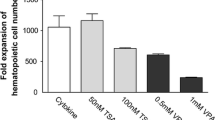
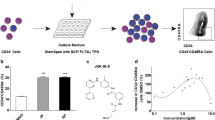
Abstract
Ex vivo expansion of hematopoietic stem cells (HSCs) is an approach for overcoming cell insufficiency for umbilical cord blood transplantation. It was suggested that in common ex vivo cultures, the stemness specificity of HSCs is rapidly reducing due to DNA hypermethylation. Here, Nicotinamide (NAM), a DNA methyltransferase and histone deacetylase inhibitor, is used with a bioengineered Bone Marrow-like niche (BLN) for HSC ex vivo expansion. The CFSE cell proliferation assay was used for tracking HSCs division. qRT-PCR was conducted to assay the HOXB4 mRNA expression levels. The morphology of BLN-cultured cells was analyzed using scanning electron microscopy (SEM). NAM boosted the induction of HSC proliferation in the BLN group compared to the control group. In addition, the ability of HSCs to colonize was more significant in the BLN group than in the control group. Our data suggest that the presence of NAM in bioengineered niches promotes HSC proliferation. The presented approach showed that small molecules could be used in the clinical setting to overcome the limited number of CD34+ cells in cord blood units.







Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BM:
-
Bone marrow
- BLN:
-
BM-like niche
- BFU-E:
-
Burst-Forming Unit-Erythroid
- CFU-GEMM:
-
Colony-Forming Unit—Granulocyte, Erythroid, Macrophage, Megakaryocyte
- CFU-GM:
-
Colony-Forming Unit—Granulocyte–Macrophage
- CFSE:
-
CarboxyFluorescein diacetate N- Succinilamidyle Ester
- DBM:
-
Demineralized Bone Matrix
- HSC:
-
Hematopoietic Stem Cell
- HDI:
-
Histone Deacetylase Inhibitors
- MSC:
-
Mesenchymal Stem cell
- NAM:
-
Nicotinamide
- NAM-CY:
-
Nicotinamide, and cytokines
- SEM:
-
Scanning Electron Microscopy
- TCC:
-
Traditional culture condition
- 2D:
-
2-Dimensional
- 3D:
-
3-Dimensional
References
Araki H, Baluchamy S, Yoshinaga K, Petro B, Petiwala S, Parajuli R et al (2009) Cord blood stem cell expansion is permissive to epigenetic regulation and environmental cues. Exp Hematol 37:1084–1095
Asri A, Sabour J, Atashi A, Soleimani M (2016) Homing in hematopoietic stem cells: focus on regulatory role of CXCR7 on SDF1a/CXCR4 axis. EXCLI J 15:134–143
Ballen KK, Lazarus H (2016) Cord blood transplant for acute myeloid leukaemia. Br J Haematol 173:25–36
Barcena-Varela M, Colyn L, Fernandez-Barrena MG (2019) Epigenetic mechanisms in hepatic stellate cell activation during liver fibrosis and carcinogenesis. Int J Mol Sci 20:2507
Barker JN, Wagner JE (2003) Umbilical-cord blood transplantation for the treatment of cancer. Nat Rev Cancer 3:526–532
Bhatlekar S, Fields JZ, Boman BM (2018) Role of HOX genes in stem cell differentiation and cancer. Stem Cells Int 2018:3569493
Boitano AE, Wang J, Romeo R, Bouchez LC, Parker AE, Sutton SE et al (2010) Aryl hydrocarbon receptor antagonists promote the expansion of human hematopoietic stem cells. Science 329:1345–1348
Bourgine PE, Klein T, Paczulla AM, Shimizu T, Kunz L, Kokkaliaris KD et al (2018) In vitro biomimetic engineering of a human hematopoietic niche with functional properties. Proc Natl Acad Sci USA 115:E5688–E5695
Bug G, Gül H, Schwarz K, Pfeifer H, Kampfmann M, Zheng X et al (2005) Valproic acid stimulates proliferation and self-renewal of hematopoietic stem cells. Cancer Res 65:2537–2541
Calkoen FGJ, Vervat C, van Pel M, de Haas V, Vijfhuizen LS, Eising E et al (2015) Despite differential gene expression profiles pediatric MDS derived mesenchymal stromal cells display functionality in vitro. Stem Cell Res 14:198–210
