mesenchymal transition: a novel role for the transcription factor Snail in inflammation and wound healing. J Gastrointest Surg. 2010;14:388–397.
Thuault S, Tan EJ, Peinado H, Cano A, et al. HMGA2 and Smads co-regulate SNAIL1 expression during induction of epithelial-to-mesenchymal transition. J Biol Chem. 2008;283:33437–33446.
Thuault S, Valcourt U, Petersen M, Manfioletti G, et al. Transforming growth factor-beta employs HMGA2 to elicit epithelial-mesenchymal transition. J Cell Biol. 2006;174:175–183.
Roy HK, Smyrk TC, Koetsier J, Victor TA, et al. The transcriptional repressor SNAIL is overexpressed in human colon cancer. Dig Dis Sci. 2005;50:42–46.
Pena C, Garcia JM, Larriba MJ, Barderas R, et al. SNAI1 expression in colon cancer related with CDH1 and VDR downregulation in normal adjacent tissue. Oncogene. 2009;28:4375–4385.
Larriba MJ, Martin-Villar E, Garcia JM, Pereira F, et al. Snail2 cooperates with Snail1 in the repression of vitamin D receptor in colon cancer. Carcinogenesis. 2009;30:1459–1468.
Moolenbeek C, Ruitenberg EJ. The “Swiss roll”: a simple technique for histological studies of the rodent intestine. Lab Anim. 1981;15:57–59.
Hollis BW, Kamerud JQ, Selvaag SR, Lorenz JD, et al. Determination of vitamin D status by radioimmunoassay with an 125I-labeled tracer. Clin Chem. 1993;39:529–533.
Osburn WO, Karim B, Dolan PM, Liu G, et al. Increased colonic inflammatory injury and formation of aberrant crypt foci in Nrf2-deficient mice upon dextran sulfate treatment. Int J Cancer. 2007;121:1883–1891.
D’Ambrosio D, Cippitelli M, Cocciolo MG, Mazzeo D, et al. Inhibition of IL-12 production by 1,25-dihydroxyvitamin D3. Involvement of NF-kappaB downregulation in transcriptional repression of the p40 gene. J Clin Invest. 1998;101:252–262.
Harant H, Wolff B, Lindley IJ. 1Alpha,25-dihydroxyvitamin D3 decreases DNA binding of nuclear factor-kappaB in human fibroblasts. FEBS Lett. 1998;436:329–334.
Cohen-Lahav M, Shany S, Tobvin D, Chaimovitz C, et al. Vitamin D decreases NFkappaB activity by increasing IkappaBalpha levels. Nephrol Dial Transpl. 2006;21:889–897.
Sun J, Kong J, Duan Y, Szeto FL, et al. Increased NF-kappaB activity in fibroblasts lacking the vitamin D receptor. Am J Physiol Endocrinol Metab. 2006;291:E315–E322.
Kastner P, Messaddeq N, Mark M, Wendling O, et al. Vitamin A deficiency and mutations of RXRalpha, RXRbeta and RARalpha lead to early differentiation of embryonic ventricular cardiomyocytes. Development. 1997;124:4749–4758.
Clapper ML, Cooper HS, Chang WC. Dextran sulfate sodium-induced colitis-associated neoplasia: a promising model for the development of chemopreventive interventions. Acta Pharmacol Sin. 2007;28:1450–1459.
Kuwata T, Wang IM, Tamura T, Ponnamperuma RM, et al. Vitamin A deficiency in mice causes a systemic expansion of myeloid cells. Blood. 2000;95:3349–3356.
Wang Y, Moser AH, Shigenaga JK, Grunfeld C, et al. Downregulation of liver X receptor-alpha in mouse kidney and HK-2 proximal tubular cells by LPS and cytokines. J Lipid Res. 2005;46:2377–2387.
Beigneux AP, Moser AH, Shigenaga JK, Grunfeld C, et al. The acute phase response is associated with retinoid X receptor repression in rodent liver. J Biol Chem. 2000;275:16390–16399.
Feingold K, Kim MS, Shigenaga J, Moser A, et al. Altered expression of nuclear hormone receptors and coactivators in mouse heart during the acute-phase response. Am J Physiol Endocrinol Metab. 2004;286:E201–E207.
Pospechova K, Rozehnal V, Stejskalova L, Vrzal R, et al. Expression and activity of vitamin D receptor in the human placenta and in choriocarcinoma BeWo and JEG-3 cell lines. Mol Cell Endocrinol. 2009;299:178–187.
Palmer HG, Gonzalez-Sancho JM, Espada J, Berciano MT, et al. Vitamin D(3) promotes the differentiation of colon carcinoma cells by the induction of E-cadherin and the inhibition of beta-catenin signaling. J Cell Biol. 2001;154:369–387.
Acknowledgments
The present work benefited from the input of Elizabeth Garrett-Mayer, PhD, a statistician (Medical University of South Carolina, Department of Public Health Sciences), from Bruce Hollis, PhD and Renee Washington (Medical University of South Carolina, Department of Pediatrics) who provided systemic vitamin D quantification and from Steven Kubalak, PhD and Jayne Bernake (Medical University of South Carolina, Department of Regenerative Medicine and Cell Biology) who provided the mice for experimentation as well as genotyping. This work was supported by NIH 2RO1CA96694 from the National Cancer Institute.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Knackstedt, R., Shaoli, S., Moseley, V. et al. The Importance of the Retinoid X Receptor Alpha in Modulating Inflammatory Signaling in Acute Murine Colitis. Dig Dis Sci 59, 753–759 (2014). https://doi.org/10.1007/s10620-013-2902-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-013-2902-8