Abstract
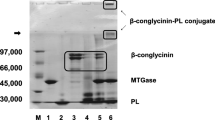
β-lactoglobulin (BLG) and ε-polylysine (PL) were bound by using microbial transglutaminase. Dextran (Dex) was further conjugated to the BLG-PL conjugate by the Maillard reaction. Confirmation of conjugation was carried out by SDS-PAGE. From the results of isoelectric focusing, it was revealed that the isoelectric point of the BLG-PL conjugate was shifted to the basic side as compared with native BLG. Immunogenicity of BLG in BALB/c mice was lowered by conjugation with PL and further lowered by conjugation with Dex. By conjugation with PL and PL-Dex, antibacterial activity against Staphylococcus aureus was endowed to BLG. Because the conjugation method in this study is a safe method, it is valuable in that it can be applicable to food processing.





Similar content being viewed by others
Data availability
The data underlying this article are available in the article and also from the corresponding author upon request.
Abbreviations
- BLG:
-
β-Lactoglobulin
- PL:
-
Epsilon polylysine
- SDS-PAGE:
-
Sodium dodecyl sulfate poly acrylamide gel electrophoresis
- CBB:
-
Coomassie brilliant blue
- IEF:
-
Isoelectric focusing
References
Armstrong JM, McKenzie HA, Sawyer WH (1967) On the fractionation of beta-lactoglobulin and alpha-lactalbumin. Biochim Biophys Acta 147:60–72. https://doi.org/10.1016/0005-2795(67)90090-6
Chen FM, Lee JH, Yang YH, Lin YT, Wang LC, Yu HH, Chiang BL (2014) Analysis of α-lactalbumin-, β-lactoglobulin-, and casein-specific IgE among children with atopic diseases in a tertiary medical center in northern Taiwan. J Microbiol Immunol Infect 47:130–136. https://doi.org/10.1016/j.jmii.2012.08.009
Foegeding EA, Kuhn PR, Hardin CC (1992) Specific divalent cation-induced changes during gelation of β-lactoglobulin. J Agric Food Chem 40:2092–2097. https://doi.org/10.1021/jf00023a011
Hattori M (2002) Functional Improvements in Food Proteins in multiple aspects by conjugation with saccharides: case studies of β-lactoglobulin-acidic polysaccharides conjugates. Food Sci Technol Res 8:291–299. https://doi.org/10.3136/fstr.8.291
Kramlová M, Přistoupil TI, Fričová V, Kraml J (1986) First experience with the use of the Pharmacia PhastSystem for the characterization of hemoglobulins by isoelectric focusing. J Chromatogr A 367:443–445. https://doi.org/10.1016/S0021-9673(00)94868-4
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
McKenzie HA (1971) Milk proteins. Academic Press, New York, pp 257–330
Phillips LG, Hawks SE, German JB (1995) Structural characteristics and foaming properties of β-lactoglobulin: effects of shear rate and temperature. J Agric Food Chem 43:613–619. https://doi.org/10.1021/jf00051a011
Sawyer L, Kontopidis G (2000) The core lipocalin, bovine β-lactoglobulin. Biochim Biophys Acta 1482:136–138. https://doi.org/10.1016/S0167-4838(00)00160-6
Shima S, Matsuoka H, Iwamoto T, Sakai H (1984) Antimicrobial action of epsilon-poly-L-lysine. J Antibiot 37:1449–1455. https://doi.org/10.7164/antibiotics.37.1449
Shimizu M, Saito M, Yamauchi K (1985) Emulsifying and structural properties of β-lactoglobulin at different pHs. Agric Biol Chem 49:189–194. https://doi.org/10.1080/00021369.1985.10866680
Virtanen T, Zeiler T, Rsutinainen J, Mäntyjärvi R (1999) Allergy to lipocalins: a consequence of misguided T-cell recognition of self and nonself? Immunol Today 20:398–400. https://doi.org/10.1016/S0167-5699(99)01515-7
Yoshida T, Tanemura M, Shimizu A, TanakaKurokawa HS, Takahashi K, Hattori M (2022) Functional improvements in β-lactoglobulin by preparing edible conjugate with microbial transglutaminase. Biosci Biotechnol Biochem 86:390–396. https://doi.org/10.1093/bbb/zbab220
Acknowledgements
We thank CHISSO CORPORATION (Tokyo, Japan) for presenting ε-polylysine. We thank Ajinomoto Co. (Tokyo,Japan) for presenting Activa TG-K (E.C.2.3.2.13). This work was supported in part by JSPS KAKENHI Grant Number JP22580126.
Author information
Authors and Affiliations
Contributions
TY: Conceptualization, Data curation, Writing manuscript, Funding acquisition, SK: Conceptualization, Data curation, Writing manuscript, RI, KH, YM: Data curation, Writing manuscript, MH: Conceptualization, Writing manuscript, Funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoshida, T., Ikeda, R., Kawai, S. et al. Reduced immunogenicity and endowed antimicrobial activity in β-lactoglobulin by preparing edible bioconjugate. Cytotechnology 75, 39–47 (2023). https://doi.org/10.1007/s10616-022-00558-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-022-00558-8