Abstract
Background
In the clinical field, increasing incidence of small intestinal ulcers associated with nonsteroidal anti-inflammatory drugs (NSAIDs) has become a topic with the advances of capsule endoscopy and balloon enteroscopy technology for the detection of small intestinal lesions. However, the pathogenesis of NSAID-induced mucosal damage, defensive mechanism of intestinal epithelial cells, and therapy for small intestinal mucosal lesion have not been fully understood. Heat shock proteins (HSPs) are involved in cytoprotection mediated by their function as a molecular chaperone. Since the function of HSP90 in the intestinal epithelial cells has not been well investigated, we examined the cytoprotective ability of HSP90-overexpressing small intestinal epithelial cells against hydrogen peroxide-induced or indomethacin-induced cell damage.
Methods
cDNA of human HSP90 gene was transfected to rat small intestinal epithelial cells (IEC-6 cells), and HSP90-overexpressing cells (IEC-6-90 cells) were selected and cloned. Anti-necrotic abilities and anti-apoptotic abilities of IEC-6-90 cells were compared with IEC-6-mock cells (transfected with vector alone). To examine the specific contribution of HSP90 on cytoprotection of IEC-6-90 cells, cytoprotective ability of IEC-6-90 cells was analyzed with or without pretreatment with functional inhibitor of HSP90, geldanamycine analog, followed by hydrogen peroxide-challenge or indomethacin-challenge.
Results
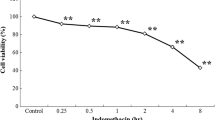
Hydrogen peroxide-induced or indomethacin-induced cell necrosis and apoptosis were significantly suppressed in IEC-6-90 cells. The cytoprotective ability of IEC-6-90 cells was suppressed by HSP90 inhibitor.
Conclusions
Our results suggest that HSP90 might play an important role in protecting small intestinal epithelial cells from hydrogen peroxide-induced or indomethacin-induced cell injury in vitro, and raised the possibility of protection of small intestinal epithelial cells by manipulation of HSP90 expression.







Similar content being viewed by others
References
Graham D, Opekun A, Willingham F, et al. Visible small-intestinal mucosal injury in chronic NSAID users. Clin Gastroenterol Hepatol. 2005;3:55–59.
Higuchi K, Umegaki E, Watanabe T, et al. Present status and strategy of NSAIDs-induced small bowel injury. J Gastroenterol. 2009;44:879–888.
Higuchi K, Yoda Y, Amagase K, et al. Prevention of NSAID-induced small intestinal mucosal injury: Prophylactic potential of lansoprazole. J Clin Biochemi Nutr. 2009;45:125–130.
Hirschowitz B. Nonsteroidal antiinflammatory drugs and the gastrointestinal tract. Gastroenterologist. 1994;2:207–223.
Sugimori S, Watanabe T, Tabuchi M, et al. Evaluation of small bowel injury in patients with rheumatoid arthritis by capsule endoscopy: effects of anti-rheumatoid arthritis drugs. Digestion. 2008;78:208–213.
Hightower L. Heat shock, stress proteins, chaperones, and proteotoxicity. Cell. 1991;66:191–197.
Tissières A, Mitchell H, Tracy U. Protein synthesis in salivary glands of Drosophila melanogaster: relation to chromosome puffs. J Mol Biol. 1974;84:389–398.
Emami A, Schwartz J, Borkan S. Transient ischemia or heat stress induces a cytoprotectant protein in rat kidney. Am J Physiol. 1991;260:F479–F485.
Li G, Li L, Liu R, et al. Heat shock protein hsp70 protects cells from thermal stress even after deletion of its ATP-binding domain. Proc Natl Acad Sci USA. 1992;89:2036–2040.
Otaka M, Odashima M, Watanabe S. Role of heat shock proteins (molecular chaperones) in intestinal mucosal protection. Biochem Biophys Res Commun. 2006;348:1–5.
Oyake J, Otaka M, Matsuhashi T, et al. Over-expression of 70-kDa heat shock protein confers protection against monochloramine-induced gastric mucosal cell injury. Life Sci. 2006;79:300–305.
Otaka M, Okuyama A, Otani S, et al. Differential induction of HSP60 and HSP72 by different stress situations in rats. Correlation with cerulein-induced pancreatitis. Dig Dis Sci. 1997;42:1473–1479.
