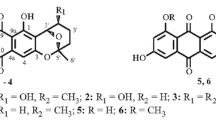
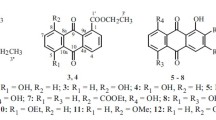
Two new anthraquinones, (11S)-1,4,6-trihydroxy-7-(1-hydroxyethyl)-3-methoxyanthracene-9,10-dione (1) and (11S)-1,6-dihydroxy-7-(1-hydroxyethyl)-3-methoxyanthracene-9,10-dione (2), together with six known compounds, including four naphthoquinones (3–6), one thiophene derivative (7), and one cyclohexanedione derivative (8), were isolated from the mangrove rhizosphere soil-derived fungus Fusarium sp. J3-2. Compound 1 displayed weak to moderate antibacterial activities against five strains of pathogenic bacteria with MIC values ranging from 25 to 50 μg/mL. Compound 3 showed weak cytotoxic activities toward human hepatocellular carcinoma cell lines, SMMC-7721 and SGC-7901, with IC50 values of 48.87 and 41.23 μg/mL, respectively. Compounds 1–7 exhibited antifouling activities against the larval settlement of the barnacle Balanus amphitrite.

Similar content being viewed by others
References
B. Haefner, Drug Discov. Today, 8, 536 (2003).
J. Xu, RSC Adv., 5, 841 (2014).
E. Ancheeva, G. Daletos, and P. Proksch, Mar. Drugs, 16, 319 (2018).
A. R. Carroll, B. R. Copp, R. A. Davis, R. A. Keyzers, and M. R. Prinsep, Nat. Prod. Rep., 37, 175 (2020).
M. Chen, Y. Y. Zheng, Z. Q. Chen, N. X. Shen, L. Shen, F. M. Zhang, X. J. Zhou, and C. Y. Wang, J. Nat. Prod., 82, 368 (2019).
Z. Y. Liang, N. X. Shen, Y. Y. Zheng, J. T. Wu, L. Miao, X. M. Fu, M. Chen, and C. Y. Wang, Bioorg. Chem., 93, 103331 (2019).
Y. Y. Zheng, X. Chen, L. M. Chen, L. Shen, X. M. Fu, Q. X. Chen, M. Chen, and C. Y. Wang, J. Ocean U. China, 19, 700 (2020).
Y. Y. Zheng, N. X. Shen, Z. Y. Liang, L. Shen, M. Chen, and C. Y. Wang, Nat. Prod. Res., 34, 378 (2020).
D. L. Zhao, D. Wang, X. Y. Tian, F. Cao, Y. Q. Li, and C. S. Zhang, Mar. Drugs, 16, 36 (2018).
F. A. Nagia and R. El-Mohamedy, Dyes Pigments, 75, 550 (2007).
A. Alvin, J. A. Kalaitzis, B. Sasia, and B. A. Neilan, J. Appl. Microbiol., 120, 1229 (2016).
J. Hashimoto, K. Motohashi, K. Sakamoto, S. Hashimoto, M. Yamanouchi, H. Tanaka, T. Takahashi, M. Takagi, and K. Shin-Ya, J. Antibiot., 62, 625 (2009).
Y. Kimura., A. Shimada, H. Nakajima, and T. Hamasaki, Agric. Biol. Chem., 52, 1253 (1988).
Y. Kimura, T. Hamasaki, and H. Nakajima, Agric. Biol. Chem., 45, 2653 (1981).
C. M. Deng, S. X. Liu, C. H. Huang, J. Y. Pang, and Y. C. Lin, Mar. Drugs, 11, 2616 (2010).
S. T. Tan, A. L. Wilkins, P. T. Holland, and T. K. McGhie, J. Agric. Food Chem., 37, 1217 (1989).
C. G. Pierce, P. Uppuluri, A. R. Tristan, F. L. Wormley Jr, E. Mowat, G. Ramage, and J. L. Lopez-Ribot, Nat. Protoc., 3, 1494 (2008).
T. J. Mosmann, J. Immunol. Meth., 65, 55 (1983).
V. Thiyagarajan, T. Harder, J. W. Qiu, and P. Y. Qian, Mar. Biol. (Berlin), 143, 543 (2003).
Acknowledgment
The National Natural Science Foundation of China (Nos. 81703411, 42177459, and 41776156) supported this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 2, March–April, 2023, pp. 202–205.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, M., Wu, JT., Hao, BC. et al. Two New Anthraquinones from the Marine-Derived Fungus Fusarium sp. J3-2. Chem Nat Compd 59, 237–241 (2023). https://doi.org/10.1007/s10600-023-03965-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-03965-1