Abstract
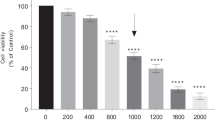
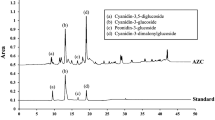
Parkinson’s disease (PD) is a common chronic neurodegenerative disease induced by the death of dopaminergic neurons. Anthocyanins are naturally found antioxidants and well-known for their preventive effects in neurodegenerative disorders. Black carrots (Daucus carota L. ssp. sativus var. atrorubens Alef.) are a rich source of anthocyanins predominantly including acylated cyanidin-based derivatives making them more stable. However, there have been no reports analysing the neuroprotective role of black carrot anthocyanins (BCA) on PD. In order to investigate the potential neuroprotective effect of BCA, human SH-SY5Y cells were treated with MPP+ (1-methyl-4-phenylpyridinium) to induce PD associated cell death and cytotoxicity. Anthocyanins were extracted from black carrots and the composition was determined by HPLC–DAD. SH-SY5Y cells were co-incubated with BCA (2.5, 5, 10, 25, 50, 100 µg/ml) and 0.5 mM MPP+ to determine the neuroprotective effect of BCA against MPP+ induced cell death and cytotoxicity. Results indicate that BCA concentrations did not have any adverse effect on cell viability. BCA revealed its cytoprotective effect, especially at higher concentrations (50, 100 µg/ml) by increasing metabolic activity and decreasing membrane damage. BCA exhibited antioxidant activity via scavenging MPP+ induced reactive oxygen species (ROS) and protecting dopaminergic neurons from ROS mediated apoptosis. These results suggest a neuroprotective effect of BCA due to its high antioxidant and antiapoptotic activity, along with the absence of cytotoxicity. The elevated stability of BCA together with potential neuroprotective effects may shed light to future studies in order to elucidate the mechanism and further neuro-therapeutic potential of BCA which is promising as a neuroprotective agent.





Similar content being viewed by others
Availability of data and materials
All data generated and/or analyzed during this study are available upon request from the corresponding author.
References
Ahmad T et al (2019) Phytochemicals in Daucus carota and their health benefits-review. Foods 8:424. https://doi.org/10.3390/foods8090424
Akhtar S, Rauf A, Imran M, Saleem Q, Riaz M, Mubarak MS (2017) Black carrot (Daucus carota L.), dietary and health promoting perspectives of its polyphenols: a review. Trends Food Sci Technol 66:36–47
Algarra M, Fernandes A, Mateus N, Freitas V, Joaquim CG, da Silva E, Casado J (2014) Anthocyanin profile and antioxidant capacity of black carrots (Daucus carota L. ssp. sativus var. atrorubens Alef.) from Cuevas Bajas, Spain. J Food Compos Anal 33:71–76
Apak R, Güçlü K, Özyürek M, Karademir SE, Erçağ E (2006) The cupric ion reducing antioxidant capacity and polyphenolic content of some herbal teas. Int J Food Sci Nutr 57:292–304
Apak R, Güçlü K, Özyürek M, Çelik ES (2008) Mechanism of antioxidant capacity assays and the CUPRAC (cupric ion reducing antioxidant capacity) assay. Microchim Acta 160:413–419
Barba-Espin G et al (2017) Foliar-applied ethephon enhances the content of anthocyanin of black carrot roots (Daucus carota ssp. sativus var. atrorubens Alef.). BMC Plant Biol 17:70. https://doi.org/10.1186/s12870-017-1021-7
Benassi B et al (2016) Extremely low frequency magnetic field (ELF-MF) exposure sensitizes SH-SY5Y cells to the pro-Parkinson’s disease toxin MPP(.). Mol Neurobiol 53:4247–4260. https://doi.org/10.1007/s12035-015-9354-4
Cassarino DS et al (1997) Elevated reactive oxygen species and antioxidant enzyme activities in animal and cellular models of Parkinson’s disease. Biochim Biophys Acta 1362:77–86. https://doi.org/10.1016/s0925-4439(97)00070-7
Chen J, Sun J, Jiang J, Zhou J (2018) Cyanidin protects SH-SY5Y human neuroblastoma cells from 1-methyl-4-phenylpyridinium-induced neurotoxicity. Pharmacology 102:126–132. https://doi.org/10.1159/000489853
Cheung YT, Lau WK, Yu MS, Lai CS, Yeung SC, So KF, Chang RC (2009) Effects of all-trans-retinoic acid on human SH-SY5Y neuroblastoma as in vitro model in neurotoxicity research. Neurotoxicology 30:127–135. https://doi.org/10.1016/j.neuro.2008.11.001
Dagda RK, Das Banerjee T, Janda E (2013) How Parkinsonian toxins dysregulate the autophagy machinery. Int J Mol Sci 14:22163–22189. https://doi.org/10.3390/ijms141122163