Itoh H, Tashima Y. The stress (heat shock) proteins. Int J Biochem. 1991;23:1185–1191.
Odashima M, Otaka M, Jin M, et al. Induction of a 72-kDa heat-shock protein in cultured rat gastric mucosal cells and rat gastric mucosa by zinc L-carnosine. Dig Dis Sci. 2002;47:2799–2804.
Odashima M, Otaka M, Jin M, et al. Zinc L-carnosine protects colonic mucosal injury through induction of heat shock protein 72 and suppression of NF-kappa B activation. Life Sci. 2006;79:2245–2250.
Hirata I, Naito Y, Handa O, et al. Heat-shock protein 70-overexpressing gastric epithelial cells are resistant to indomethacin-induced apoptosis. Digestion. 2009;79:243–250.
Wada I, Otaka M, Jin M, et al. Expression of HSP72 in the gastric mucosa is regulated by gastric acid in rats-correlation of HSP72 expression with mucosal protection. Biochem Biophys Res Commun. 2006;20:611–618.
Otaka M, Odashima M, Tamaki K, et al. Expression and function of stress (heat shock) proteins in gastrointestinal tract. Int J Hyperthermia. 2009;25:634–640.
Bradford M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analyt Biochem. 1976;72:248–254.
Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA. 1979;76:4350–4354.
Ritossa F. A new puffing pattern induced by temperature shock and DNP in Drosophila. Experientia. 1962;18:571–573.
Hightower L. Cultured animal cells exposed to amino acid analogues or puromycin rapidly synthesize several polypeptides. J Cell Physiol. 1991;102:407–427.
Bukau B, Weissman J, Horwich A. Molecular chaperones and protein quality control. Cell. 2006;125:443–451.
Yamamoto S, Nakano S, Owari K, et al. Gentamicin inhibits HSP70-assisted protein folding by interfering with substrate recognition. FEBS Lett. 2010;584:645–651.
Nakamura K, Rokutan K, Marui N, et al. Induction of heat shock proteins and their implication in protection against ethanol-induced damage in cultured guinea pig gastric mucosal cells. Gastroenterology. 1991;101:161–166.
Petrof E, Chiancio M, Chang E. Role and regulation of intestinal epithelial heat shock proteins in health and disease. Chin J Dig Dis. 2004;5:45–50.
Otaka M, Odashima M, Izumi Y, et al. Target molecules of molecular chaperone (HSP70 family) in injured gastric mucosa in vivo. Life Sci. 2009;84:664–667.
Watanabe D, Otaka M, Mikami K, et al. Expression of a 72-kDa heat shock protein, and its cytoprotective function, in gastric mucosa in cirrhotic rats. J Gastroenterol. 2004;39:724–733.
Odashima M, Otaka M, Matsuhashi T, et al. Mechanical strain stress suppresses expression of HSP70 and wound restoration in gastric mucosal cells. Dig Dis Sci. 2007;52:3087–3091.
Jin M, Otaka M, Okuyama A, et al. Association of 72-kDa heat shock protein expression with adaptation to aspirin in rat gastric mucosa. Dig Dis Sci. 1999;44:1401–1407.
Young J, Moarefi I, Hartl F. Hsp90: A specialized but essential protein-folding tool. J Cell Biol. 2001;154:267–273.
Sreedhar A, Kalmár E, Csermely P, Shen Y. Hsp90 isoforms: functions, expression and clinical importance. FEBS Lett. 2004;562:11–15.
Budas G, Churchill E, Disatnik M, et al. Mitochondorial import of PKCepsilon is mediated by HSP90: a role in cardioprotection from ischaemia and reperfusion injury. Cardiovasc Res. 2010;88:83–92.
Takada M, Otaka M, Takahashi T, et al. Overexpression of a 60-kDa heat shock protein enhances cytoprotective function of small intestinal epithelial cells. Life Sci. 2010;86:499–504.
Koga F, Kihara K, Neckers L. Inhibition of cancer invasion and metastasis by targeting the molecular chaperone heat-shock protein 90. Anticancer Res. 2009;29:797–807.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tamaki, K., Otaka, M., Takada, M. et al. Evidence for Enhanced Cytoprotective Function of HSP90-Overexpressing Small Intestinal Epithelial Cells. Dig Dis Sci 56, 1954–1961 (2011). https://doi.org/10.1007/s10620-010-1558-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-010-1558-x