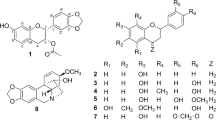
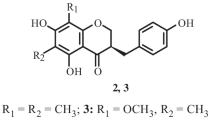
Two new flavonoids, 8,2′-diprenyl-3,5,7,5′-tetrahydroxyflavone (1) and 8-(1-methoxy-2,3-dihydroxy-3-methylbutyl)naringenin (2), along with twelve known compounds (3–14), were isolated from the leaves of Macaranga pustulata. The structures of the new compounds 1 and 2 were established by detailed spectroscopic data analysis and ECD computational methods. The inhibitory effects of compounds 1–6 against NLRP3 inflammasome activation as well as the cytotoxic activities of compounds 1–4 against MCF-7 and HCT-116 cancer cell lines were investigated. Compound 3 showed weak cytotoxicity against HCT-116 (IC50 = 55.747 ± 3.212 μM).

Similar content being viewed by others
References
J. J. Magadula, J. Med. Plants Res., 8, 489 (2014).
R. M. Ngoumfo, G. E. Ngounou, C. V. Tchamadeu, M. I. Qadir, C. D. Mbazoa, A. Begum, F. N. Ngninzeko, D. Lontsi, and M. I. Choudhary, J. Nat. Prod., 71, 1906 (2008).
M. Tanjung, D. Mujahidin, E. H. Hakim, A. Darmawan, and Y. M. Syah, Nat. Prod. Comm., 5, 1209 (2010).
M. Tanjung, E. H. Hakim, and Y. M. Syah, Chem. Nat. Compd., 53, 215 (2017).
T. Peresse, G. Jezequel, P. M. Allard, V. C. Pham, D. T. M. Huong, F. Blanchard, J. Bignon, H. Levaique, J. L. Wolfender, and M. J. Litaudon, J. Nat. Prod., 80, 2684 (2017).
T. Wang, B. Liu, S. Hua, T. Li, and G. Chen, J. Chin. Herb. Med., 31, 372 (2008).
T. Wang, B. Liu, M. Ji, and G. Chen, J. Nat. Sci., 22, 49 (2009).
T. Wang, L. Huang, S. Zhao, Z. Chen, and G. Chen, J. Chin. Pharm., 21, 1392 (2010).
I. Zakaria, N. Ahmat, F. M. Jaafar, and A. Widyawaruyanti, Fitoterapia, 83, 968 (2012).
P. Luo, Z. Y. Yin, Z. J. Sun, H. Chen, C. J. Li, H. H. Zhou, Q. Gu, and J. Xu, Fitoterapia, 129, 162 (2018).
W. Xu, G. Zhou, and X. Yao, Nat. Prod. Res. Dev., 22, 945 (2010).
M. Maver, E. F. Queiroz, J. L. Wolfender, and K. Hostettmann, J. Nat. Prod., 68, 1094 (2005).
H. D. T. Mai, T. P. Toan, G. T. Huu, T. N. Le, V. T. K. Oanh, N. T. M. Hang, H. T. Thu, V. M. Chau, M. Litaudon, and V. C. Pham, Nat. Prod. Res., 34, 2772 (2020).
P. Basabe, M. de Roman, I. S. Marcos, D. Diez, A. Blanco, O. Bodero, F. Mollinedo, B. G. Sierra, and J. G. Urones, Eur. J. Med. Chem., 45, 4258 (2010).
K. Mukaide, S. Honda, B. Vongsak, and S. Kumazawa, Phytochem. Lett., 43, 88 (2021).
L. Siracusa, F. Cimino, A. Saija, and G. Ruberto, Advan. Chem. Res., 44,133 (2018).
Z. Gao, Z. Wei, J. Zhang, and Y. Su, Chem. Nat. Compd., 57, 650 (2021).
S. B. Banerjee, Eur. J. Biom. Pharm. Sci., 8, 215 (2021).
H. Joshi, V. Gajera, and A. Katariya, Int. J. Pharm. Sci. Res., 12, 3567 (2021).
H. Lee, S. Lee, J. Y. Baek, C. S. Seo, H. Yun, S. N. Kim, K. S. Kang, and Y. K. Choi, Bioorg. Med. Chem. Lett., 40, 127919 (2021).
H. Abd el-Fattah, A. Gohar, S. el-Dahmy, and A. Hubaishi, Acta Pharm. Hung., 64, 83 (1994).
Y. Li, Z. Yu, D. Zhao, and D. Han, Life Sci., 277, 119483 (2021).
J. L. Tian, T. L. Liu, J. J. Xue, W. Hong, Y. Zhang, D. X. Zhang, C. C. Cui, M. C. Liu, and S. L. Niu, Ind. Crop Prod., 138, 111445 (2019).
P. C. Zhang, S. W. Y. Wang, R. Y. Chen, and D. Q. Yu, J. Nat. Prod., 64, 1206 (2001).
D. B. Pu, X. J. Zhang, D. W. Bi, J. B. Gao, Y. Yang, X. L. Li, J. Lin, X. N. Li, R. H. Zhang, and W. L. Xiao, J. Nat. Prod., 83, 2191 (2020).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox, Gaussian 09, Revision E. 01.Wallingford CT: Gaussian, Inc. (2010).
T. Bruhn, A. Schaumloeffel, Y. Hemberger, and G. Bringmann, Chirality, 25, 243 (2013).
D. B. Pu, T. Wang, X. J. Zhang, J. B. Gao, R. H. Zhang, X. N. Li, Y. M. Wang, X. L. Li, H. Y. Wang, and W. L. Xiao, RSC Adv., 8, 6425 (2018).
Acknowledgment
This work was financially supported by the National Natural Science Foundation of China (81860615, 82260682, 21762048, 81903541, and 22167021), Applied Basic Research Foundation of Yunnan Province (2018FY001, 2018FA048, 202101AS070022, and 202101AT070195), the Program for Changjiang Scholars and Innovative Research Team in University (IRT_17R94), Project of Innovative Research Team of Yunnan Province (202005AE160005), and Yun Ling Scholar Project to Wei-Lie Xiao, National Key R&D Program of China (2019YFE0109200), and a grant (No. 2021FK004) from YNCUB. Authors thank the Advanced Analysis and Measurement Center of Yunnan University for the sample testing service.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2023, pp. 42–46.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaunda, J.S., Chen, YS., Xu, YJ. et al. Two New Prenylated Flavonoids from the Leaves of Macaranga pustulata. Chem Nat Compd 59, 46–51 (2023). https://doi.org/10.1007/s10600-023-03914-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-023-03914-y