Espinosa-Acosta G et al (2018) Stability analysis of anthocyanins using alcoholic extracts from black carrot (Daucus carota ssp. sativus Var. atrorubens Alef.). Molecules 23:1. https://doi.org/10.3390/molecules23112744
Fall CP, Bennett JP Jr (1999) Characterization and time course of MPP+-induced apoptosis in human SH-SY5Y neuroblastoma cells. J Neurosci Res 55:620–628
Gao X, Cassidy A, Schwarzschild MA, Rimm EB, Ascherio A (2012) Habitual intake of dietary flavonoids and risk of Parkinson disease. Neurology 78:1138–1145. https://doi.org/10.1212/WNL.0b013e31824f7fc4
Hu Q, Wang G (2016) Mitochondrial dysfunction in Parkinson’s disease. Transl Neurodegener 5:14. https://doi.org/10.1186/s40035-016-0060-6
Hung KC, Huang HJ, Lin MW, Lei YP, Lin AM (2014) Roles of autophagy in MPP+-induced neurotoxicity in vivo: the involvement of mitochondria and alpha-synuclein aggregation. PLoS ONE 9:e91074. https://doi.org/10.1371/journal.pone.0091074
Jiang X, Jin T, Zhang H, Miao J, Zhao X, Su Y, Zhang Y (2019) Current progress of mitochondrial quality control pathways underlying the pathogenesis of Parkinson’s disease. Oxid Med Cell Longev 2019:4578462. https://doi.org/10.1155/2019/4578462
Jing P, Bomser JA, Schwartz SJ, He J, Magnuson BA, Giusti MM (2008) Structure-function relationships of anthocyanins from various anthocyanin-rich extracts on the inhibition of colon cancer cell growth. J Agric Food Chem 56:9391–9398. https://doi.org/10.1021/jf8005917
Khoo HE, Azlan A, Tang ST, Lim SM (2017) Anthocyanidins and anthocyanins: colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr Res 61:1361779. https://doi.org/10.1080/16546628.2017.1361779
Kim HG et al (2010) Mulberry fruit protects dopaminergic neurons in toxin-induced Parkinson’s disease models. Br J Nutr 104:8–16. https://doi.org/10.1017/S0007114510000218
Kim MJ et al (2015) Transduced PEP-1-PON1 proteins regulate microglial activation and dopaminergic neuronal death in a Parkinson’s disease model. Biomaterials 64:45–56. https://doi.org/10.1016/j.biomaterials.2015.06.015
Li D, Wang P, Luo Y, Zhao M, Chen F (2017) Health benefits of anthocyanins and molecular mechanisms: update from recent decade. Crit Rev Food Sci Nutr 57:1729–1741. https://doi.org/10.1080/10408398.2015.1030064
Lin MT, Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443:787–795. https://doi.org/10.1038/nature05292
Meng L et al (2018) Anthocyanins extracted from Aronia melanocarpa protect SH-SY5Y cells against amyloid-beta (1–42)-induced apoptosis by regulating Ca(2+) homeostasis and inhibiting mitochondrial dysfunction. J Agric Food Chem 66:12967–12977. https://doi.org/10.1021/acs.jafc.8b05404
Montilla EC, Arzaba MR, Hillebrand S, Winterhalter P (2011) Anthocyanin composition of black carrot (Daucus carota ssp. sativus var. atrorubens Alef.) cultivars Antonina, Beta Sweet, Deep Purple, and Purple Haze. J Agric Food Chem 59:3385–3390. https://doi.org/10.1021/jf104724k
Moriya C et al (2015) New acylated anthocyanins from purple yam and their antioxidant activity. Biosci Biotechnol Biochem 79:1484–1492. https://doi.org/10.1080/09168451.2015.1027652
Netzel MNG, Kammerer DR, Schieber A, Carle R, Simons LBI, Bitsch R, Konczak I (2007) Cancer cell antiproliferation activity and metabolism of black carrot anthocyanins. Innov Food Sci Emerg Technol 8:365–372
Rababah TM, Ereifej KI, Esoh RB, Al-u’datt MH, Alrababah MA, Yang W (2011) Antioxidant activities, total phenolics and HPLC analyses of the phenolic compounds of extracts from common Mediterranean plants. Nat Prod Res 25:596–605. https://doi.org/10.1080/14786419.2010.488232
Rabiei Z, Solati K, Amini-Khoei H (2019) Phytotherapy in treatment of Parkinson’s disease: a review. Pharm Biol 57:355–362. https://doi.org/10.1080/13880209.2019.1618344
Ramalingam M, Kim SJ (2016) The neuroprotective role of insulin against MPP(+)-induced Parkinson’s disease in differentiated SH-SY5Y cells. J Cell Biochem 117:917–926. https://doi.org/10.1002/jcb.25376
Sevimli-Gur C, Cetin B, Akay S, Gulce-Iz S, Yesil-Celiktas O (2013) Extracts from black carrot tissue culture as potent anticancer agents. Plant Foods Hum Nutr 68:293–298. https://doi.org/10.1007/s11130-013-0371-z
Shishido T et al (2019) Synphilin-1 has neuroprotective effects on MPP(+)-induced Parkinson’s disease model cells by inhibiting ROS production and apoptosis. Neurosci Lett 690:145–150. https://doi.org/10.1016/j.neulet.2018.10.020
