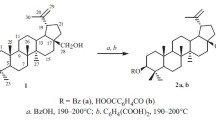
Synthetic methods for betulin 3-acetate-28-benzoate, betulin 3-acetate-28-p-bromobenzoate, betulin 3-acetate-28-cinnamate, betulin 3-acetate-28-succinate, and betulin 3-caetate-28-phthalate based on the reaction of betulin 3-acetate with fused benzoic, p-bromobenzoic, cinnamic, succinic, and phthalic acids at 185–195°C for 5–6 min are proposed for the first time.
Similar content being viewed by others
References
V. V. Grishko, I. A. Tolmacheva, and A. V. Pereslavtseva, Chem. Nat. Compd., 51, 1 (2015).
S. C. Jonnalagadda, P. Suman, D. C. Morgan, and J. N. Seay, Stud. Nat. Prod. Chem., 53, 45 (2017).
U. Bildziukevich, Z. Ozdemir, and Z. Wimmer, Molecules, 24 (19), 3546 (2019).
S. Amiri, S. Dastghaib, M. Ahmadi, P. Mehrbod, F. Khadem, H. Behrouj, M.-R. Aghanoori, F. Machaj, M. Ghamsari, J. Rosik, A. Hudecki, A. Afkhami, M. Hashemi, M. J. Los, P. Mokarram, T. Madrakian, and S. Ghavami, Biotechnol. Adv., 38, 107409 (2020).
P. A. Krasutsky, R. M. Carlson, and R. Karim, US Pat. No. 6,642,217, Nov. 4, 2003.
O. B. Flekhter, L. T. Karachurina, V. V. Poroikov, L. P. Nigmatullina, L. A. Baltina, F. S. Zarudii, V. A. Davydova,
L. V. Spirikhin, I. P. Baikova, F. Z. Galin, and G. A. Tolstikov, Russ. J. Bioorg. Chem., 26 (3), 192 (2000).
7. O. B. Flekhter, N. I. Medvedeva, L. T. Karachurina, L. A. Baltina, F. Z. Galin, F. S. Zarudii, and G. A. Tolstikov, Pharm. Chem. J., 39 (8), 401 (2005).
8. L. Pohjala, S. Alakurtti, T. Ahola, J. Yli-Kauhaluoma, and P. Tammela, J. Nat. Prod., 72 (11), 1917 (2009).
9. V. A. Levdansky, A. A. Kondrasenko, A. V. Levdansky, and B. N. Kuznetsov, Chem. Nat. Compd., 56, 951 (2020).
10. V. A. Levdansky, A. V. Levdansky, and B. N. Kuznetsov, J. Sib. Fed. Univ.: Chem., 5 (3), 274 (2012).
Acknowledgment
The research was supported by Russian Science Foundation Grant No. 23-23-00336; https://rscf.ru/project/23-23-00336/. The work used equipment at the Krasnoyarsk Regional Center for Common Use of FRC, KSC, SB, RAS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2023, pp. 727–729
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Levdansky, A.V., Garyntseva, N.V. & Levdansky, V.A. Esterification of Betulin 3-Acetate in Melts of Some Organic Acids. Chem Nat Compd 59, 862–864 (2023). https://doi.org/10.1007/s10600-023-04136-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-04136-y