Cantero Peral S, Burkhart HM, Oommen S, Yamada S, Nyberg SL, Li X et al (2015) Safety and feasibility for pediatric cardiac regeneration using epicardial delivery of autologous umbilical cord blood-derived mononuclear cells established in a porcine model system. Stem Cells Transl Med 4:195–206
Chang Y, Hsieh P-H, Chao CC-K (2009) The efficiency of Percoll and Ficoll density gradient media in the isolation of marrow derived human mesenchymal stem cells with osteogenic potential. Chang Gung Med J 32:264–275
Chaurasia P, Gajzer DC, Schaniel C, Souza S, Hoffman R (2014) Epigenetic reprogramming induces the expansion of cord blood stem cells. J Clin Investig 124:2378–2395
Chotinantakul K, Leeanansaksiri W (2012) Hematopoietic stem cell development, niches, and signaling pathways. Bone Marrow Res 2012:270425
Crowley LC, Marfell BJ, Scott AP, Waterhouse NJ (2016) Quantitation of apoptosis and necrosis by annexin V binding, propidium iodide uptake, and flow cytometry. Cold Spring Harb Protoc. https://doi.org/10.1101/pdb.prot087288
Eckschlager T, Plch J, Stiborova M, Hrabeta J (2017) Histone deacetylase inhibitors as anticancer drugs. Int J Mol Sci 18:1414
Hashimoto Y, Funamoto S, Kimura T, Nam K, Fujisato T, Kishida A (2011) The effect of decellularized bone/bone marrow produced by high-hydrostatic pressurization on the osteogenic differentiation of mesenchymal stem cells. Biomaterials 32:7060–7067
Houshmand M, Soleimani M, Atashi A, Saglio G, Abdollahi M, Zarif MN (2017) Mimicking the acute myeloid leukemia niche for molecular study and drug screening. Tissue Eng Part C Methods 23:72–85
Hwang ES, Song SB (2017) Nicotinamide is an inhibitor of SIRT1 in vitro, but can be a stimulator in cells. Cell Mol Life Sci 74:3347–3362
Ingavle G, Vaidya A, Kale V (2019) Constructing three-dimensional microenvironments using engineered biomaterials for hematopoietic stem cell expansion. Tissue Eng Part B Rev 25:312–329
Kelly SS, Sola CBS, de Lima M, Shpall E (2009) Ex vivo expansion of cord blood. Bone Marrow Transpl 44:673–681. https://doi.org/10.1038/bmt.2009.284
Kojabad AA, Ghaleh HEG, Shahriary A, Farzanehpour M (2022) Human hematopoietic stem cells co-cultured in 3D with stromal support to optimize lentiviral vector-mediated gene transduction. Indian J Hematol Blood Transfus. https://doi.org/10.1007/s12288-022-01576-4
Kramer A, Challen GA (2017) The epigenetic basis of hematopoietic stem cell aging. Semin Hematol 54:19–24
Lee H, Bao S, Qian Y, Geula S, Leslie J, Zhang C et al (2019) Stage-specific requirement for Mettl3-dependent m(6)A mRNA methylation during haematopoietic stem cell differentiation. Nat Cell Biol 21:700–709
Nielsen LK, Kelly SS, Sola CBS, De Lima M, Shpall EJ, Sieber S et al (2009) Making sense of hematopoietic stem cell niches. Blood 5:1–12
Nikas IP, Paschou SA, Ryu HS (2020) The role of nicotinamide in cancer chemoprevention and therapy. Biomolecules 10:477
Norkin M, Lazarus HM, Wingard JR (2013) Umbilical cord blood graft enhancement strategies: has the time come to move these into the clinic[quest]. Bone Marrow Transpl 48:884–889
Peled T, Shoham H, Aschengrau D, Yackoubov D, Frei G, Rosenheimer GN et al (2012) Nicotinamide, a SIRT1 inhibitor, inhibits differentiation and facilitates expansion of hematopoietic progenitor cells with enhanced bone marrow homing and engraftment. Exp Hematol 40:342–355