Smeriglio A, Denaro M, Barreca D, D’Angelo V, Germano MP, Trombetta D (2018) Polyphenolic profile and biological activities of black carrot crude extract (Daucus carota L. ssp sativus var. atrorubens Alef.). Fitoterapia 124:49–57. https://doi.org/10.1016/j.fitote.2017.10.006
Song FL, Gan RY, Zhang Y, Xiao Q, Kuang L, Li HB (2010) Total phenolic contents and antioxidant capacities of selected chinese medicinal plants. Int J Mol Sci 11:2362–2372. https://doi.org/10.3390/ijms11062362
Strathearn KE et al (2014) Neuroprotective effects of anthocyanin- and proanthocyanidin-rich extracts in cellular models of Parkinsons disease. Brain Res 1555:60–77. https://doi.org/10.1016/j.brainres.2014.01.047
Torma PD et al (2017) Hydroethanolic extracts from different genotypes of acai (Euterpe oleracea) presented antioxidant potential and protected human neuron-like cells (SH-SY5Y). Food Chem 222:94–104. https://doi.org/10.1016/j.foodchem.2016.12.006
Tsutsumi A et al (2019) Acylated anthocyanins derived from purple carrot (Daucus carota L.) induce elevation of blood flow in rat cremaster arteriole. Food Funct 10:1726–1735. https://doi.org/10.1039/c8fo02125b
Vagiri M, Ekholm A, Andersson SC, Johansson E, Rumpunen K (2012) An optimized method for analysis of phenolic compounds in buds, leaves, and fruits of black currant (Ribes nigrum L.). J Agric Food Chem 60:10501–10510. https://doi.org/10.1021/jf303398z
Vargas CG et al (2018) Bioactive compounds and protective effect of red and black rice brans extracts in human neuron-like cells (SH-SY5Y). Food Res Int 113:57–64. https://doi.org/10.1016/j.foodres.2018.06.069
Winter AN, Bickford PC (2019) Anthocyanins and their metabolites as therapeutic agents for neurodegenerative disease. Antioxidants. https://doi.org/10.3390/antiox8090333
Wright OR, Netzel GA, Sakzewski AR (2013) A randomized, double-blind, placebo-controlled trial of the effect of dried purple carrot on body mass, lipids, blood pressure, body composition, and inflammatory markers in overweight and obese adults: the QUENCH trial. Can J Physiol Pharmacol 91:480–488. https://doi.org/10.1139/cjpp-2012-0349
Xie HR, Hu LS, Li GY (2010) SH-SY5Y human neuroblastoma cell line: in vitro cell model of dopaminergic neurons in Parkinson’s disease. Chin Med J 123:1086–1092
Yousuf B, Gul K, Wani AA, Singh P (2016) Health benefits of anthocyanins and their encapsulation for potential use in food systems: a review. Crit Rev Food Sci Nutr 56:2223–2230. https://doi.org/10.1080/10408398.2013.805316
Zeng G et al (2010) Salvianolic acid B protects SH-SY5Y neuroblastoma cells from 1-methyl-4-phenylpyridinium-induced apoptosis. Biol Pharm Bull 33:1337–1342. https://doi.org/10.1248/bpb.33.1337
Zeng XS, Geng WS, Jia JJ, Chen L, Zhang PP (2018) Cellular and molecular basis of neurodegeneration in Parkinson disease. Front Aging Neurosci 10:109. https://doi.org/10.3389/fnagi.2018.00109
Zhang J et al (2019) Neuroprotective effects of anthocyanins and its major component cyanidin-3-O-glucoside (C3G) in the central nervous system: an outlined review. Eur J Pharmacol 858:172500. https://doi.org/10.1016/j.ejphar.2019.172500
Acknowledgements
We thank Cihan Altintas and Cagin Cevik for technical support.
Funding
This work was supported by a grant from Nisantasi University Scientific Research Committee (Project No: BAP00017).
Author information
Authors and Affiliations
Contributions
MZ designed and performed the experiments, analyzed the results, wrote the paper, and approved the final version; AM analyzed the data, revised the manuscript and approved the final version; IK revised the manuscript and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests regarding the publication of this paper.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10616_2021_500_MOESM1_ESM.tif
Supplementary file1 (TIF 96328 kb) Supplementary Fig. 1 HPLC chromatogram and calibration curve of cyanidin-3-O-glucoside. a HPLC chromatogram of cyanidin-3-O-glucoside standard monitored at 520 nm detection. b Calibration curve of cyanidin-3-glucoside standard.
Rights and permissions
About this article
Cite this article
Zaim, M., Kara, I. & Muduroglu, A. Black carrot anthocyanins exhibit neuroprotective effects against MPP+ induced cell death and cytotoxicity via inhibition of oxidative stress mediated apoptosis. Cytotechnology 73, 827–840 (2021). https://doi.org/10.1007/s10616-021-00500-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-021-00500-4