Pineault N, Abu-Khader A (2015) Advances in umbilical cord blood stem cell expansion and clinical translation. Exp Hematol 43:498–513
Pinho S, Frenette PS (2019) Haematopoietic stem cell activity and interactions with the niche. Nat Rev Mol Cell Biol 20:303
Santana BP, Nedel F, Perelló Ferrúa C, Marques e Silva R, da Silva AF, Demarco FF et al (2015) Comparing different methods to fix and to dehydrate cells on alginate hydrogel scaffolds using scanning electron microscopy. Microsc Res Tech 78:553–561
Sauvageau G, Thorsteinsdottir U, Eaves CJ, Lawrence HJ, Largman C, Lansdorp PM et al (1995) Overexpression of HOXB4 in hematopoietic cells causes the selective expansion of more primitive populations in vitro and in vivo. Genes Dev 9:1753–1765
Schuster JA, Stupnikov MR, Ma G, Liao W, Lai R, Ma Y et al (2012) Expansion of hematopoietic stem cells for transplantation: current perspectives. Exp Hematol Oncol 1:12
Seita J, Weissman IL (2010) Hematopoietic stem cell: self-renewal versus differentiation. Wiley Interdiscip Rev Syst Biol Med 2:640–653
Shah Z, Filonenko E, Ramensky V, Fan C, Wang C, Sonin A et al (2019) MYB is an essential regulator of primitive human hematopoiesis in pluripotent stem cell differentiation cultures. SSRN Electron J. https://doi.org/10.2139/ssrn.3406382
Sharma MB, Limaye LS, Kale VP (2012) Mimicking the functional hematopoietic stem cell niche in vitro: recapitulation of marrow physiology by hydrogel-based three-dimensional cultures of mesenchymal stromal cells. Haematologica 97:651
Stanevsky A, Goldstein G, Nagler A (2009) Umbilical cord blood transplantation: pros, cons and beyond. Blood Rev 23:199–204
Tajer P, Pike-Overzet K, Arias S, Havenga M, Staal F (2019) Ex vivo expansion of hematopoietic stem cells for therapeutic purposes: lessons from development and the niche. Cells 8:169
Takizawa H, Schanz U, Manz MG (2011) Ex vivo expansion of hematopoietic stem cells: mission accomplished? Swiss Med Wkly 141:w13316
Tatetsu H, Armant M, Wang F, Gao C, Ueno S, Tian X et al (2019) Maintenance and enhancement of human peripheral blood mobilized stem/progenitor cell engraftment after ex vivo culture via an HDACi/SALL4 axis (3465). Exp Hematol 75:53–63
Torisawa Y, Spina CS, Mammoto T, Mammoto A, Weaver JC, Tat T et al (2014) Bone marrow–on–a–chip replicates hematopoietic niche physiology in vitro. Nat Methods 11:663–669. https://doi.org/10.1038/nmeth.2938
Tothova Z, Gilliland DG (2007) FoxO transcription factors and stem cell homeostasis: insights from the hematopoietic system. Cell Stem Cell 1:140–152
Weidner CI, Walenda T, Lin Q, Wölfler MM, Denecke B, Costa IG et al (2013) Hematopoietic stem and progenitor cells acquire distinct DNA-hypermethylation during in vitro culture. Sci Rep 3:3372. https://doi.org/10.1038/srep03372
Xia Y, Whitesides GM (1998) Soft Lithography. Annu Rev Mater Sci 28:153–184. https://doi.org/10.1146/annurev.matsci.28.1.153
Funding
This study was supported by High Institute for Research and Education in Transfusion Medicine, Tehran, Iran.
Author information
Authors and Affiliations
Contributions
MHAA, MS and MNZ wrote the main manuscript text and AAK prepared figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Askari, M.H.A., Shahabi, M., Kojabad, A.A. et al. Reconstruction of bone marrow microenvironment for expansion of hematopoietic stem cells by a histone deacetylase inhibitor. Cytotechnology 75, 195–206 (2023). https://doi.org/10.1007/s10616-022-00564-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-022-00564-w